 New Zealand Pharmacovigilance Centre
New Zealand Pharmacovigilance Centre
University of Otago
PO Box 913, Dunedin, New Zealand
Telephone: 64-3-479 7185
Fax: 64-3-479 7150
Email: nzphvc@otago ac nz
Website: www otago ac nz/carm
CAVEAT DOCUMENT
Accompanying statement to data released from the
NEW ZEALAND CENTRE FOR ADVERSE REACTIONS MONITORING
The Centre for Adverse Reactions Monitoring (CARM) has only limited details about each suspected adverse
reaction contained in its Database. It is important that the limitations and qualifications which apply to the
information and its use are understood.
The data made available represent the collection of spontaneous reports in the CARM database associated
with therapeutic products/vaccines granted regulatory approval for use in New Zealand.
Reports have been submitted to the Centre since April 1965 and in many instances describe no more than
suspicions which have arisen from observation of an unexpected or unwanted event. This level of reporting
is due to CARM encouraging reporters to report events they suspect may be associated with a
pharmaceutical product/vaccine irrespective of whether or not they believe it was the cause. CARM accepts
all reports and proof of causality is not required when submitting a report to CARM. Coincidental events that
may be unrelated to pharmaceutical product/vaccine exposure may be reported. This is particularly possible
when the product has widespread use, or is used in targeted strategies such as vaccination campaigns.
In most instances it cannot be proven that a pharmaceutical product or ingredient is the cause of an event in
the Database. Reports vary in quality, completeness and detail and may include detail that is incorrect.
Consequently, a report in the CARM database of an event does not confirm that the pharmaceutical
product/vaccine caused the event.
The volume of reports for a particular product may be influenced by the extent of use of the product,
publicity, nature of reactions and other factors which vary over time and from product to product. It is
generally accepted internationally that systems such as CARM are subject to underreporting which may
result in scant reports for events perceived by the reporter to be minor or well recognised, whilst more
serious or unexpected events are possibly more likely to be reported, even if they are coincidental.
Moreover, no information is provided on the number of patients exposed to the product.
The data contained in these tables are further subject to ongoing internal quality controls, review and
updating and therefore may be subject to change, particularly if follow-up information is received.
For the above reasons interpretations of adverse reaction data, and particularly those based on comparisons
