Issues raised by medical practitioners in New Zealand concerning
COVID vaccines
Report for Medsafe: Professor P McIntyre, University of Otago & Dr Ian Town, Chief Science
Advisor, Ministry of Health
Background
A group of medical practitioners, (‘NZ Doctors’, hereafter referred to ‘the authors’) registered with
the Medical Council of New Zealand, has raised a number of concerns in communications with the
Medical Council and Medsafe. These concerns are under the broad headings of exaggeration of the
severity of the disease (COVID-19) caused by SARS-CoV-2 infection and the safety and efficacy of
vaccines in comparison to therapeutic agents. The issues raised are considered sequentially below.
Exaggerated disease burden
The authors assert that it is becoming increasingly clear that the all-age infection fatality rate (IFR) of
COVID-19 is similar to seasonal influenza. They cite a review by Professor John Ioannidis (Stanford
University) from October 2020 (Ref 1), as well as his summary of six reviews including his own from
February 2021 in support of this contention (Ref 2). However, a close reading of these two papers shows
they are primarily devoted to methodological issues important for critical interpretation of the IFR
rather than comparison of IFRs for COVID-19 and influenza. Calculation of the IFR is dependent on the
accuracy of its numerator (estimated deaths, derived by various means) and its denominator
(estimated infections, mostly derived from population-based serological studies). Both measures are
subject to several potential biases that Professor Ioannidis enumerates (Refs 1,2), resulting in substantial
uncertainty. Although it is true that non-pharmacological interventions (NPIs), such as border closure
and physical distancing/mask-wearing, will reduce IFR if they are successfully applied to shield high-
risk vulnerable populations, the authors do not justify comparison of COVID-19 IFR with seasonal
influenza against which comparable NPIs are not applied. Similarly, while it is also true that in-hospital
mortality from COVID-19 in highly resourced settings has reduced considerably with increasing
experience in management of the disease and its complications (Ref 3), this does not directly address
the issue of comparative mortality with influenza.
Comparison with influenza
A recent national study from France presents a direct comparison between all patients hospitalised
for COVID-19 from 1 March to 30 April 2020, and all patients hospitalised for influenza between
1 December 2018 and 28 February 2019 (Ref 4). In-hospital mortality for COVID-19 was about three-fold
1
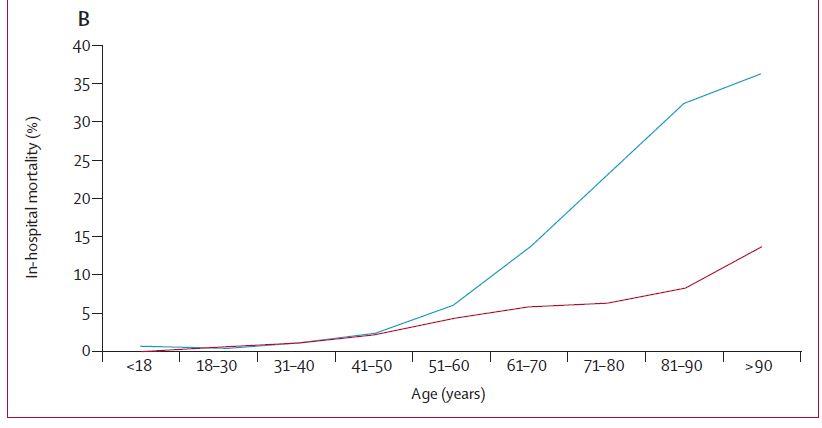
that of influenza (16·9 percent of 89,530 vs 5·8 percent of 45,819). In-hospital COVID-19 mortality
increased much more steeply with age over 50 years than influenza – age-standardised mortality ratio
was 2·82. Figure 1 – taken from reference 4 – shows COVID-19 in blue and influenza in purple.
Figure 1: Intensive care support and mortality of patients hospitalised in France for COVID-19 or seasonal influenza, by age
at admission. Dates are for patients who were hospitalised for COVID-19 between 1 March and 30 April 2020 and for patients
who were hospitalised for seasonal influenza between 1 December 2018 and 28 February 2019.
