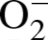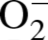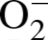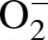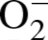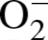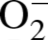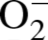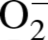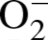NIH Public Access
Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
NIH-PA Author Manuscript
Published in final edited form as:
Inhal Toxicol. 2008 July ; 20(9): 829–838. doi:10.1080/08958370801895121.
Acute Inhalation Exposure to Vaporized Methamphetamine
Causes Lung Injury in Mice
Sandra M. Wells and
Mary C. Buford
Center for Environmental Health Sciences, Department of Biomedical and Pharmaceutical
Sciences, University of Montana, Missoula, Montana, USA
Sarah N. Braseth and
James D. Hutchison
Montana State Crime Laboratory, Missoula, Montana, USA
Andrij Holian
Center for Environmental Health Sciences, Department of Biomedical and Pharmaceutical
Sciences, University of Montana, Missoula, Montana, USA
NIH-PA Author Manuscript
Abstract
Methamphetamine (MA) is currently the most widespread illegally used stimulant in the United
States. Use of MA by smoking is the fastest growing mode of administration, which increases
concerns about potential pulmonary and other medical complications. A murine exposure system
was developed to study the pulmonary affects of inhaled MA. Mice were exposed to 25–100 mg
vaporized MA and assessments were made 3 h following initiation of exposure to model acute lung
injury. Inhalation of MA vapor resulted in dose-dependent increases in MA plasma levels that were
in the range of those experienced by MA users. At the highest MA dose, histological changes were
observed in the lung and small but significant increases in lung wet weight to body weight ratios
(5.656 ± 0.176 mg/g for the controls vs. 6.706± 0.135 mg/g for the 100 mg MA-exposed mice) were
found. In addition, there was 53% increase in total protein in bronchoalveolar lavage (BAL) fluid,
greater than 20% increase in albumin levels in the BAL fluid, greater than 2.5-fold increase in lactate
dehydrogenase levels in the BAL fluid, and reduced total BAL cell numbers (approximately 77% of
controls). Levels of the early response cytokines tumor necrosis factor (TNF)-
α and interleukin (IL)-6
were dose-dependently increased in BAL fluid of MA-exposed mice. Exposure to 100 mg MA
significantly increased free radical generation in the BAL cells to 107–146% of controls and to
NIH-PA Author Manuscript
approximately 135% of the controls in lung tissue in situ. Together, these data show that acute
inhalation exposure to relevant doses of volatilized MA is associated with elevated free radical
formation and significant lung injury.
Methamphetamine hydrochloride (MA) is a substituted amphetamine with potent central
nervous system stimulant effects, and is currently the most widespread illegally used stimulant
in the United States (NIDA, 2006). Like cocaine, MA can be snorted, injected, ingested, or
smoked. According to the 2007 National Drug Threat Assessment Report by the National Drug
Intelligence Center, smoking is the fastest growing primary mode of administration. From 1993
to 2004, use of MA by smoking grew from 15% to nearly 60% (NDIC, 2006). Smoking MA
may result in more rapid addiction to the drug than snorting or injection because smoking
causes a nearly instantaneous, intense, and longer lasting high (NIDA, 2002). These factors
Copyright © Informa Healthcare USA, Inc.
Address correspondence to Sandra M. Wells, PhD, Center for Environmental Health Sciences, Department of Biomedical and
Pharmaceutical Sciences, University of Montana, Skaggs Building, Room 285C, Missoula, MT 59812, USA.
[email address].
Wells et al.
Page 2
may lead to increased rates of addiction that will further strain the resources of public health
agencies.
NIH-PA Author Manuscript
Studies of other inhaled drugs including marijuana and cocaine have shown that these
substances can cause a variety of pulmonary complications. Chronic marijuana use is
associated with increased prevalence of symptoms of chronic bronchitis, including chronic
cough, sputum production, wheeze, and increased outpatient visits for respiratory illness
(Bloom et al., 1987; Tashkin et al., 1987; Taylor et al., 2000; Taylor et al., 2002). Marijuana
use also affects airway pathology by causing lower-airway injury, inflammation, and
impairment in the antimicrobial and tumoricidal function of alveolar macrophages (Fligiel et
al., 1997; Roth et al., 1998). Alkaloidal cocaine (free-base or crack cocaine) smoking is
associated with a number of acute pulmonary complications, including acute respiratory
symptoms, (Tashkin et al., 1992), severe exacerbations of asthma (Haim et al., 1995), and an
acute lung injury syndrome (Forrester et al., 1990).
Increasing evidence suggests that MA-induced neurotoxicity involves the production of
reactive oxygen and reactive nitrogen species (ROS and RNS), resulting in induction of
oxidative stress (Stephans & Yamamoto, 1994). Furthermore, there is evidence that free
radicals may play a role in amphetamine-related lung injury. In an isolated rat lung model,
Huang et al. demonstrated that the injury observed following amphetamine administration was
associated with overproduction of free radicals (Huang et al., 2002). Although most of the
NIH-PA Author Manuscript
reported side effects of smoking MA have been systemic, case reports of pulmonary
complications including acute noncardiogenic pulmonary edema (Nestor et al., 1989a,
1989b) and fatal pulmonary hypertension (Schaiberger et al., 1993) have been reported.
However, to date there are no published studies on the pulmonary effects of vaporized MA.
Based on the published results with other drugs and the case studies reported for MA, we
hypothesized that vaporized MA may cause lung injury accompanied with oxidant stress. The
present study was designed to test this hypothesis.
MATERIALS AND METHODS
Animals
BALB/
c mice used in these studies were bred and maintained in microisolator units in the
University of Montana specific pathogen-free animal facility. The mice were given water ad
libitum. Mice, 7–10 wk of age, were used for all animal experiments. All animal procedures
were approved by the University of Montana Institutional Animal Care and Use Committee
(IACUC).
NIH-PA Author Manuscript
Methamphetamine Exposure
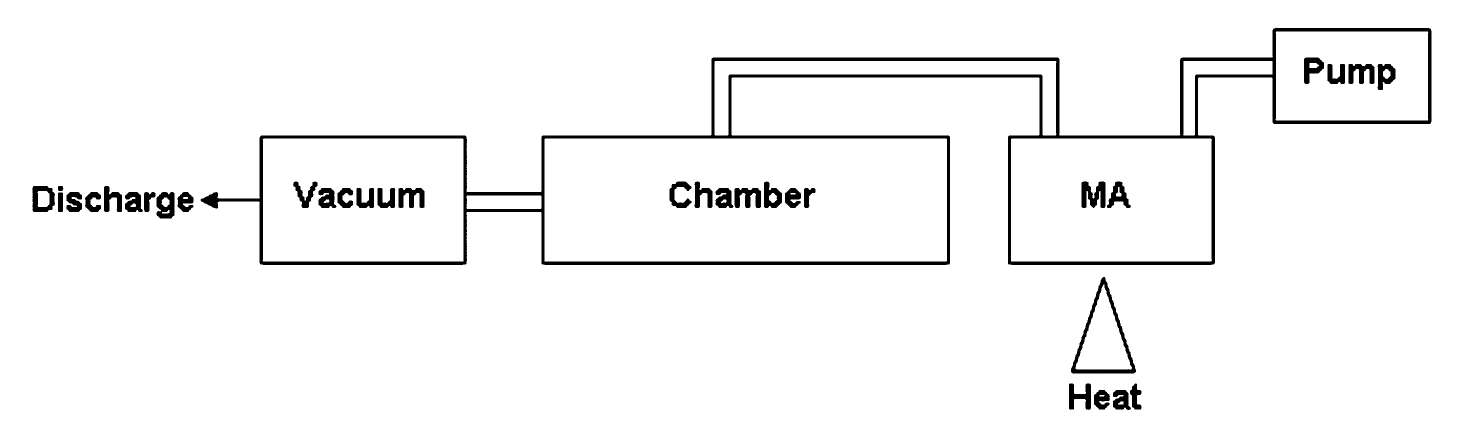
Mice were exposed to vapor from 25–100 mg heated MA utilizing the exposure system
described in Figure 1. The exposure chamber consisted of a 100-ml heating vessel above a
propane heat source connected to the animal chamber. The animal chamber was designed and
built by Dr. Lung-Chi Chen at New York University School of Medicine and holds up to 32
mice in individual compartments. Air was pumped into the heating vessel at a rate of 1.5 L/
min using a small pump connected to the top of the heating vessel. The air moved from the
heating vessel into the stainless-steel chamber and was distributed evenly throughout the
chamber. Air flow through the chamber was kept continuous with the use of a constant vacuum
of approximately 1.5 L/min. Temperature and humidity were both monitored inside the
chamber using a remote temperature/humidity probe and remained unchanged throughout the
exposures.