between pharmaceutical products, may be misleading. Any use of this information must take into account at
least the above. Although this information is now released, it is strongly recommended that prior to any use
under the Official Information Act 1982
of such information, CARM is contacted for interpretation.
Any publication, in whole or in part, of the obtained information must have published with it a statement:
(i)
of the source of the information
(ii)
that the information is not homogenous at least with respect to origin or likelihood that the
pharmaceutical product/vaccine caused the adverse reaction
(iii)
that the information does not represent the opinion of the NZPhvC or CARM.
Released
Director
New Zealand Pharmacovigilance Centre
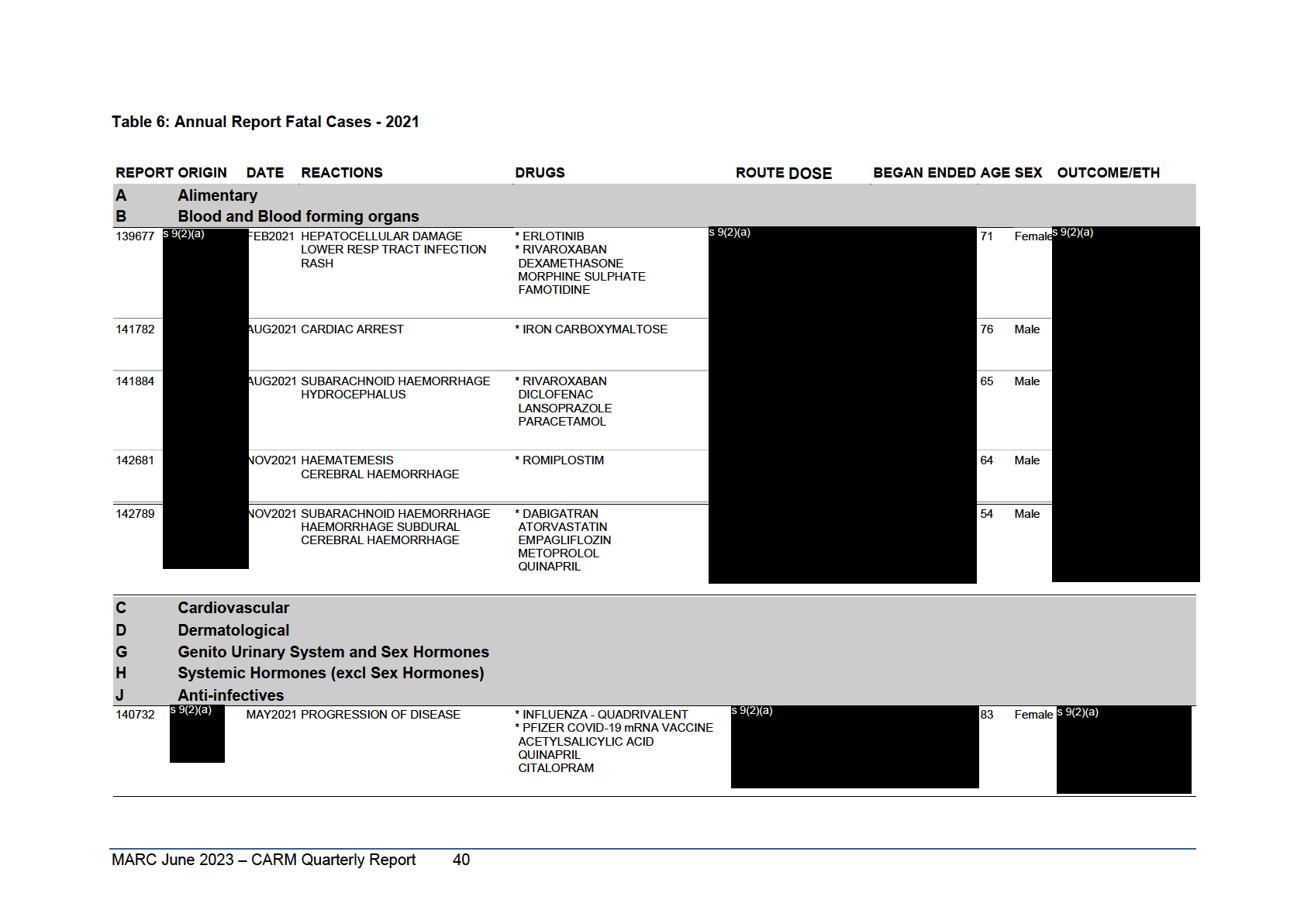
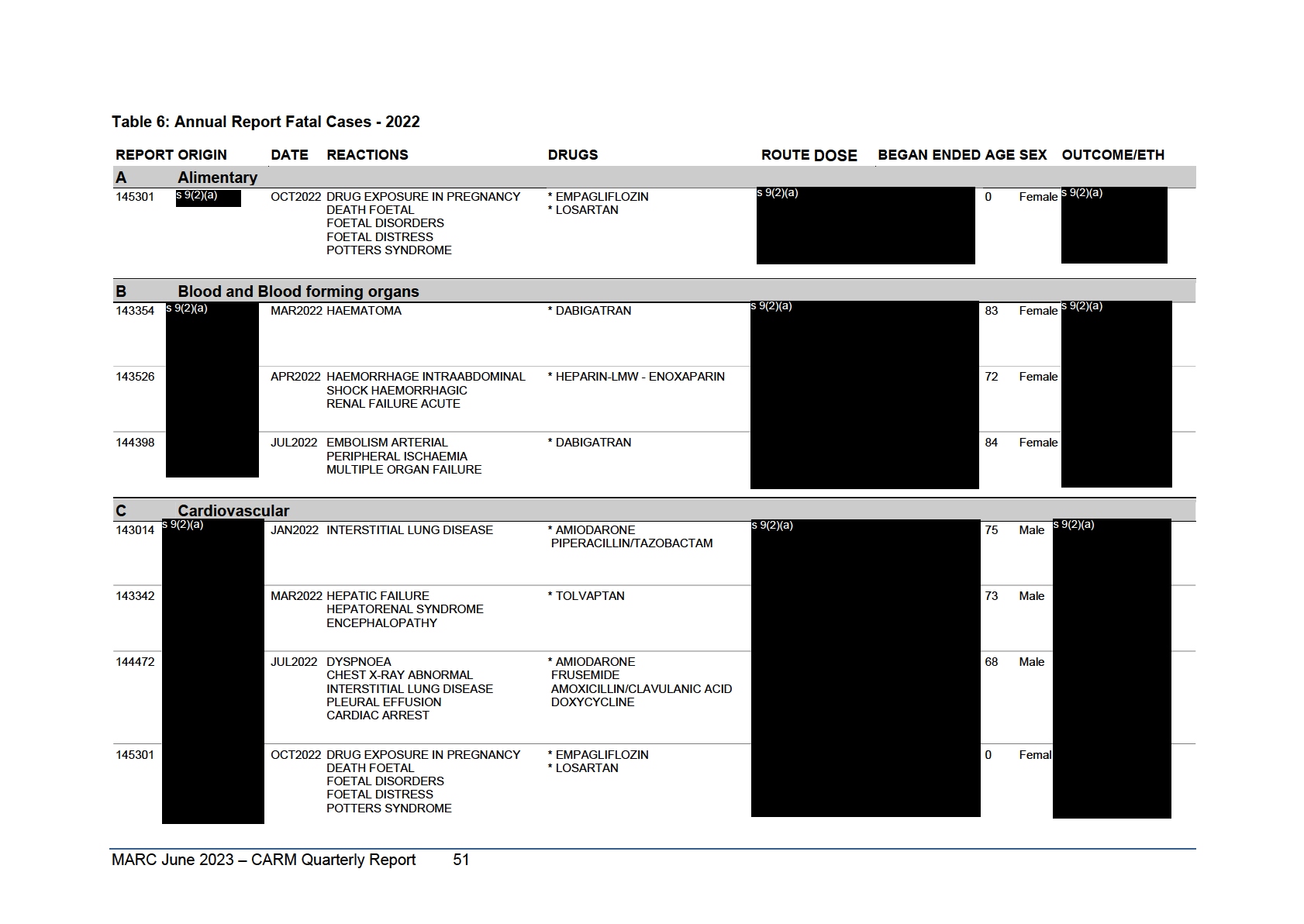
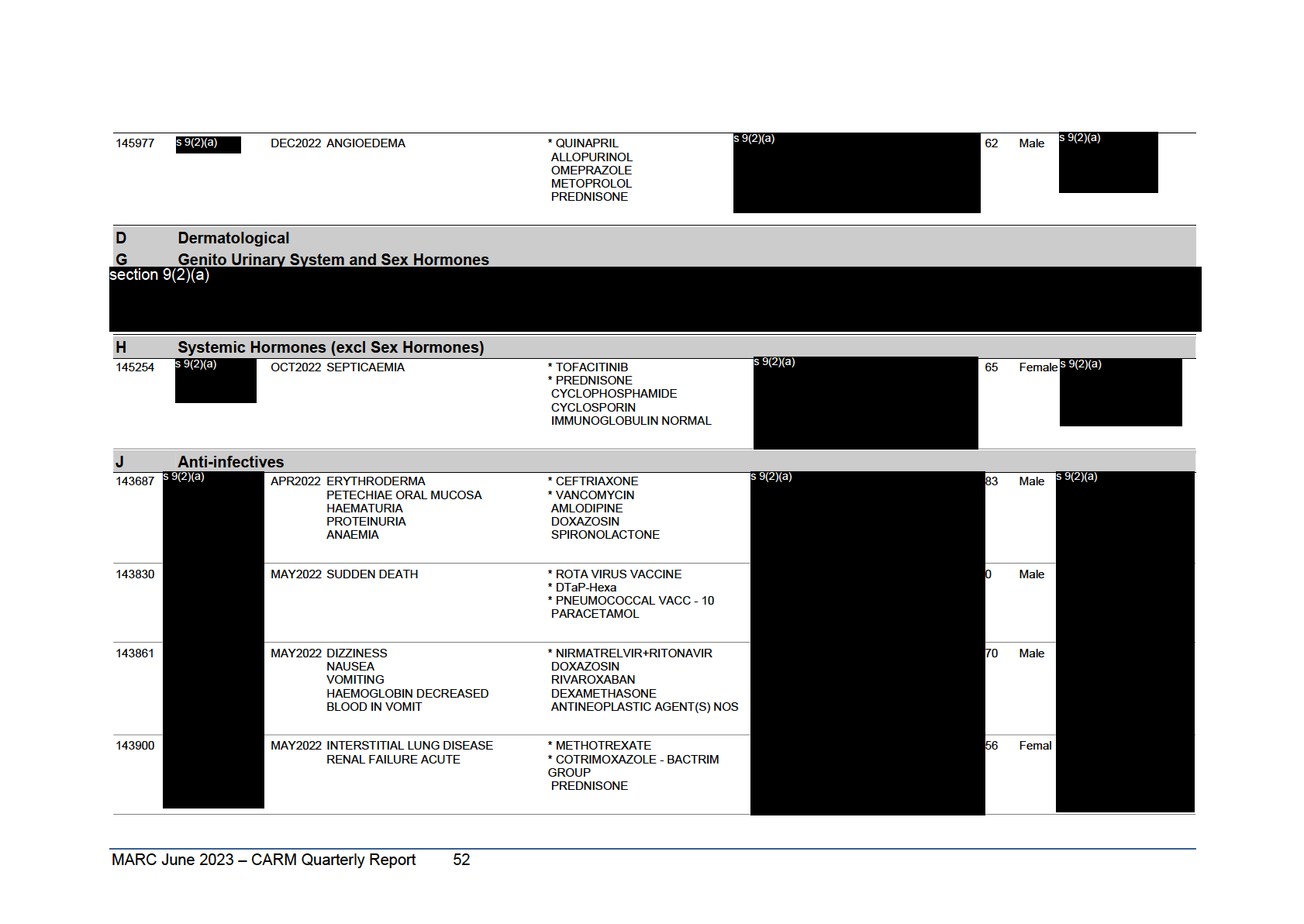
Table 6: Annual Report Fatal Cases January to December 2020
1982
Reports are grouped according to the ATC classification of the suspect agent(s). Cases where there is more than one suspect agent are listed once in each relevant ATC classification and those cases appearing in more
than one ATC classification are identified with ***** below the Report Number. This listing covers the 107 reports, identifying 129 suspect medicines, which resulted in a fatal outcome regardless of causality.