At the global level, the most recent modelling of deaths due to influenza estimated that 390,000
deaths occurred per year, with an upper limit of about 520,000 (Ref 5). This compares with the
(acknowledged minimum) estimate for COVID 19 of around 3.5 million deaths to 30 May 2021 (Ref 6).
Safety and efficacy of COVID-19 vaccines
Under-reporting of vaccine adverse effects
The authors assert they are aware of serious vaccine adverse events following Comirnaty vaccine that
‘do not seem to be in the public domain’ and that Medsafe should ‘carry out its own assessment of
deaths being reported by CDC/VAERS, Eudra Vigilance early warning mechanisms’.
Response:
a. Deaths occurring after receiving a COVID-19 vaccine.
To assess this, it is necessary to be aware of the strengths and limitations of passive reporting of
adverse events occurring after receiving a vaccine or vaccines. The VAERS safety database,
maintained by the US Centers for Disease Control (CDC), is the national passive adverse event
reporting system for the United States. VAERS had 2,342 deaths among the 56,869 adverse events
following COVID vaccines reported to it from 14 December 2020 to 1 April 2021. At first glance, it
is understandable this number of reported deaths appears alarming and the CDC website on
31 May had the number of deaths reported at 4,863. However, this must be viewed in context.
First, more than 285 million doses of COVID-19 vaccines had been administered by 24 May in the
United States. Second, a large proportion of those receiving vaccines during the first part of the
2
US rollout were older than 65 years, among whom deaths from a variety of causes would be
expected to occur with or without vaccination. The authors assert there has been an ‘apparent
surge in deaths and disability following vaccination, often patients below 50 who are extremely
unlikely to die from COVID-19’ but do not provide any evidence to support this statement.
VAERS is an open system, with any member of the public able to report. However, just as occurs
with the Centre for Adverse Drug Reporting (CARM) in New Zealand, CDC and FDA physicians
review each case report of death as soon as it is notified to VAERS, and then request medical
records to further assess reports. CDC, in its most recent summary of reported deaths, states
‘review of available clinical information including death certificates, autopsy, and medical records
revealed no evidence that vaccination contributed to patient deaths’ (Ref 7).
Enhanced surveillance in place in New Zealand for the COVID-19 vaccine programme includes
establishment of an Independent Safety Monitoring Board that has eight members representing
a range of clinical and statistical specialties and a consumer representative (Ref 8). Any unexplained
death occurring after receipt of a COVID-19 vaccine will be reviewed in detail by the ISMB
. The
Ministry of Health has also agreed to implement a cellphone-based application that will enquire
about adverse events directly with vaccine recipients.
b. Deaths prevented by COVID-19 vaccines in persons over 60 years of age – United Kingdom.
In the United Kingdom (UK), COVID-19 vaccines were rolled out progressively, starting with people
over 80 years, then those 70-79 years, then those 60-69 years. Self-evidently, these age groups
have high rates of death from a number of causes. Figure 2 shows data from Public Health England
to the end of March 2021 with actual numbers of deaths observed (shown in blue) compared with
numbers of deaths expected in these age groups if COVID-19 disease trends in the UK population
had continued but in the absence of vaccination (shown in ochre). These graphs started to diverge
within weeks of the UK vaccine rollout commencing and continued to diverge over the following
three months (Ref 9).