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 3
Mice were placed in the chamber and were allowed to acclimate for 5 min. The heat source
was ignited and the indicated amount of MA (Sigma Chemical, St. Louis, MO) heated for 20
NIH-PA Author Manuscript
min. Following the 20-min exposure, the heat source was turned off and the mice remained in
the chamber for an additional 5 min. Due to risk of low levels of MA exposures, control mice
were subjected to the same conditions as the exposed mice (e.g., transfer to and from the
exposure chamber), except for being placed inside the exposure chamber. All mice were
sacrificed approximately 3 h following initiation of exposure. Mice receiving the 100 mg dose
displayed self-injurious behavior. Therefore, the 3-h time point was chosen to allow for
maximal time between the exposure and assessments before significant injury occurred.
Determination of MA Plasma Levels
Mice were euthanized by a lethal injection of Euthasol. Blood was collected by cardiac bleeds
into 2.0-ml Vacutainer tubes containing sodium fluoride and potassium oxalate (Becton
Dickenson, Franklin Lakes, NJ). A basic, liquid—liquid extraction procedure was used to
extract MA and amphetamine from the mouse blood. Stock standards (10 ng/
μl, 1 ng/
μl, 0.1
ng/
μl) of MA were used as positive controls. An internal standard (1 ng/
μl) MA d-14 was also
prepared and 7.5
μl was added to all samples to aid in the quantification of MA. One half
milliliter of mouse blood was diluted to 1.0 ml by adding 0.5 ml deionized water. The standards
were prepared for quantification in 0.5 ml of negative control blood, and were also diluted to
1.0 ml using 0.5 ml deionized water. Extraction was done by addition of 1.0 ml of 0.5
N NaOH,
NIH-PA Author Manuscript
4.0 ml
n-butyl chloride, mixing for 2 min, and centrifugation for 3 min at 3400 rpm. The
supernatant was transferred to new tubes and dried at 37°C for 10 min following the addition
of 100
μl 1% methonolic hydrochloride. The dried samples were reconstituted with 100
μl 1%
formic acid in water, centrifuged at 14,000 rpm for 1 min, and supernatants were transferred
to vials for liquid chromatography—tandem mass spectrometric (LC/MS) analysis.
Chromatographic separation was achieved on a 4.6 mm × 150 mm, 3.5
μm Eclipse Plus C18
column (Agilent Technologies, Palo Alto, CA) at a column temperature of 55°C. The mobile
phase consisted of 1% formic acid in water and methanol and the flow rate was 1.00 ml/min
with a 20-min stop time and a 5-min post time. Signals were produced using selected ion
monitoring (SIM) parameters and an atmospheric pressure ionization—electrospray ionization
mode with positive polarity. SIM parameters were set to scan for ions at 136, 147, 150, and
164. The spray chamber parameters were 300°C gas temperature, 13.0 L/min drying gas, a
capillary voltage of 3.0 kV.
Collection of Bronchoalveolar Lavage Cells and Fluid
Lungs were removed with the heart and then lavaged with five 1.0-ml aliquots of cold
phosphate-buffered saline (PBS). The first 1.0 ml was centrifuged at 400 × g for 5 min and the
NIH-PA Author Manuscript
lavage fluid was saved for protein, cytokine, and lactate dehydrogenase (LDH) assessments.
The remaining 4 aliquots were centrifuged at 400 × g for 5 min and the cells from all 5 aliquots
were pooled. The cell pellet was resuspended in 1 ml PBS and a 40-
μl sample was counted
using a Z1 Coulter particle counter (Beckman Coulter, Fullerton, CA).
Determination of Protein Levels
Total bronchoalveolar lavage (BAL) fluid protein levels were analyzed for total protein in
triplicate using the Bio-Rad Laboratories protein assay (Hercules, CA) according to the
manufacturer’s instructions. Bovine serum albumin was used as the standard. Samples were
measured undiluted. Colormetric analysis was performed with the Spectra Max 340 plate
reader (GE Healthcare, United Kingdom) at 595 nm. Data are expressed as micrograms total
protein per milliliter of BAL fluid.
Mouse albumin levels in BAL fluid were measured in triplicate using a mouse albumin enzyme-
linked immunosorbent assay (ELISA) kit purchased from Bethyl Laboratories (Mont-gomery,
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
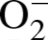
Wells et al.
Page 4
TX) according to the manufacturer’s instructions. Samples were diluted 1:200. Colormetric
analysis was performed with the Spectra Max 340 plate reader (GE Healthcare) at 450 nm.
NIH-PA Author Manuscript
Data are expressed as micrograms albumin per milliliter of BAL fluid. The detection limit for
this ELISA was 7 ng/ml.
Determination of Lung Weight to Body Weight (LW/BW) Ratios
Lung weight to body weight (LW/BW) ratios were determined by weighing whole mice and
whole excised lung. Only nonlavaged lungs were utilized for the determination of LW/BW
ratio. The LW/BW ratio is expressed in milligrams per gram.
Determination of LDH in BAL Fluid
LDH activity in the BAL fluid was determined by measuring the oxidation of lactate to
pyruvate, which then reacts with tetrazolium salt to form formazan (Decker & Lohmann-
Matthes, 1988), using an LDH-cytotoxicity assay kit by BioVision Research Products
(Mountain View, CA) according to the manufacture’s instructions. Absorbance was measured
by spectrophotometric analysis at 500 nm with the Spectra Max 340 plate reader (GE
Healthcare). Data are expressed as optical density at 500 nm.
Histology
NIH-PA Author Manuscript
Lungs from each mouse were inflation fixed with 3% paraformaldehyde and immersed in the
same fixative. Tissue was fixed overnight, washed with PBS, dehydrated, and embedded in
paraffin. Tissue sections (7
μm) were stained with hematoxylin—eosin (H&E) for histology.
Determination of BAL Fluid Cytokine Levels
BAL fluid from the first 1.0-ml aliquot was assayed for cytokines with commercially available
kits according to the manufacturer’s protocol. Interleukin (IL)-6 and interferon (IFN)-
α
measurements were determined by using Duo-set kits (R & D Systems). Samples were diluted
1:2. Colormetric analysis was performed with the Spectra Max 340 plate reader (GE
Healthcare) at 450 nm. Data are expressed as picograms per milliliter of retrieved culture
supernatant.
Measurement of ROS and RNS in BAL Cells
For analysis of intracellular
production (referred to as ROS) and intracellular ONOO−
production (referred to as RNS), dihydroethidium (DHE) and dihydrorhodamine 123 (DHR)
levels were determined as previously described (Haugen et al., 1999). Briefly, 1.5 × 105 cells
were incubated on ice with DHE (25
μM) or DHR (25
μM) for 20 min and analyzed using a
NIH-PA Author Manuscript
FACSCalibur (Becton Dickinson). Data were processed using the Cell Quest software (Becton
Dickinson). The membrane-permeable fluorescent dye DHE is oxidized in the presence of ROS
and fluoresces red (absorption/emission 518/605 nm) upon DNA intercalation. The membrane-
permeable nonfluorescent dye DHR is oxidized to the fluorescent product rhodamine, which
has absorption/emission spectra of 505/534 nm. Increased cellular fluorescence, as measured
by flow cytometry, indicates increased production of ROS or RNS as is reported as mean
channel fluorescence.