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
Act
Alimentary
s 9(2)(a)
s 9(2)(a)
138845 s 9(2)(a) NOV2020 PNEUMONIA
*
CLOZAPINE
67
M
*****
SEPSIS
*
OMEPRAZOLE
BRONCHIECTASIS
NICOTINE
BUDESONIDE/EFORMOTEROL
AMISULPRIDE
Blood and Blood forming organs
s 9(2)(a)
Information
137735
JUL2020 INTESTINAL
NECROSIS
*
DABIGATRAN
76
M
*****
*
IDARUCIZUMAB
137934
AUG2020 PULMONARY
HAEMORRHAGE
*
DABIGATRAN
74
F
137603
JUL2020 HAEMORRHAGE
RETROPERITONEAL
*
HEPARIN-LMW - ENOXAPARIN
79
M
Official
136017
FEB2020
CEREBRAL HAEMORRHAGE
*
RIVAROXABAN
91
F
THROMBOSIS CEREBRAL
ACE INHIBITOR (NOS)
the
THYROXINE
PARACETAMOL
UNCLASSIFIED
AGENT
137732
JUL2020
HAEMORRHAGE INTRACRANIAL
*
RIVAROXABAN
85
F
FALL
FRACTURE
under
135764
JAN2020
MYOCARDIAL INFARCTION
*
ELTROMBOPAG
82
M
THROMBOCYTOSIS
FELODIPINE
ARM
PAIN
CANDESARTAN
CHEST
PAIN
METFORMIN
CITALOPRAM
MARC September 2021 – CARM Quarterly Report
41
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
137416 s 9(2)(a)
JUN2020 CHILLS
*
HUMAN SERUM ALBUMIN
73
F
PARACETAMOL
ACETYLSALICYLIC ACID
137738
JUL2020 HYPOTENSION
*
IMMUNOGLOBULIN
NORMAL
58
F
*****
BACK PAIN
MORPHINE SULPHATE
VOMITING
*
PLATELETS
RECTAL
BLEEDING
LAXSOL
INFUSION
REACTION
FAMOTIDINE
Cardiovascular
s 9(2)(a)
136174
FEB2020
SUDDEN DEATH
*
VALSARTAN/SACUBITRIL
67
M
137186
JUN2020
ABDOMINAL PAIN
*
ALIROCUMAB
79
F
ANOREXIA
BLOATING
MELAENA
Genito Urinary System and Sex Hormones
137895 s 9(2)(a)
AUG2020 UTERINE
CARCINOMA
*
OESTRIOL
71
F
Systemic Hormones (excl Sex Hormones)
s 9(2)(a)
135878
JAN2020
COLLAPSE CIRCULATORY
*
OCTREOTIDE LAR
68
F
under the Official Information Act 1982
HEART DISORDER
MARC September 2021 – CARM Quarterly Report
42
Released
Antiinfectives
1982
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
136687
MAY2020 DRESS
SYNDROME
*
AMOXICILLIN
80
F
NEPHRITIS INTERSTITIAL
Act
136929
MAY2020 ANAEMIA
APLASTIC
*
CHLORAMPHENICOL
70
F
PREDNISONE
136250
MAR2020 JAUNDICE
*
FLUCLOXACILLIN
83
F
HEPATITIS CHOLESTATIC
RIVAROXABAN
DEATH
BETAMETHASONE
PARACETAMOL
METOPROLOL
135633
JAN2020 HYPERKALAEMIA
*
AMPHOTERICIN, LIPOSOMAL
42
F
*****
CARDIAC ARREST
*
GABAPENTIN
Information
*
METHADONE
PIPERACILLIN/TAZOBACTAM
MULTIVITAMINS
138123
AUG2020
CONGESTIVE HEART FAILURE
*
INTRAGAM P
71
M
HYPOXIA
DOXYCYCLINE
HYPOTENSION
VALACICLOVIR
RENAL IMPAIRMENT
METOPROLOL
Official
HYPERKALAEMIA
TIOTROPIUM
/OLODATEROL
135879
JAN2020 CHOLELITHIASIS
* V
IEKIRA PAK-RBV
61
M
DYSPNOEA
SALBUTAMOL
the
FATIGUE
MYOCARDIAL
ISCHAEMIA
CORONARY
ARTERY
DISORDER
137738
JUL2020 HYPOTENSION
*
IMMUNOGLOBULIN NORMAL
58
F
*****
BACK PAIN
MORPHINE SULPHATE
VOMITING
*
PLATELETS
under
RECTAL
BLEEDING
LAXSOL
INFUSION
REACTION
FAMOTIDINE
137514
JUL2020 SUDDEN
DEATH
*
DTaP-Hexa
4m
M
*
PNEUMOCOCCAL VACC - 10