3
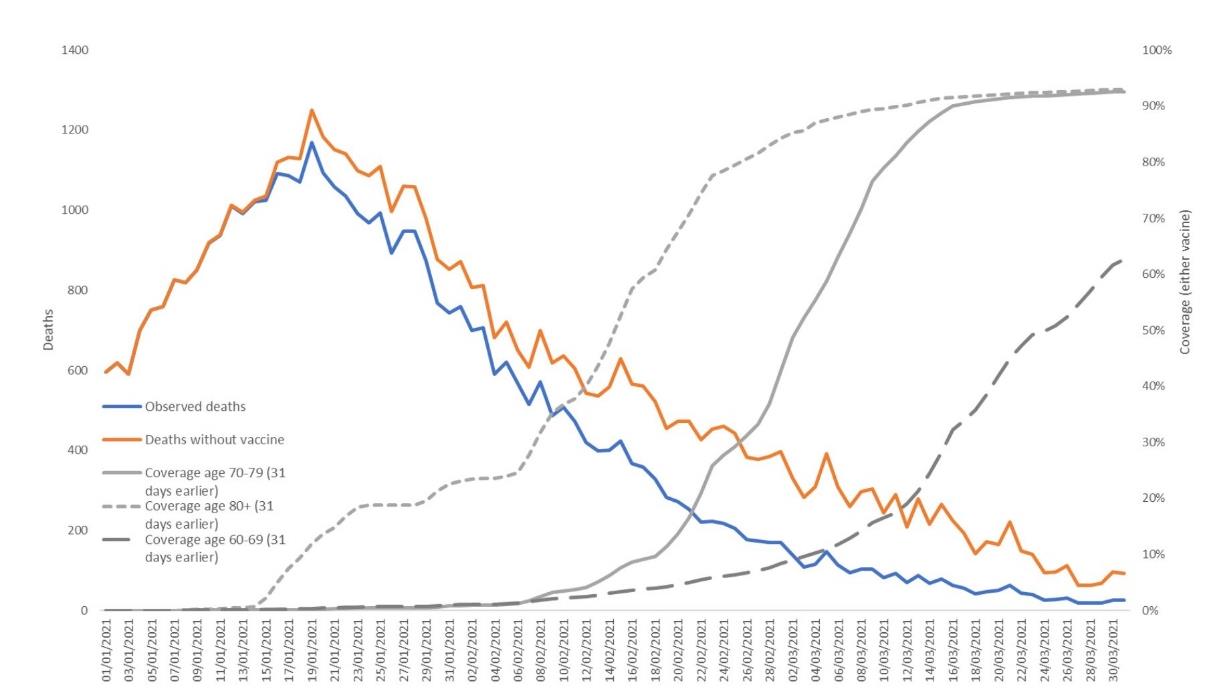
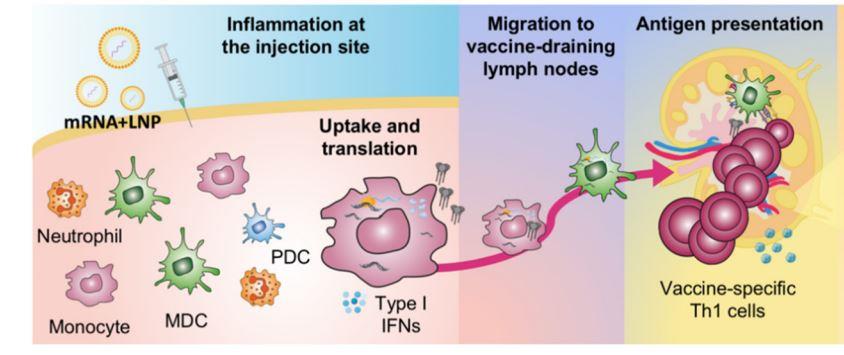
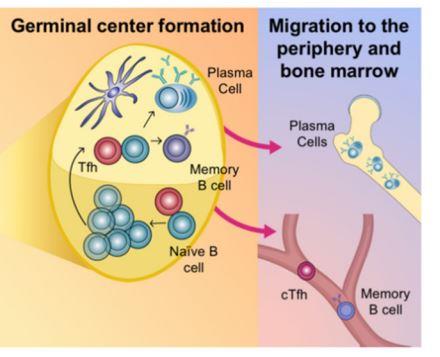 Figure 2: Daily Observed COVID-19 deaths aged 60+ and expected numbers in the absence of vaccination using the back-
Figure 2: Daily Observed COVID-19 deaths aged 60+ and expected numbers in the absence of vaccination using the back-
calculation method.
Other assertions about potential adverse effects
c. Concern that permanent alteration of DNA may occur.
Response:
Messenger RNA is unstable, which is why it must be stored at very low temperatures prior to use.