In Situ Detection of ROS
In situ production of ROS can be evaluated microscopically in frozen sections using DHE
(Miller et al., 1998; Paravicini et al., 2002; Grobe et al., 2006). Briefly, lungs were inflated
with 0.5 ml of a 1:1 ratio of Tissue-Tek OCT compound (Sakura Finetek USA, Torrance, CA)
and water. They were then frozen by placing immediately above liquid nitrogen until rigid and
then submerging in liquid nitrogen for 10 min, and they were stored at −80°C. Tissues were
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
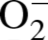
Wells et al.
Page 5
cryosectioned at 7
μm, collected onto Superfrost plus slides (VWR Scientific, West Chester,
PA), allowed to air-dry at room temperature, and stored at −80°C until used. Slides were placed
NIH-PA Author Manuscript
into PBS for 30 min at room temperature and then stained with DHE (10
μM) in PS for 30 min
in a moist chamber in the dark. The slides were rinsed extensively with PBS, coverslipped, and
imaged with a Kodak DC120 digital camera. DHE fluorescence was quantified with a
CompuCyte LSC laser scanning cytometer (CompuCyte, Cambridge, MA). The desired area
of analysis was located visually using epifluorescence visual microscopy on the instrument.
Scan areas were set and the scan initiated using the 20× objective and the argon laser operating
at 10 mW. Phantom contours (Megyeri et al., 2005) were used to obtain fluorescence
measurements of both airways and interstitial tissue. Long red fluorescence was collected with
a 650–700 nm bandpass filter/PMT set. Four different measurements were taken and averaged
for each animal. Maximum pixel median values and percent positive over background statistics
were used. Data were acquired and analyzed with WinCyte acquisition software (CompuCyte).
To confirm specificity of DHE for
, slides were also pretreated with polyethylene glycol
superoxide dismutase (PEG-SOD) (50 U/slide) before being stained with DHE as described
earlier. PEG-SOD was obtained from Sigma Chemical.
Statistical Analysis
The mean ± SEM was calculated for all samples and
p values were calculated using an unpaired
NIH-PA Author Manuscript
t-test or a one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison
to a single control group. For calculation of correlation between MA dose and plasma levels,
a Pearson correlation test was utilized. All data are expressed as mean × SEM. Less than 5%
probability of type I error was accepted as statistically significant.
RESULTS
Mouse Plasma MA Levels
To determine the relative exposure of the mice to the volatized MA, we first established whether
there was a correlation between the amount of MA that was heated and the levels of MA in
plasma of the exposed mice. We exposed mice to 25 mg, 50 mg, 75 mg, or 100 mg heated MA.
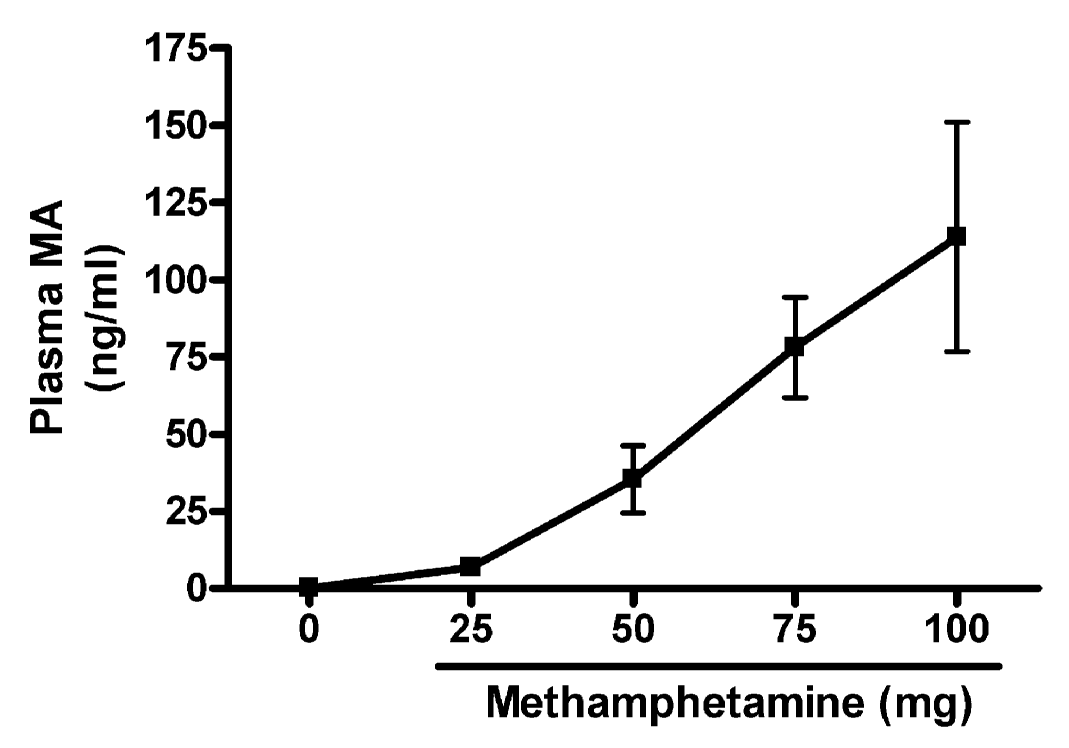
Plasma levels of MA for the exposed mice and the unexposed controls are shown in Figure 2.
We observed a relative linear increase in plasma MA levels following increasing doses of
heated MA. A significant (
p < .001) correlation between MA dose and plasma levels was
determined using a Pearson correlation test (
r = .7450).
Lung Injury Following Exposure to Vaporized MA
NIH-PA Author Manuscript
To explore the effects of vaporized MA in the lungs of mice following inhalation we assessed
several parameters of lung injury and inflammation following exposure to increasing MA
doses. Protein leak across the alveolar—capillary barrier can be determined by measurement
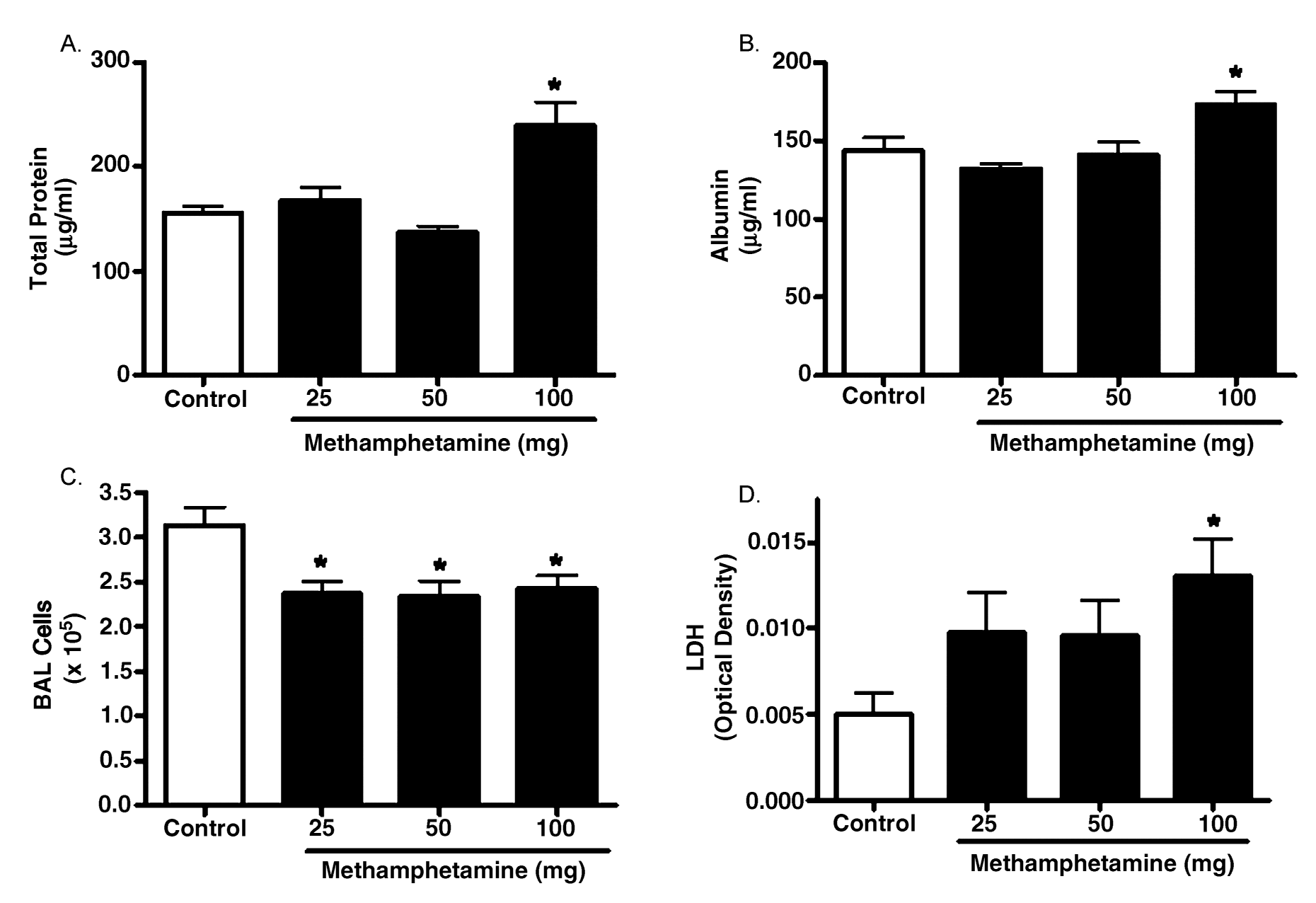
of total protein concentration in the BAL fluid (Kenyon et al., 2002). We measured total protein
levels in the BAL fluid of control and exposed mice (Figure 3A) and found a significant (53%)
increase in total protein in BAL fluid of animals exposed to the 100-mg dose of MA. In addition,
BAL fluid albumin levels were significantly increased in 100 mg MA-exposed animals (Figure
3B; 143.4 ± 8.738
μg/ml in controls vs. 173.0 ± 8.053
μg/ml in MA-exposed mice;
p < .05).