MARC September 2021 – CARM Quarterly Report
43
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
137890
AUG2020
SUDDEN DEATH
*
ROTA VIRUS VACCINE
1m
M
1982
*
DTaP-Hexa
*
PNEUMOCOCCAL VACC - 10
CHOLECALCIFEROL
FERROUS
SULPHATE
Act
138219
SEP2020
SUDDEN INFANT DEATH SYNDROME
*
DTaP-Hexa
6m
M
*
PNEUMOCOCCAL VACC - 10
136512
MAR2020 CHRONIC
OBSTRUCT AIRWAYS DISEASE
*
INFLUENZA - QUADRIVALENT
67
F
136521
APR2020 SUDDEN
DEATH
*
INFLUENZA - QUADRIVALENT
67
M
ATORVASTATIN
BENDROFLUAZIDE
FELODIPINE
ACETYLSALICYLIC
ACID
Information
136522
APR2020 SUDDEN
DEATH
*
INFLUENZA - QUADRIVALENT
71
F
ATENOLOL
ENALAPRIL
FRUSEMIDE
137611
JUL2020 UNCONSCIOUSNESS
*
INFLUENZA - QUADRIVALENT
85
M
Official
FIXED PUPILS
137514
JUL2020 SUDDEN
DEATH
* DTaP-Hexa
4m
M
*
PNEUMOCOCCAL VACC - 10
the
137890
AUG2020
SUDDEN DEATH
*
ROTA VIRUS VACCINE
1m
M
*
DTaP-Hexa
*
PNEUMOCOCCAL VACC - 10
CHOLECALCIFEROL
FERROUS
SULPHATE
under
138219
SEP2020
SUDDEN INFANT DEATH SYNDROME
*
DTaP-Hexa
6m
M
*
PNEUMOCOCCAL VACC - 10
MARC September 2021 – CARM Quarterly Report
44
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
137890
AUG2020 SUDDEN
DEATH
*
ROTA VIRUS VACCINE
1m
M
1982
*
DTaP-Hexa
*
PNEUMOCOCCAL VACC - 10
CHOLECALCIFEROL
FERROUS
SULPHATE
Act
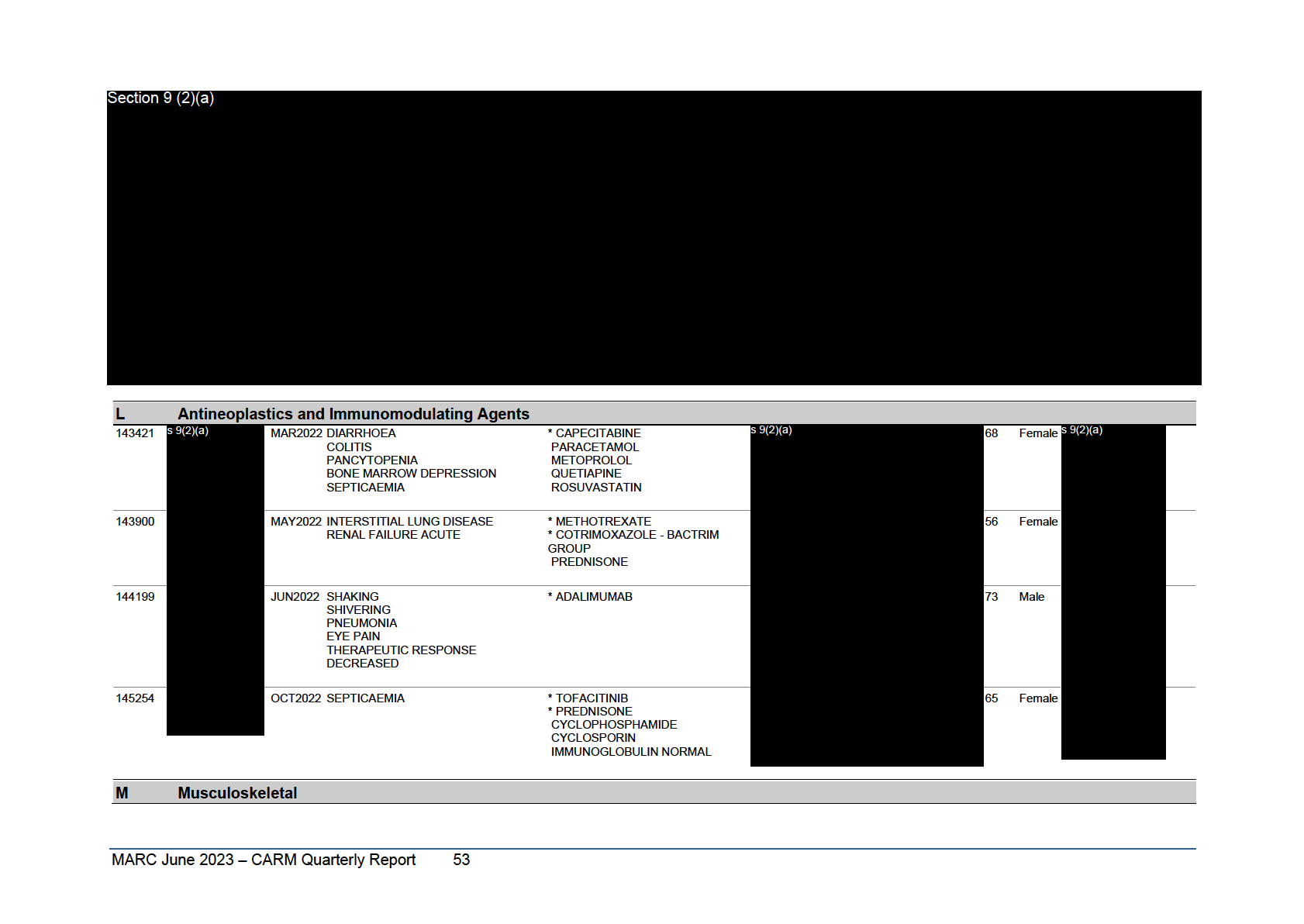
Antineoplastics and Immunomodulating Agents
s 9(2)(a)
138095
AUG2020 MYALGIA
*
ALECTINIB
74
F
PNEUMONITIS
DABIGATRAN
PROGRESSION OF DISEASE
FELODIPINE
CHOLECALCIFEROL
SALBUTAMOL
136300
MAR2020 CARDIOMYOPATHY
* DAUNORUBICIN
63
M
Information
DEATH
*
ANTHRACYCLINE NOS
*
DAUNORUBICIN
136027
FEB2020 LYMPHANGITIS
*
ATEZOLIZUMAB
64
F
PULMONARY INFILTRATION
136028
FEB2020 FEVER
*
ATEZOLIZUMAB
29
M
Official
FATIGUE
*
COBIMETINIB
PROGRESSION OF DISEASE
139042
NOV2020
PROGRESSION OF DISEASE
*
ATEZOLIZUMAB
71
M
the
*
BEVACIZUMAB
138444
SEP2020 LEUKOENCEPHALOPATHY
* RITUXIMAB
64
F
PROGRESSION OF DISEASE
*
BENDAMUSTINE
IMMUNE (IRIS) SYNDROME
FLUDARABINE
PNEUMONIA
MITOXANTRONE
RESPIRATORY
ARREST
under
139042
NOV2020
PROGRESSION OF DISEASE
*
ATEZOLIZUMAB
71
M
*
BEVACIZUMAB
MARC September 2021 – CARM Quarterly Report
45
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
137180
JUN2020
PROGRESSION OF DISEASE
*
PEMBROLIZUMAB
69
M
1982
MEDICINE
INEFFECTIVE
*
CARBOPLATIN
*
GEMCITABINE
Act
137280
JUN2020 INTESTINAL
PERFORATION
*
CETUXIMAB
58
M
ANTINEOPLASTIC AGENT(S) NOS
136028
FEB2020 FEVER
* ATEZOLIZUMAB
29
M
FATIGUE
*
COBIMETINIB
PROGRESSION OF DISEASE
137495
JUN2020
PROGRESSION OF DISEASE
*
COBIMETINIB
M
*
VEMURAFENIB
137623
JUL2020 CONVULSIONS
*
COBIMETINIB
M
DYSPHAGIA
*
VEMURAFENIB
Information
136300
MAR2020 CARDIOMYOPATHY
*
DAUNORUBICIN
63
M
DEATH
*
ANTHRACYCLINE
NOS
*
DAUNORUBICIN
136300
MAR2020 CARDIOMYOPATHY