As mRNA does not enter the nucleus and is rapidly broken down by the cell after protein
transcription has occurred, it is unable to alter DNA (Ref 10). This is shown in the graphic below (Ref 11).
d. Concern about ‘the potential for immune suppression and exaggerated activation, as per previous
unsuccessful coronavirus vaccine animal trials, from pathogenic priming, antibody dependent
enhancement and vaccine interference. Comirnaty underwent no such animal trials’.
Response:
While it is true that there were concerns about heightened immune response to SARS-CoV-2 virus
infection post COVID-19 vaccines, based on data from experimental animals with SARS-CoV-1
vaccine development, there has been no evidence to support this in pre-clinical animal studies or
in humans. Pre-clinical trials for Comirnaty are well described in the literature (Ref 11). Studies of
4
health care workers recurrently exposed to SARS-CoV-2 infection in the UK (the SIREN study) have
demonstrated no adverse outcomes following exposure to infection in vaccinated participants
(Ref 12). There will be continued follow-up of both vaccine trial participants and cohorts such as the
SIREN study to provide longer-term monitoring for any evidence of enhanced disease among
COVID-19 vaccine recipients on re-exposure to SARS-Co-V-2 infection.
e. Concern about the spike protein causing endothelial cell mitochondrial damage.
Response:
The authors refer to a paper by Lei et al entitled “SARS-CoV-2 Spike Protein Impairs Endothelial
Function via Downregulation of ACE2” (Ref 13) with reference to direct damage to endothelial cells
from spike protein. In this study a SARS-CoV-2 pseudovirus, producing spike protein, was
administered directly into the trachea of the Syrian Hamster, an experimental animal shown to be
especially susceptible to SARS-CoV-2 infection (Ref 13). The study demonstrated a potential
mechanism for COVID-19-associated vascular disease via the spike protein via a mechanism
comparable to infection, not vaccination. In human mRNA vaccine recipients, spike protein is
produced intracellularly through mRNA transcription in the ribosome and then displayed on the
surface of the antigen presenting cell to induce B cells to produce antibody to spike protein (see
graphic above and reference 11) – this is entirely different and protective.
f. Concern about ‘the concept of shedding (presumably of spike protein) after vaccination, affecting
unvaccinated bystanders, particularly in the form of hormonal disruption and/or clotting
abnormalities in females of all ages’.
Response:
There is no evidence of shedding of spike protein or other viral proteins produced through vaccine
responses and no evidence of bystander infection. As Comirnaty is a non-live vaccine, this concern
is not scientifically credible. Inclusion in the Pfizer trial protocol of protection advice for both
investigators and close partners of vaccinees is a routine part of good clinical practice guidelines
for phase 1 and 2 studies and does not imply any concern about transmission.
g. Concern that ‘through molecular mimicry of syncitin-1, and with reports of increased miscarriages,
there is concern in pregnancy and for fertility’.
Response:
Numerous internet posts have falsely claimed that mRNA vaccines used against COVID-19 target
a protein called syncytin-1, which is needed for placental formation and successful pregnancies.
Many of these posts also baselessly imply that the vaccines will make people infertile. The origin
of these claims is uncertain as syncytin-1 is not contained in the SARS-CoV-2 spike protein and the
5
SARS-CoV-2 spike protein and syncytin-1 are not very similar. The protein targeted by their vaccine
only shared a sequence of four amino acids with syncytin-1, which is made up of 538 amino acids.
This is explained in detail in an article for the Conversation by Catherine Thornton, a Professor of
Human Immunology at Swansea University. (Footnote 1)
h. Concern about ‘the potential for autoimmune reactions, or impaired or exaggerated infection
responses, years after vaccination that short or even medium-term trials would not disclose’.
Response:
Although triggering of adverse immune responses in individuals who have significant autoimmune
disease cannot be excluded, based on current evidence the Australasian Society for Clinical
Immunology and Allergy (ASCIA) recommends that patients with autoimmunity or severe allergy
who are requiring specific therapies should not discontinue these and are recommended to
receive COVID-19 vaccines(Footnote 2). Very recently, early results from work in mice has suggested
that mRNA vaccines may offer benefit in the treatment of multiple sclerosis (Ref 14).