Finally, the lung wet weight/body weight ratio (LW/W) was assessed and was significantly
higher in the MA-exposed animals compared to controls (5.656 ± 0.176 mg/g for the controls
vs. 6.706 ± 0.135 mg/g for the MA-exposed mice;
n = 4,
p < .05).
BAL total cellularity was also determined and found to be mildly but significantly decreased
in animals exposed to all doses of MA (Figure 3C). At the 100-mg dose, exposed mice had a
total BAL cell number of 2.42 ± 0.151 × 105 cells vs. 3.13 ± 0.204 × 105 cells in the control
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
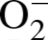
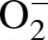
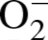
Wells et al.
Page 6
mice (
p <.05). In both control and MA-exposed mice, the cells present in the BAL were
predominantly macrophages and the BAL return was similar. To assess whether this decrease
NIH-PA Author Manuscript
in cell number could be loss through cell death, we assessed lactate dehydrogenase (LDH)
levels in BAL fluid. We observed a greater than 2.5-fold increase in BAL fluid LDH levels in
mice exposed to the highest dose of MA (Figure 3D). Lower doses of MA also increased BAL
fluid LDH; however, these increases were not significant.
MA-Induced Changes in Lung Histopathology
Histopathologic assessment of the lung was performed to assess whether the physiological
indices reported earlier in this article correlated with any morphological changes. For this
assessment, we used 100 mg dose of MA, since exposure to this level of MA resulted in plasma
MA levels below the mean MA plasma level of 730 ng/ml reported for human MA users
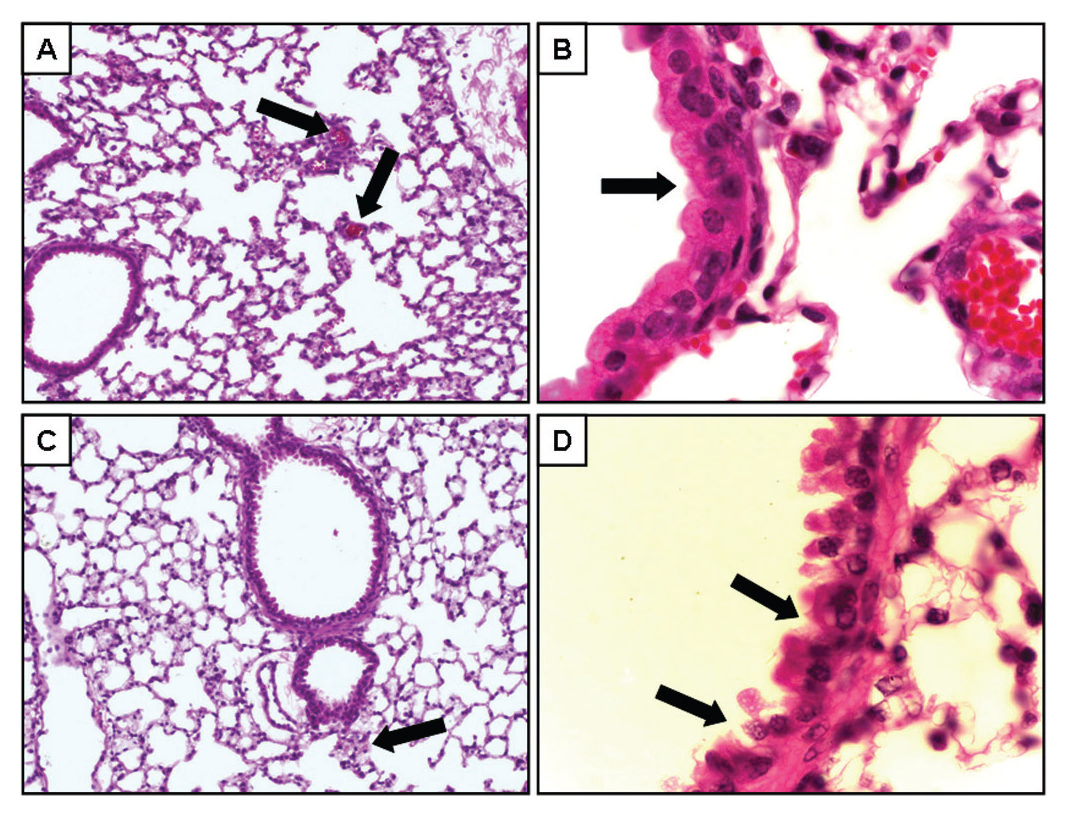
(Schwilke et al., 2006). Representative H&E sections from control (Figure 3, panels A—B)
and MA-exposed (Figure 3, panels C—D) mice indicate that inhalation of vaporized MA
appeared to have caused subtle but detectable lung injury (Figure 4). Compared to control mice
(panel A), there were fewer small arterioles in the peripheral lung tissue of MA-exposed mice
(panel C). In addition, MA exposure appears to result in damage to airway epithelial cells as
evidenced by exposed nuclei and exposed basement membrane (panel D). This is in contrast
to the epithelial cell layer in control mice, which was uniform and intact (panel B). Taken
together, these results indicate that exposure to relevant levels of MA resulted in detectable
NIH-PA Author Manuscript
lung injury.