* DAUNORUBICIN
63
M
DEATH
*
ANTHRACYCLINE
NOS
Official
*
DAUNORUBICIN
137180
JUN2020
PROGRESSION OF DISEASE
*
PEMBROLIZUMAB
69
M
MEDICINE INEFFECTIVE
*
CARBOPLATIN
the
*
GEMCITABINE
136172
FEB2020
PROGRESSION OF DISEASE
*
PEMBROLIZUMAB
50
M
137180
JUN2020
PROGRESSION OF DISEASE
*
PEMBROLIZUMAB
69
M
under
MEDICINE INEFFECTIVE
*
CARBOPLATIN
*
GEMCITABINE
137486
JUN2020
PROGRESSION OF DISEASE
*
PEMBROLIZUMAB
70
F
MARC September 2021 – CARM Quarterly Report
46
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
137885
AUG2020 PNEUMONIA
*
PEMBROLIZUMAB
63
M
1982
138323
SEP2020 RASH
*
PEMBROLIZUMAB
62
M
Act
RHINITIS
PEMETREXED
LACRIMATION
INCREASED
CARBOPLATIN
PROGRESSION OF DISEASE
138952
NOV2020
CONVULSIONS AGGRAVATED
*
PEMBROLIZUMAB
67
F
138444
SEP2020 LEUKOENCEPHALOPATHY
*
RITUXIMAB
64
F
PROGRESSION OF DISEASE
*
BENDAMUSTINE
IMMUNE (IRIS) SYNDROME
FLUDARABINE
PNEUMONIA
MITOXANTRONE
RESPIRATORY
ARREST
Information
137118
MAY2020
PROGRESSION OF DISEASE
*
TRASTUZUMAB
54
F
139195
DEC2020
PROGRESSION OF DISEASE
*
TRASTUZUMAB
F
Official
137420
JUN2020
PROGRESSION OF DISEASE
*
VELIPARIB
20
M
TEMOZOLOMIDE
137495
JUN2020
PROGRESSION OF DISEASE
*
COBIMETINIB
M
the
*
VEMURAFENIB
137623
JUL2020 CONVULSIONS
*
COBIMETINIB
M
DYSPHAGIA
*
VEMURAFENIB
138728
OCT2020
CARCINOMA COLON
*
ADALIMUMAB
81
M
under
METASTASES NOS
135703
JAN2020
RENAL FAILURE ACUTE
*
INFLIXIMAB
53
F
MARC September 2021 – CARM Quarterly Report
47
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
137053
MAY2020
PROGRESSION OF DISEASE
*
LENALIDOMIDE
71
M
1982
DEXAMETHASONE
CYCLOPHOSPHAMIDE
Act
137487
JUN2020
PROGRESSION OF DISEASE
*
LENALIDOMIDE
78
M
136365
MAR2020
PROGRESSION OF DISEASE
*
POMALIDOMIDE
69
F
Nervous System
s 9(2)(a)
137875
AUG2020 RESPIRATORY
ARREST
*
CODEINE
62
F
CARDIAC ARREST
Information
INTENTIONAL
OVERDOSE
135633
JAN2020 HYPERKALAEMIA
*
AMPHOTERICIN,
LIPOSOMAL
42
F
*****
CARDIAC ARREST
*
GABAPENTIN
*
METHADONE
PIPERACILLIN/TAZOBACTAM
MULTIVITAMINS
Official
136416
MAR2020 RESPIRATORY
DEPRESSION
*
MIDAZOLAM
54
F
CARDIAC
ARREST
*
MORPHINE SULPHATE
AGITATION
*
MORPHINE
SULPHATE
DEXAMETHASONE
the
136416
MAR2020 RESPIRATORY
DEPRESSION
*
MIDAZOLAM
54
F
CARDIAC ARREST
*
MORPHINE SULPHATE
AGITATION
*
MORPHINE SULPHATE
DEXAMETHASONE
under
137860
AUG2020
QT PROLONGED
*
OLANZAPINE
45
M
FIBRILLATION
VENTRICULAR
*
MORPHINE SULPHATE
CARDIAC
ARREST
METHAMPHETAMINE
MULTIPLE ORGAN FAILURE
CONVULSIONS
MARC September 2021 – CARM Quarterly Report
48
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
135633
JAN2020 HYPERKALAEMIA
* AMPHOTERICIN,
LIPOSOMAL
42
F
1982
*****
CARDIAC ARREST
*
GABAPENTIN
*
METHADONE
PIPERACILLIN/TAZOBACTAM
MULTIVITAMINS
Act
136332
MAR2020 CONVULSIONS
*
LAMOTRIGINE
35
F