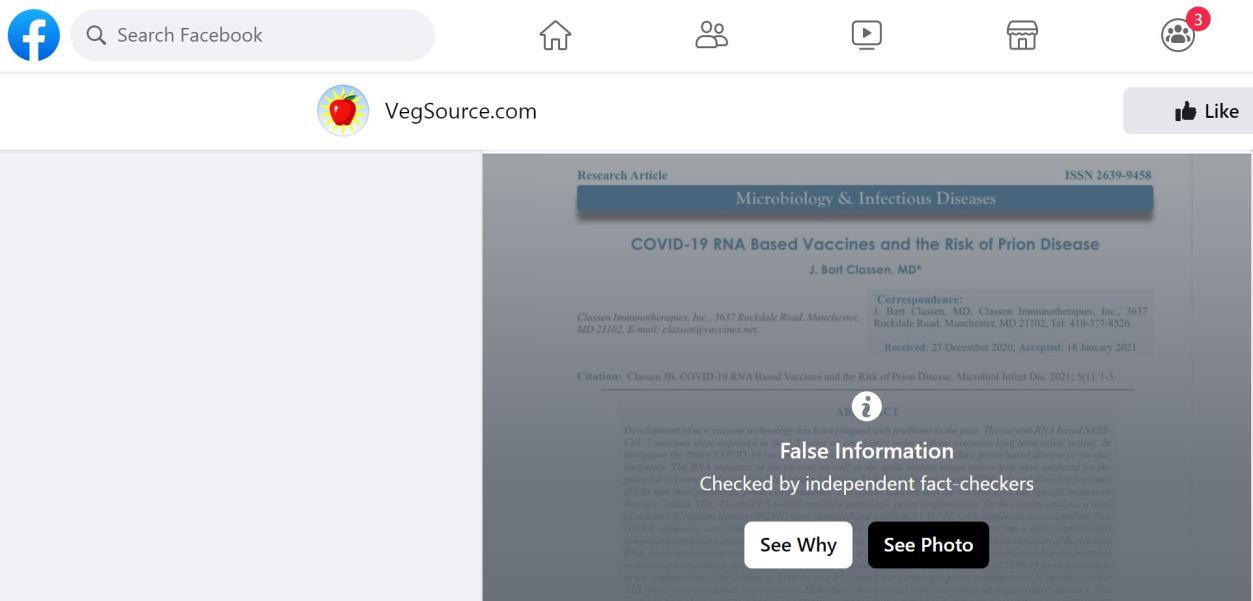
i. Concern about ‘the potential for spike protein-as-prion disease’ in the distant future.
Response:
This assertion was reviewed by USA Today through interviews with experts including Dr Angela
Rasmussen, a virologist affiliated with Georgetown University. She stated: ‘the publication which
gave rise to this theory (Classen JB COVID-19 RNA Based Vaccines and the Risk of Prion Disease)
in Microbiology & Infectious Diseases held no scientific weight at all’ and that the journal was ‘not
a reputable or reliable’. It is not Medline listed. Notably, Classen in 1999 claimed the influenza
vaccine
caused type 1 diabetes, a
claim disproven by Johns Hopkins University's Institute for
Vaccine Safety. This critique was sufficient for VegSource.com to rate the claim as false on
Facebook (see screen shot below).
1https://theconversation.com/covid-19-vaccines-do-not-make-women-infertile-153550
2https://www.allergy.org.au/images/stories/pospapers/ASCIA_HP_Guide_Immunodeficiency_Autoimmunity_
COVID-19_Vaccination_2021-04-14.pdf
6
 j. Concern that ‘vaccinated people may not have prolonged or broad-based immunity, including to
the many new variants of concern, and have higher death rates when exposed’.
Response:
j. Concern that ‘vaccinated people may not have prolonged or broad-based immunity, including to
the many new variants of concern, and have higher death rates when exposed’.