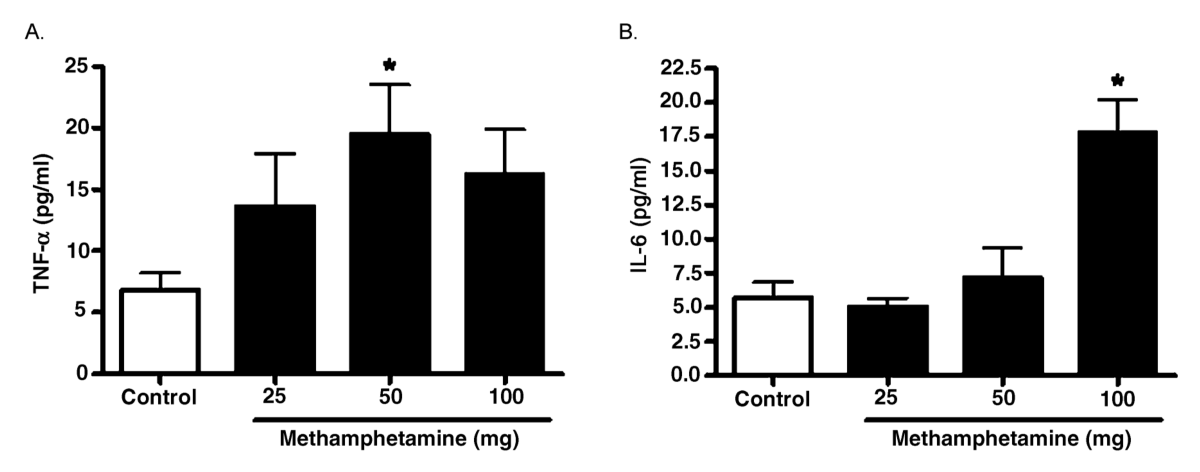
Early-Response Cytokines Following Acute Exposure to Vaporized MA
To determine whether the induction of lung injury correlated with increased expression of early
response cytokines, TNF-
α and interleukin IL-6 were measured in the BAL fluid of control
and MA-exposed mice (Figure 5). TNF-
α levels were significantly increased following
exposure to the 50 mg doses of MA. TNF-
α levels also appeared higher at the 100 mg dose of
MA; however, this increase did not reach statistical significance. IL-6 levels were only
significantly increased following exposure to 100 mg MA. Consistent with other agents that
cause lung injury, these results demonstrate that the MA-induced lung injury was associated
with rapid elevation of early response cytokines.
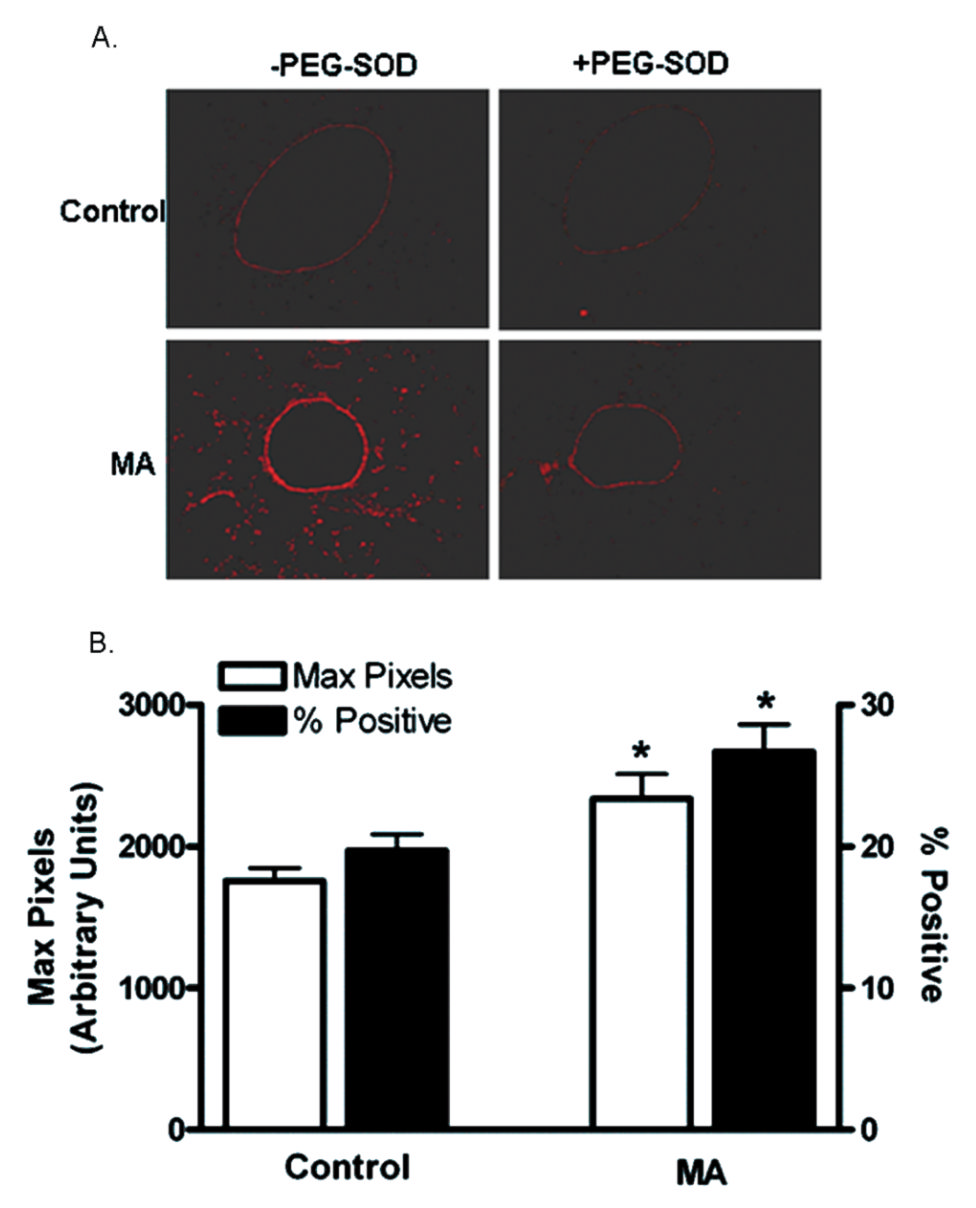
Oxidative Stress in Lung Cells and Tissue Following Exposure to Vaporized MA
To test for possible mechanistic explanations for MA-induced lung injury, we evaluated
whether the observed lung injury following exposure to 100 mg MA was associated with
increased oxidative stress. The fluorescent dyes DHE and DHR were utilized to detect
increased ROS and RNS, respectively, in the BAL cells and lung tissue from exposed mice.
NIH-PA Author Manuscript
The oxidation of DHE has been reported to be relatively specific for
with minimal oxidation
from H2O2 or ONOO− and has been used as a marker for
production (referred to as ROS)
(Benov et al., 1998; Fink et al., 2004). Peroxynitrite efficiently oxidizes the nonfluorescent
molecule DHR to the fluorescent product rhodamine. This method has been employed for
detection of ONOO− (referred to as RNS) and can react efficiently with ONOO− even at very
low concentrations (Kooy et al., 1994). As previously described (Haugen et al., 1999), DHE
and DHR fluorescence was measured by flow cytomentry in intact BAL cells to determine the
effect of MA exposure on the level of intracellular ROS and RNS levels (Table 1). The mean
channel fluorescence for both DHE and DHR was significantly increased in the BAL cells
from mice exposed to heated MA, indicating elevated intracellular ROS and RNS levels
following exposure.
The relative
levels in lungs in situ were also assessed utilizing DHE staining of frozen tissue
sections (Figure 6A). The regions of DHE staining correlated with the airway epithelial cells
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
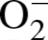
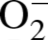
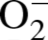
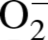
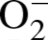
Wells et al.
Page 7
that were injured, as shown in Figure 4D. The intensity of DHE fluorescence was quantified
utilizing Laser Scanning Microscopy (Figure 6B). These studies revealed that the DHE
NIH-PA Author Manuscript
fluorescence was significantly higher (1756 ± 91.33 in controls vs. 2339 ± 172.5 in MA-
exposed;
n = 4,
p < .05) and there was an increase in the percent DHE-positive cells in mice
exposed to MA compared to unexposed controls (19.70% ± 1.167% in controls vs. 26.70% ±
1.950% in MA-exposed;
n =4,
p < .05).
It has been reported that concentrations higher than 1
μM DHE can result in
-independent
fluorescence from nonspecific ethidium cation binding to nuclear DNA, resulting in an
enhancement of fluorescence (Budd et al., 1997). Furthermore, DHE can be oxidized by routes
independent of
such as ferricytochrome
c oxidation in conditions where mitochondria are
the primary source
production or cytochrome
c release into the cytosol during apoptosis
(Green & Reed, 1998). Therefore, DHE oxidation was also assessed in the presence of PEG-
SOD (50 U/slide) to confirm that the increased DHE oxidation following exposure to MA was
due to
generation (Grobe et al., 2006). Pretreatment of tissue sections with PEG-SOD
significantly reduced the DHE fluorescence in both control and exposed animals, confirming
that this fluorescence is likely due to the presence of
(Figure 6A) rather than to nonspecific
DHE oxidation.