THERAPEUTIC RESPONSE DECREASED
LEVETIRACETAM
137761
JUL2020 SUDDEN
DEATH
*
LAMOTRIGINE
22
F
138645
OCT2020 SUDDEN
DEATH
*
LAMOTRIGINE
19
M
TOPIRAMATE
MIDAZOLAM
Information
137879
AUG2020 ASPIRATION PNEUMONITIS
*
LEVODOPA/CARBIDOPA (NOS)
69
M
RIGORS
LORAZEPAM
NEUTROPENIA
QUETIAPINE
HYPERKALAEMIA
GABAPENTIN
DOMPERIDONE
138733
OCT2020
NEOPLASM MALIGNANT
*
CHLORPROMAZINE
M
*
PARALDEHYDE
Official
135869
JAN2020
MYOCARDIAL ISCHAEMIA
*
CLOZAPINE
55
M
the
135870
JAN2020 SUICIDE
*
CLOZAPINE
29
M
DIAZEPAM
OMEPRAZOLE
MYLANTA
135871
JAN2020 PNEUMONIA
*
CLOZAPINE
50
M
under
CHRONIC OBSTRUCT AIRWAYS DISEASE
MORPHINE SULPHATE
PREDNISONE
135872
JAN2020 PNEUMONIA
*
CLOZAPINE
84
M
MARC September 2021 – CARM Quarterly Report
49
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
135873
JAN2020 PARKINSONISM
*
CLOZAPINE
68
M
1982
ASPIRATION
PNEUMONITIS
LEVODOPA/CARBIDOPA (100/25)
CODEINE
FLUDROCORTISONE
LORAZEPAM
Act
135874
JAN2020 ARRHYTHMIA
*
CLOZAPINE
68
F
PARKINSONISM
MIDAZOLAM
DEMENTIA
ACETYLSALICYLIC
ACID
TRANSIENT
ISCHAEMIC
ATTACK
MADOPAR
SYNCOPE
ROPINIROLE
135875
JAN2020 DEMENTIA
*
CLOZAPINE
81
M
135876
JAN2020
MYOCARDIAL ISCHAEMIA
*
CLOZAPINE
67
F
CORONARY ARTERY DISORDER
Information
135877
JAN2020 PARKINSONISM
*
CLOZAPINE
58
M
MYOCARDIAL INFARCTION
PNEUMONIA
135906
JAN2020
CARDIAC ARREST
*
CLOZAPINE
43
M
Official
135907
JAN2020 APPENDICITIS
*
CLOZAPINE
77
M
ASPIRATION
LORATADINE
TERAZOSIN
the
LAXSOL
135908
JAN2020
MEGACOLON ACQUIRED
*
CLOZAPINE
54
M
STROKE
135909
JAN2020
SUDDEN DEATH
*
CLOZAPINE
38
M
under
MARC September 2021 – CARM Quarterly Report
50
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
135910
JAN2020 ASPIRATION PNEUMONITIS
*
CLOZAPINE
40
M
1982
BOWEL
MOTILITY
DISORDER
LEVOMEPROMAZINE
DIAZEPAM
HYOSCINE
TOPICAL
VALPROATE
SODIUM
Act
135911
JAN2020 PNEUMONIA
*
CLOZAPINE
66
M
135912
JAN2020 MELANOMA
MALIGNANT
*
CLOZAPINE
64
F
135913
JAN2020 CONSTIPATION
*
CLOZAPINE
56
M
RECTAL ULCER
DIAZEPAM
INTESTINAL
PERFORATION
LORAZEPAM
PERITONITIS
PAROXETINE
PROPRANOLOL
Information
135914
JAN2020
INTENTIONAL OVERDOSE
*
CLOZAPINE
48
M
135915
JAN2020 ASPIRATION
*
CLOZAPINE
67
M
INTESTINAL
OBSTRUCTION
RESPIRATORY
ARREST
Official
RENAL
FAILURE
AGGRAVATED
135916
JAN2020 HYPERTENSION
*
CLOZAPINE
64
F
HEART DISORDER
the
LIVER
FATTY
HEPATIC
CIRRHOSIS
135917
JAN2020
CARDIAC ARREST
*
CLOZAPINE
58
M
under
135918
JAN2020
CARDIAC FAILURE
*
CLOZAPINE
75
F
DIABETES MELLITUS
MARC September 2021 – CARM Quarterly Report
51
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
135982
FEB2020 MEGACOLON
ACQUIRED
*
CLOZAPINE
43
M
1982
SEPSIS SECONDARY
COLITIS
DEATH
Act
137377
JUN2020 PNEUMONIA
*
CLOZAPINE
55
F
137378
JUN2020
SUDDEN DEATH
*
CLOZAPINE
55
M
DRUG TOXICITY
137379
JUN2020