Response:
Knowledge about immunity from variants of concern is rapidly evolving, but the available
information for all vaccines in current use suggests that protection against severe disease to
variants is well-preserved in both vaccine recipients and persons who have recovered from proven
SARS-CoV-2 infection. See reference to the SIREN study (see section d above) (Ref 12).
k. Concern that ‘the symptoms, oxidative stress and inflammatory phenotype of “long covid”
syndrome may be produced solely by the spike protein, without presence of whole virus’.
Response:
Knowledge about the pathogenesis and clinical course of long COVID is evolving. To the extent
that it is due to spike protein, effective vaccines that induce potent immune responses to spike
protein would be expected to prevent it.
l. Concern that ‘the effects on children (two reports on VAERS of deaths of young children in clinical
trials), pregnancy, fertility, the elderly and infirm, interactions with medications, other chronic
conditions, have not been studied’.
Response:
It is true that evidence is only accumulating now about COVID-19 vaccines including Comirnaty
when given to people with a range of chronic conditions including immunocompromising
conditions, pregnant women and those who are very frail. However, in the context of countries
where there is community transmission of SARS-CoV-2 and substantially increased risk of severe
disease in these groups, the benefits of vaccination have been universally determined to greatly
7
exceed the risks. After hundreds of millions of doses, no evidence of adverse effects has emerged.
However, particularly in persons living with HIV or who have other forms of immune suppression,
vaccine responses and protection may be reduced. Vaccine trials in children underway or just
completed have shown strong immune responses and evidence of significant protection. Children
from 12 to 16 years, although having a low risk of severe disease, have a higher risk of infection
and severe disease compared with younger children, especially if they have comorbidities
including obesity (Ref 4). With respect to interpreting deaths among children in clinical trials see
sections a and b above.
Therapies for COVID-19 that may prevent or successfully treat disease
The authors assert that the notion that vaccination is New Zealand’s best or only hope for beating
COVID-19 is being undermined by strong evidence in favour of therapies including ivermectin,
hydroxychloroquine plus zinc and vitamin D. They go on to assert that mRNA vaccines against COVID-
19 represent ‘novel technology gene therapies’, and are, as such, more akin to a medical device than
a vaccine. Based on these considerations, their contention that COVID-19 is no more severe than
seasonal influenza, and what they believe to be the proven benefits and superior safety of the
therapeutic alternatives listed, they argue for suspending the COVID-19 vaccine programme pending
further evidence of its relative safety and efficacy.
Response:
Reasons to reject the contentions that mRNA vaccines represent gene therapy, and that COVID-19 is
equivalent in severity to seasonal influenza, have been discussed above.
Therapies for COVID-19 disease and its complications are of high ongoing importance, as it is inevitable
that some individuals will develop disease despite vaccination or because they have not received
vaccine. Therapies are continuously and rigorously reviewed in ‘living guidelines’, a process sponsored
by WHO, with reporting summarised by the British Medical Journal under its ‘rapid recommendation’