DISCUSSION
NIH-PA Author Manuscript
This study employed an MA inhalation exposure system to explore the acute pulmonary effects
of MA. To our knowledge, this is the first report of pulmonary effects in mice following
inhalation exposure to MA. We show that an acute exposure to MA causes lung injury and
increased production of free radicals in the lungs of exposed mice. It has been previously
reported that MA is readily vaporized upon heating, and that approximately 81–91% of
methamphetamine-HCl is recovered intact when heated at temperatures used to smoke the drug
(NIDA, 1991). Furthermore, in humans, MA is rapidly and well absorbed by inhalation of the
vapor (Cook et al., 1993). In our study, we observed dose-dependent increases in MA plasma
levels following exposure to the drug, suggesting that MA is also rapidly absorbed by inhalation
of the vapor in our animal exposure model. We measured a linear increase in MA plasma
concentrations following increasing doses of MA. In humans, several studies have
demonstrated that the fraction of MA excreted in urine decreases with increasing drug dose,
and the amount of MA in urine is not proportional to the dose of the drug absorbed (Cook et
al., 1993; Kim et al., 2004). This suggests that detoxification by renal clearance might become
saturated with only moderate doses of MA.
Because the lungs are the principal organ exposed to the products of smoked substances, it is
NIH-PA Author Manuscript
expected that pulmonary complications would be prominent among the health consequences
of smoking. The present results show that acute inhalation of MA caused increased
microvascular permeability as measured by protein and albumin levels in the BAL fluid and
lung weight gain. Furthermore, increased LDH levels in the BAL fluid suggest that the decrease
is likely through cell death. There was a decrease in lavagable lung cells that may be due to
MA-induced cytotoxicity. Alternatively, this could be due to increased cell adherence
preventing macrophages from being lavaged or increased trafficking of the cells out of the
lung. Further studies will be required to confirm the cause of decreased BAL cellularity. It is
important to note that these acute effects on the lungs occurred at MA plasma levels below
those seen in MA users.
This study focused on acute response of the lung to MA smoke exposure. Preliminary studies
of mice exposed to the 50-mg MA dose at up to 24 h postexposure suggest that any detectable
injury resolves (data not shown). The 100-mg MA dose resulted in self-injurious behavior not
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 8
allowing long-term evaluation beyond the 3-h time point. However, it will be important to
evaluate repeated and long-term MA exposure at lower doses to mimic human exposures.
NIH-PA Author Manuscript
These findings are consistent with observed effects of other smoked substances including
tobacco, marijuana, and alkaloidal cocaine. Habitual marijuana use in humans is associated
with histopathologic evidence of airway injury (Roth et al., 1998) and increased frequency and
severity of epithelial inflammation and loss of ciliated surface epithelium (Gong et al., 1987;
Fligiel et al., 1997). Similarly, long-term effects of tobacco smoking, with or without other
substances, include increases in alveolar epithelial permeability (Susskind et al., 1991; Tashkin
et al., 1997). Although there are no data available on the lung patho-physiology of MA
inhalation exposures, it has been well documented that MA can cause neurotoxic damage,
including degeneration of monoaminergic terminals and apoptosis of non-monoaminergic cells
in the brain (Preston et al., 1985; Ricaurte & McCann, 1992; Deng et al., 1999, 2001; Jayanthi
et al., 2001). Furthermore, numerous studies indicate that MA-induced neurotoxic damage is
due, at least in part, to the formation of ROS (LaVoie & Hastings, 1999; Davidson et al.,
2001), and MA-induced neurotoxicity can be attenuated in transgenic mice that overexpress
copper-zinc superoxide dismutase (Cadet et al., 1994). Recently, Mori et al. demonstrated that
oxidative stress mediated by the activation of neuronal nitric oxide synthase is associated with
MA-induced self-injurious behavior following MA administration (Mori et al., 2007). Our data
demonstrate that inhalation of MA also results in cellular toxicity in the lung, and this toxicity
NIH-PA Author Manuscript
is associated with increased cellular ROS and RNS. An important target of toxicity may be
airway epithelial cells, as evidenced by increased DHE staining (oxidant stress) seen in Figure
6A that correlated with morphological injury shown in Figure 4D.
The production of free radicals may be responsible for tissue injury in various cardiopulmonary
diseases (Yu, 1994), and acute lung injury and consequent acute respiratory distress syndrome
have been shown to result from free radical formation (Downey et al., 1999). Furthermore,
Huang et al. demonstrated that free radicals mediate amphetamine-induced acute pulmonary
edema in isolated rat lung (Huang et al., 2002). Therefore, it is reasonable to suggest that in
the model reported here, increased free radical formation results in cellular toxicity contributing
to pulmomary edema.
In the present study, there was a significant increase in the early response cytokines TNF-
α
and IL-6 in BAL fluid of mice exposed to MA. In human brain endothelial cells, Lee et al.
demonstrated that MA-induced disturbances in cellular redox status and activation of AP-1
and NF-
κB can play critical roles in the signaling pathways leading to upregulation of
inflammatory genes (Lee et al., 2001). In vitro studies have shown that humoral-mediated and
cell-mediated responses are affected by MA (Zule & Desmond, 1999). For example, it has
NIH-PA Author Manuscript
been demonstrated that MA affects immune function with a significant suppression of IL-2,
but not IL-4 production by T lymphocytes, as well as a suppression of B lymphocyte
proliferation (House et al., 1994). In vivo, chronic intraperitoneal administration of MA in a
mouse model significantly decreased the production of IL-2 and interferon (IFN)-
γ , while
significantly increasing the production of TNF-
α by cultured splenocytes (Yu et al., 2002),
further suggesting that MA may have immunomodulatory activity (Yu et al., 2002). The results
reported here are consistent with MA having an immunomodulatory effect in the lung, although
the mechanisms of this remain to be discovered.
Currently, there are no peer-reviewed reports on the relationship between individuals exposed
to MA smoke and resultant health consequences. There have been anecdotal reports of
increased asthma, pulmonary fibrosis, and upper respiratory complaints (Nestor et al.,
1989b; Schaiberger et al., 1993); however, no documented health statistics are available at this
time. The data presented here show that a single inhalation exposure to MA can result in lung
injury in mice and support the need to conduct further studies to determine whether there may
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 9
be a link between MA exposures and respiratory effects in humans. Given that most exposures
to MA are chronic in nature, the data presented here support the need to conduct additional
NIH-PA Author Manuscript
studies of the effects of chronic, repeated exposure to MA. Demonstrating that inhalation of
MA can result in lung injury should prove useful for consideration of improved respiratory
treatment of patients removed from situations where MA exposures may have occurred.
Acknowledgments
This work was supported in part by the National Center for Research Resources (NCRR; P20RR017670 to A. H.) and
the National Heart, Lung, and Blood Institute (NHLBI; 1F32HL086154 to S. M. W.), both components of the National
Institutes of Health (NIH). Its contents are solely the responsibility of the authors and not necessarily the official views
of NCRR, NHLBI, or NIH. The authors thank R. Hamilton for statistical support, L. Hoerner for technical assistance
with the laboratory animals, and P. Shaw for technical assistance with the LSC measurements. In addition, we thank
A. Harris and B. Klietz at the Montana State Crime Laboratory for their technical assistance with the development of
the exposure model.
REFERENCES
Benov L, Sztejnberg L, Fridovich I. Critical evaluation of the use of hydroethidine as a measure of
superoxide anion radical. Free Radical Biol. Med 1998;25:826–831. [PubMed: 9823548]
Bloom JW, Kaltenborn WT, Paoletti P, Camilli A, Lebowitz MD. Respiratory effects of non-tobacco
cigarettes. Br. Med. J. Clin. Res. Ed 1987;295:1516–1518.
NIH-PA Author Manuscript
Budd SL, Castilho RF, Nicholls DG. Mitochondrial membrane potential and hydroethidine-monitored
superoxide generation in cultured cerebellar granule cells. FEBS Lett 1997;415:21–24. [PubMed:
9326361]
Cadet JL, Sheng P, Ali S, Rothman R, Carlson E, Epstein C. Attenuation of methamphetamine-induced
neurotoxicity in copper/zinc superoxide dismutase transgenic mice. J. Neurochem 1994;62:380–383.
[PubMed: 7505315]
Cook CE, Jeffcoat AR, Hill JM, Pugh DE, Patetta PK, Sadler BM, White WR, Perez-Reyes M.
Pharmacokinetics of methamphetamine self-administered to human subjects by smoking S-(+)-
methamphetamine hydrochloride. Drug Metab. Dispos 1993;21:717–723. [PubMed: 8104133]
Davidson C, Gow AJ, Lee TH, Ellinwood EH. Methamphetamine neurotoxicity: necrotic and apoptotic
mechanisms and relevance to human abuse and treatment. Brain Res. Brain Res. Rev 2001;36:1–22.
[PubMed: 11516769]
Decker T, Lohmann-Matthes ML. A quick and simple method for the quantitation of lactate
dehydrogenase release in measurements of cellular cytotoxicity and tumor necrosis factor (TNF)
activity. J. Immunol. Methods 1988;115:61–69. [PubMed: 3192948]
Deng X, Ladenheim B, Tsao LI, Cadet JL. Null mutation of c-fos causes exacerbation of
methamphetamine-induced neurotoxicity. J. Neurosci 1999;19:10107–10115. [PubMed: 10559418]
Deng X, Wang Y, Chou J, Cadet JL. Methamphetamine causes widespread apoptosis in the mouse brain:
NIH-PA Author Manuscript
evidence from using an improved TUNEL histochemical method. Brain Res. Mol. Brain Res
2001;93:64–69. [PubMed: 11532339]
Downey GP, Dong Q, Kruger J, Dedhar S, Cherapanov V. Regulation of neutrophil activation in acute
lung injury. Chest 1999;116:46S–54S. [PubMed: 10424590]
Fink B, Laude K, McCann L, Doughan A, Harrison DG, Dikalov S. Detection of intracellular superoxide
formation in endothelial cells and intact tissues using dihydroethidium and an HPLC-based assay.
Am. J. Physiol. Cell Physiol 2004;287:C895–902. [PubMed: 15306539]
Fligiel SE, Roth MD, Kleerup EC, Barsky SH, Simmons MS, Tashkin DP. Tracheobronchial
histopathology in habitual smokers of cocaine, marijuana, and/or tobacco. Chest 1997;112:319–326.
[PubMed: 9266864]
Forrester JM, Steele AW, Waldron JA, Parsons PE. Crack lung: An acute pulmonary syndrome with a
spectrum of clinical and histopathologic findings. Am. Rev. Respir. Dis 1990;142:462–467.
[PubMed: 2382909]
Gong H Jr. Fligiel S, Tashkin DP, Barbers RG. Tracheobronchial changes in habitual, heavy smokers of
marijuana with and without tobacco. Am. Rev. Respir. Dis 1987;136:142–149. [PubMed: 3496818]
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 10
Green DR, Reed JC. Mitochondria and apoptosis. Science 1998;281:1309–1312. [PubMed: 9721092]
Grobe AC, Wells SM, Benavidez E, Oishi P, Azakie A, Fineman JR, Black SM. Increased oxidative
NIH-PA Author Manuscript
stress in lambs with increased pulmonary blood flow and pulmonary hypertension: Role of NADPH
oxidase and endothelial NO synthase. Am. J. Physiol. Lung Cell. Mol. Physiol 2006;290:L1069–
1077. [PubMed: 16684951]
Haim DY, Lippmann ML, Goldberg SK, Walkenstein MD. The pulmonary complications of crack
cocaine. A comprehensive review. Chest 1995;107:233–240. [PubMed: 7813284]
Haugen TS, Skjonsberg OH, Kahler H, Lyberg T. Production of oxidants in alveolar macrophages and
blood leukocytes. Eur. Respir. J 1999;14:1100–1105. [PubMed: 10596697]
House RV, Thomas PT, Bhargava HN. Comparison of immune functional parameters following in vitro
exposure to natural and synthetic amphetamines. Immunopharmacol. Immunotoxicol 1994;16:1–21.
[PubMed: 8169319]
Huang KL, Shaw KP, Wang D, Hsu K, Huang TS, Chen HI. Free radicals mediate amphetamine-induced
acute pulmonary edema in isolated rat lung. Life Sci 2002;71:1237–1244. [PubMed: 12106589]
Jayanthi S, Deng X, Bordelon M, McCoy MT, Cadet JL. Methamphetamine causes differential regulation
of pro-death and anti-death Bcl-2 genes in the mouse neocortex. FASEB J 2001;15:1745–1752.
[PubMed: 11481222]
Kenyon NJ, van der Vliet A, Schock BC, Okamoto T, McGrew GM, Last JA. Susceptibility to ozone-
induced acute lung injury in iNOS-deficient mice. Am. J. Physiol. Lung Cell. Mol. Physiol
2002;282:L540–545. [PubMed: 11839550]
Kim I, Oyler JM, Moolchan ET, Cone EJ, Huestis MA. Urinary pharmacokinetics of methamphetamine
NIH-PA Author Manuscript
and its metabolite, amphetamine following controlled oral administration to humans. Ther. Drug
Monit 2004;26:664–672. [PubMed: 15570192]
Kooy NW, Royall JA, Ischiropoulos H, Beckman JS. Peroxynitrite-mediated oxidation of
dihydrorhodamine 123. Free Radical Biol. Med 1994;16:149–156. [PubMed: 8005510]
LaVoie MJ, Hastings TG. Dopamine quinone formation and protein modification associated with the
striatal neurotoxicity of methamphetamine: evidence against a role for extracellular dopamine. J.
Neurosci 1999;19:1484–1491. [PubMed: 9952424]
Lee YW, Hennig B, Yao J, Toborek M. Methamphetamine induces AP-1 and NF-kappaB binding and
transactivation in human brain endothelial cells. J. Neurosci. Res 2001;66:583–591. [PubMed:
11746378]
Megyeri A, Bacso Z, Shields A, Eliason JF. Development of a stereological method to measure levels of
fluoropyrimidine metabolizing enzymes in tumor sections using laser scanning cytometry. Cytometry
A 2005;64:62–71. [PubMed: 15729713]
Miller FJ Jr. Gutterman DD, Rios CD, Heistad DD, Davidson BL. Superoxide production in vascular
smooth muscle contributes to oxidative stress and impaired relaxation in atherosclerosis. Circ. Res
1998;82:1298–1305. [PubMed: 9648726]
Mori T, Ito S, Kita T, Narita M, Suzuki T, Matsubayashi K, Sawaguchi T. Oxidative stress in
methamphetamine-induced self-injurious behavior in mice. Behav. Pharmacol 2007;18:239–249.
NIH-PA Author Manuscript
[PubMed: 17426488]National Drug Intelligence Center. National drug threat assessment 2007.
National Drug Intelligence Center, U.S. Department of Justice; Washington, DC: 2006.
National Institute on Drug Abuse. Methamphetamine abuse: Epidemiologic issues and implications.
Miller, NJKMA., editor. National Institute on Drug Abuse; Washington, DC: 1991. Research
Monograph 115
National Institute on Drug Abuse. Research report: Methamphetamine: Abuse and addiction. National
Institute on Drug Abuse, National Institutes of Health; Washington, DC: 2002.
National Institute on Drug Abuse. Methamphetamine abuse and addiction. National Institute on Drug
Abuse, National Institutes of Health; Washington, DC: 2006.