series see below.
8

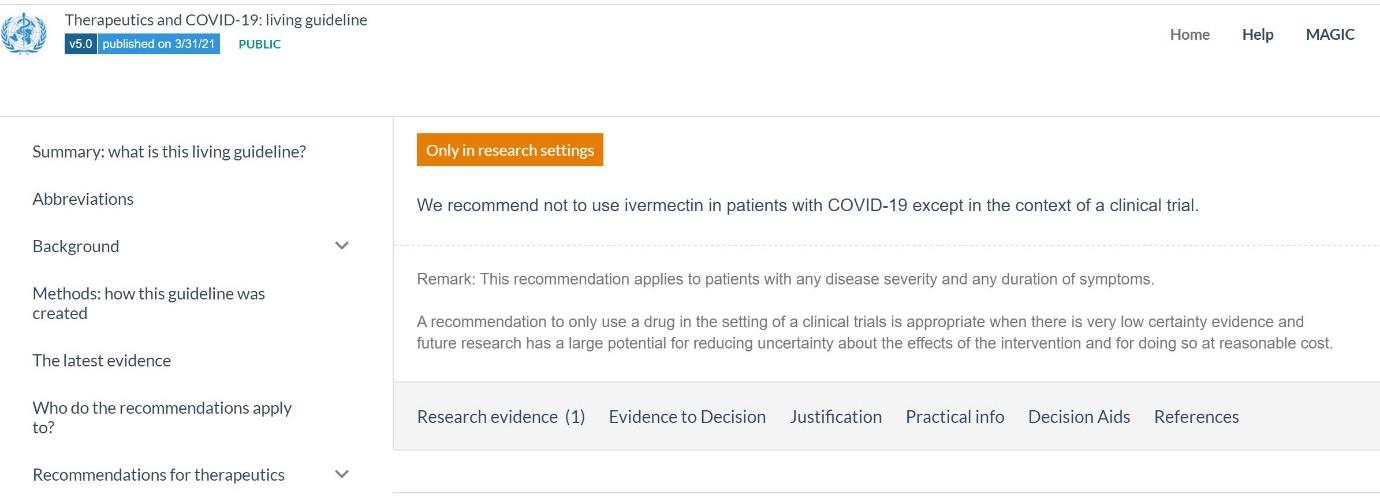
Currently, only dexamethasone is graded as ‘recommended’ and this recommendation is restricted to
persons with severe or critical disease. Remdesivir is graded as ‘to be considered’. Ivermectin is
deemed to be only appropriate for use in the context of an approved clinical trial, given substantial
uncertainty about its benefit:risk ratio (see below). The living guideline website provides a detailed
rationale for this classification based on recommendations of a diverse panel of advisors including lay
representatives. Even if additional therapies including ivermectin meet criteria for approval as
therapies for COVID-19, this will still be clearly less preferable than prevention of disease by the range
of highly effective vaccines available. No one would argue that treatment of meningococcal disease
with penicillin was to be preferred to prevention of meningococcal disease by vaccination, as
exemplified by the 1990s experience with epidemic meningococcal disease in New Zealand.
The weight of medical and scientific opinion
The authors express concern that eminent 'insider' scientists with unique expertise and insights are
sounding alarms, risking their careers and reputations to draw attention to important concerns, and
have knowledge and views that should make us pause first, then investigate further, ‘with the
9
implication that these individuals should be accorded whistleblower status’. The authors contend that
these scientists have expertise ‘which eclipses (that which) anyone in New Zealand has to offer’.
Response:
The doctors and scientists named are Pfizer's Peter Yeadon (this appears to be Dr Michael Yeadon),
Prof Sukharit Bhakdi, Dr Geert Vanden Bossche, Dr Peter McCulloch and Prof Dolores Cahill. Although
these individuals do have eminent careers, it is notable that none of them prior to 2020 had any
specific expertise in vaccines or immunisation apart from Dr Vanden Bossche. Vanden Bossche is a
veterinary doctor who has a Ph.D. in virology and has worked in senior management positions,
including senior program officer for vaccine discovery at the Bill and Melinda Gates Foundation.
However, his academic publications essentially stop in 1995, except fo
r one 2017 article about his
natural killer cell vaccine idea published in a journal belonging to a publishing group
sued by the
Federal Trade Commission for deceptive practices. Vanden Bossche’s recent advocacy against
vaccines has been compared to that of Andrew Wakefield. Yeadon worked in allergy and respiratory
disease with Pfizer, Bhakdi is a retired Thai-German microbiologist, Peter A. McCullough is a
cardiologist, and Dolores Cahill is a Professor of Medical Genetics. None of this disqualifies these
scientists from having views about COVID-19 vaccines but does call into question the depth of their
knowledge in this area.
Summary
While the concerns of NZ Doctors (the authors) are undoubtedly sincerely held, and based on
questions and issues of importance, we argue that their central contentions of over-stated disease
severity, substantive concerns about vaccine safety and that available therapies have equivalent or
superior risk-benefit profiles to COVID-19 vaccines, in particular Comirnaty vaccine, are not well
founded. Based on the evidence we cite here, and much broader high quality literature from clinical
trials and real world experience with COVID-19 vaccines, we strongly believe that their urgent rollout
is crucial to the welfare of the New Zealand population and should be strongly supported by all health
professionals.