Nestor TA, Tamamoto WI, Kam TH, Schultz T. Acute pulmonary oedema caused by crystalline
methamphetamine. Lancet 1989a;2:1277–1278. [PubMed: 2573788]
Nestor TA, Tamamoto WI, Kam TH, Schultz T. Crystal methamphetamine-induced acute pulmonary
edema: a case report. Hawaii Med. J 1989b;48:457–458. 460. [PubMed: 2592191]
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 11
Paravicini TM, Gulluyan LM, Dusting GJ, Drummond GR. Increased NADPH oxidase activity,
gp91phox expression, and endothelium-dependent vasorelaxation during neointima formation in
rabbits. Circ. Res 2002;91:54–61. [PubMed: 12114322]
NIH-PA Author Manuscript
Preston KL, Wagner GC, Schuster CR, Seiden LS. Long-term effects of repeated methylamphetamine
administration on monoamine neurons in the rhesus monkey brain. Brain Res 1985;338:243–248.
[PubMed: 2411342]
Ricaurte GA, McCann UD. Neurotoxic amphetamine analogues: Effects in monkeys and implications
for humans. Ann. NY Acad. Sci 1992;648:371–382. [PubMed: 1379014]
Roth MD, Arora A, Barsky SH, Kleerup EC, Simmons M, Tashkin DP. Airway inflammation in young
marijuana and tobacco smokers. Am. J. Respir. Crit. Care Med 1998;157:928–937. [PubMed:
9517614]
Schaiberger PH, Kennedy TC, Miller FC, Gal J, Petty TL. Pulmonary hypertension associated with long-
term inhalation of “crank” methamphetamine. Chest 1993;104:614–616. [PubMed: 8101799]
Schwilke EW, dos Santos M. I. Sampaio, Logan BK. Changing patterns of drug and alcohol use in fatally
injured drivers in Washington State. J. Forens. Sci 2006;51:1191–1198.
Stephans SE, Yamamoto BK. Methamphetamine-induced neurotoxicity: Roles for glutamate and
dopamine efflux. Synapse 1994;17:203–209. [PubMed: 7974204]
Susskind H, Weber DA, Volkow ND, Hitzemann R. Increased lung permeability following long-term
use of free-base cocaine (crack). Chest 1991;100:903–909. [PubMed: 1914604]
Tashkin DP, Coulson AH, Clark VA, Simmons M, Bourque LB, Duann S, Spivey GH, Gong H.
Respiratory symptoms and lung function in habitual heavy smokers of marijuana alone, smokers of
NIH-PA Author Manuscript
marijuana and tobacco, smokers of tobacco alone, and nonsmokers. Am. Rev. Respir. Dis
1987;135:209–216. [PubMed: 3492159]
Tashkin DP, Khalsa ME, Gorelick D, Chang P, Simmons MS, Coulson AH, Gong H Jr. Pulmonary status
of habitual cocaine smokers. Am. Rev. Respir. Dis 1992;145:92–100. [PubMed: 1731605]
Tashkin DP, Kleerup EC, Hoh CK, Kim KJ, Webber MM, Gil E. Effects of ’crack’ cocaine on pulmonary
alveolar permeability. Chest 1997;112:327–335. [PubMed: 9266865]
Taylor DR, Fergusson DM, Milne BJ, Horwood LJ, Moffitt TE, Sears MR, Poulton R. A longitudinal
study of the effects of tobacco and cannabis exposure on lung function in young adults. Addiction
2002;97:1055–1061. [PubMed: 12144608]
Taylor DR, Poulton R, Moffitt TE, Ramankutty P, Sears MR. The respiratory effects of cannabis
dependence in young adults. Addiction 2000;95:1669–1677. [PubMed: 11219370]
Yu BP. Cellular defenses against damage from reactive oxygen species. Physiol. Rev 1994;74:139–162.
[PubMed: 8295932]
Yu Q, Zhang D, Walston M, Zhang J, Liu Y, Watson RR. Chronic methamphetamine exposure alters
immune function in normal and retrovirus-infected mice. Int. Immunopharmacol 2002;2:951–962.
[PubMed: 12188036]
Zule WA, Desmond DP. An ethnographic comparison of HIV risk behaviors among heroin and
methamphetamine injectors. Am. J. Drug Alcohol Abuse 1999;25:1–23. [PubMed: 10078975]
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 12
NIH-PA Author Manuscript
FIG. 1.
Heated methamphetamine exposure system. MA was heated over a propane heat source in the
heating vessel. The resultant MA vapor was moved through the animal chamber by constant
air flow of 1.5 L/min generated by an air pump and a vacuum source.
NIH-PA Author Manuscript
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 13
NIH-PA Author Manuscript
NIH-PA Author Manuscript
FIG. 2.
MA plasma levels with increasing doses of MA. Plasma MA levels were determined by LC/
MS. Following exposure to the vapors from increasing amounts of heated MA, MA plasma
levels were increased in the exposed mice. Values are means ± SEM from four mice in each
group. Correlation between MA dose and plasma levels was calculated using a Pearson
correlation test (
r = .7450,
p <.001).
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 14
NIH-PA Author Manuscript
NIH-PA Author Manuscript
FIG. 3.
Assessment of lung injury following acute exposure to vaporized MA. Mice were exposed to
vapors from 25 to 100 mg of heated MA. Tissues were collected 3 h following exposure to
assess parameters of lung injury. (A) Total protein in the BAL fluid was assessed (controls,
n = 15; 25–100 mg MA,
n = 5 each). (B) Albumin in the BAL fluid was assessed (
n = 4 for all
groups). (C) Cell numbers from whole lung lavage was determined (controls,
n = 15; 25 mg
MA,
n = 8; 50 mg MA,
n = 11; 100 mg MA,
n = 9). (D) LDH in BAL fluid was measured (
n
= 8 for all groups). Asterisk indicates significant at
p < .05 versus controls. Data are shown as
means ± SEM.
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 15
NIH-PA Author Manuscript
FIG. 4.
NIH-PA Author Manuscript
Assessment of lung histopathology following acute exposure to vaporized MA. Lung
morphology was evaluated by H&E staining of paraffin sections. Representative sections from
control mice (
n = 4; panels A—B) and mice 3 h after MA exposure (
n = 4; panels C—D).
Panels A and B show representative images of lung tissue (original magnification, 20×). In
panel A, the capillaries (black arrows) are apparent throughout the tissue. In panel C, no clear
capillaries could be identified, but what appears to be the remains of a small arteriole is shown
(black arrow). Higher magnification (original magnification 100×) of the airway epithelial cells
are shown in panels B and D. The black arrows in panel D point to areas of disruption of normal
cellular architecture.
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 16
NIH-PA Author Manuscript
FIG. 5.
Levels of early response cytokines in BAL fluid following acute exposure to vaporized MA.
Three hours following 25–100 mg MA exposure, TNF-
α production (A) and IL-6 production
(B) in BAL fluid was determined (
n = 8 for all groups). Asterisk indicates significant at
p < .
05 versus controls. Data are shown as means ± SEM.
NIH-PA Author Manuscript
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 17
NIH-PA Author Manuscript
NIH-PA Author Manuscript
FIG. 6.
Determination of oxidative stress in lung cells and tissue following exposure to vaporized MA.
Frozen sections were stained with DHE ± PEG-SOD (see Materials and Methods), imaged,
and analyzed by laser scanning cytometry (LSC). (A) DHE staining in lung sections.
Representative images from control and MA-exposed mice are shown. (B) The maximum pixel
values and percentages of DHE-positive cells are graphed (
n = 4). Asterisk indicates significant
at
p < .05 versus controls. Data are shown as means ± SEM.
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.
Wells et al.
Page 18
TABLE 1
Effect of Inhaled Methamphetamine on Intracellular Reactive Oxygen and Reactive Nitrogen Species
NIH-PA Author Manuscript
Control
Methamphetamine
DHR MCF
2527 ± 120
3690 ± 160
*
DHE MCF
11584 ± 147
12490 ± 220
*
Note: Data are means ±
SEM. DHR, dihydrorhodamine-123; DHE, dihydroethidium; MCF, mean channel fluorescence. n = 4–5;
*P < 0.05 vs. control.
NIH-PA Author Manuscript
NIH-PA Author Manuscript
Inhal Toxicol. Author manuscript; available in PMC 2009 September 29.