10
link to page 2 link to page 2 link to page 2 link to page 2 link to page 4 link to page 4
List of Figures
Figure 1: Intensive care support and mortality of patients hospitalised in France for COVID-19 or
seasonal influenza, by age at admission. Dates are for patients who were hospitalised for COVID-19
between 1 March and 30 April 2020 and for patients who were hospitalised for seasonal influenza
between 1 December 2018 and 28 February 2019. ............................................................................... 2
Figure 2: Daily Observed COVID-19 deaths aged 60+ and expected numbers in the absence of
vaccination using the back-calculation method. .................................................................................... 4
11
References
1. Ioannidis JPA. Infection fatality rate of COVID-19 inferred from seroprevalence data. Bull World
Health Organ. 2021; 99:19-33F. doi: 10.2471/BLT.20.265892.
2. Ioannidis JPA. Reconciling estimates of global spread and infection fatality rates of COVID-19: An
overview of systematic evaluations. Eur J Clin Invest. 2021; 51:e13554. doi: 10.1111/eci.13554
3. Horwitz LI, Jones SA, Cerfolio RJ, Francois F, Greco J, Rudy B, Petrilli CM. Trends in COVID-19 Risk-
Adjusted Mortality Rates. J Hosp Med. 2021; 16:90-92. doi: 10.12788/jhm.3552
4. Piroth L, Cottenet J, Mariet AS, Bonniaud P, Blot M, Tubert-Bitter P, Quantin C. Comparison of the
characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: a nationwide,
population-based retrospective cohort study. Lancet Respir Med. 2021; 9:251-259. doi:
10.1016/S2213-2600(20)30527-0
5. Paget J, Spreeuwenberg P, Charu V, Taylor RJ, Iuliano AD, Bresee J, Simonsen L, Viboud C; Global
Seasonal Influenza-associated Mortality Collaborator Network and GLaMOR Collaborating
Teams*. Global mortality associated with seasonal influenza epidemics: New burden estimates
and predictors from the GLaMOR Project. J Glob Health. 2019; 9:020421. doi:
10.7189/jogh.09.020421
6.
COVID-19 Data Repository by the Center for Systems Science and Engineering (CSSE) at Johns
Hopkins University. https://github.com/CSSEGISandData/COVID-19
7. CDC: Selected adverse events reported after COVID vaccination accessed May 31st, 2021
https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/adverse-events.html
8. NZ
Doctor
https://www.nzdoctor.co.nz/article/undoctored/covid-19-independent-safety-
monitoring-board
9. Public Health England Impact of COVID-19 vaccines on mortality in England December 2020 to
March 2021 Accessed May 31st 2021 at
https://assets.publishing.service .gov.uk/
government/uploads/system/uploads/attachment_data/file/977249/PHE_COVID-
19_vaccine_impact_on_mortality_March.pdf
10. CDC: Understanding mRNA COVID vaccines Accessed May 31st 2021
at https://www.cdc.gov /
coronavirus/2019-ncov/vaccines/different-vaccines/mrna.html
11. Cagigi A, Loré K. Immune Responses Induced by mRNA Vaccination in Mice, Monkeys and Humans.
Vaccines (Basel). 2021; 9:61. doi: 10.3390/vaccines9010061.
12
12. Hall VJ, Foulkes S, Saei A, Andrews N, Oguti B, Charlett A et al SIREN Study Group. COVID-19
vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine
against infection (SIREN): a prospective, multicentre, cohort study. Lancet. 2021;
397(10286):1725-1735. doi: 10.1016/S0140-6736(21)00790-X.
13. Lei Y, Zhang J, Schiavon CR et al SARS-CoV-2 Spike Protein Impairs Endothelial Function via
Downregulation of ACE2. bioRxiv [Preprint]. 2020 Dec 4:2020.12.04.409144. doi:
10.1101/2020.12.04.409144. Update in: Circ Res. 2021 Mar 31
14. Wardell CM, Levings MK. mRNA vaccines take on immune tolerance. Nat Biotechnol. 2021
Apr;39(4):419-421. doi: 10.1038/s41587-021-00880-0.
13