MYOCARDIAL INFARCTION
*
CLOZAPINE
55
M
137380
JUN2020 ARRHYTHMIA
*
CLOZAPINE
79
F
Information
137381
JUN2020
THROMBOSIS CORONARY
*
CLOZAPINE
49
M
PANTOPRAZOLE
IMIPRAMINE
AMISULPRIDE
SALBUTAMOL
Official
137382
JUN2020
THROMBOSIS CORONARY
*
CLOZAPINE
47
M
137383
JUN2020
SUDDEN DEATH
*
CLOZAPINE
17
M
the
METFORMIN
LAXSOL
ARIPIPRAZOLE
137385
JUN2020
SUDDEN DEATH
*
CLOZAPINE
43
M
METFORMIN
BENZTROPINE MESYLATE
under
PROPRANOLOL
RISPERIDONE
MARC September 2021 – CARM Quarterly Report
52
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
137451
JUN2020 ASPIRATION PNEUMONITIS
*
CLOZAPINE
78
M
1982
PNEUMONIA
MIDODRINE
ACETYLSALICYLIC
ACID
DOCUSATE + SENNOSIDES
FLUDROCORTISONE
Act
137452
JUN2020
MYOCARDIAL INFARCTION
*
CLOZAPINE
61
M
RESPIRATORY DISORDER
137453
JUN2020 STROKE
*
CLOZAPINE
76
M
DEMENTIA
LEVOMEPROMAZINE
137454
JUN2020 CARCINOMA
*
CLOZAPINE
54
M
MOCLOBEMIDE
ATORVASTATIN
METFORMIN
CHOLECALCIFEROL
Information
137455
JUN2020 PNEUMONIA
*
CLOZAPINE
78
M
138843
NOV2020 PNEUMONIA
*
CLOZAPINE
73
M
NEOPLASM MALIGNANT
PROGRESSION OF DISEASE
Official
138844
NOV2020 PNEUMONIA
*
CLOZAPINE
70
M
ATRIAL
FIBRILLATION
OLANZAPINE
TACHYCARDIA
VENTRICULAR
the
CARDIAC
ARREST
138845
NOV2020 PNEUMONIA
*
CLOZAPINE
67
M
*****
SEPSIS
*
OMEPRAZOLE
BRONCHIECTASIS
NICOTINE
BUDESONIDE/EFORMOTEROL
AMISULPRIDE
under
138846
NOV2020 PARKINSONISM
*
CLOZAPINE
73
F
APOMORPHINE
LEVODOPA
AMANTADINE
MIRTAZAPINE
MARC September 2021 – CARM Quarterly Report
53
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
138847
NOV2020
SUDDEN DEATH
*
CLOZAPINE
64
M
1982
ATENOLOL
FRUSEMIDE
LAXSOL
OMEPRAZOLE
Act
138848
NOV2020
SUDDEN DEATH
*
CLOZAPINE
67
M
MYOCARDIAL INFARCTION
BENZTROPINE MESYLATE
ATORVASTATIN
138849
NOV2020 ASPIRATION PNEUMONITIS
*
CLOZAPINE
91
M
CONFUSION
ALENDRONATE + CHOLECALCIFERO
SOMNOLENCE
DONEPEZIL
FALL
ESCITALOPRAM
FLUDROCORTISONE
138850
NOV2020 METASTASES
NOS
*
CLOZAPINE
52
M
Information
NEOPLASM
MALIGNANT
NEUTROPHILIA
LEUKOCYTOSIS
138851
NOV2020 DEMENTIA
*
CLOZAPINE
72
M
HEAD INJURY
Official
138852
NOV2020 DEMENTIA
*
CLOZAPINE
72
F
DONEPEZIL
DOCUSATE + SENNOSIDES
LORAZEPAM
VENLAFAXINE
the
139041
NOV2020 PNEUMONIA
*
CLOZAPINE
80
F
ESCITALOPRAM
OMEPRAZOLE
FLUTICASONE/SALMETEROL
DORZOLAMIDE/TIMOLOL
under
136416
MAR2020 RESPIRATORY
DEPRESSION
*
MIDAZOLAM
54
F
CARDIAC ARREST
*
MORPHINE SULPHATE
AGITATION
*
MORPHINE
SULPHATE
DEXAMETHASONE
MARC September 2021 – CARM Quarterly Report
54
Released
REPORT ORIGIN
DATE
REACTIONS
DRUGS
ROUTE DOSE/UNIT
BEGAN ENDED
AGE SEX OUTCOME
s 9(2)(a)
s 9(2)(a)
137860
AUG2020
QT PROLONGED
*
OLANZAPINE
45
M
Died
drug may be contributory
1982
FIBRILLATION
VENTRICULAR
*
MORPHINE
SULPHATE
Maori
CARDIAC
ARREST
METHAMPHETAMINE
MULTIPLE ORGAN FAILURE
CONVULSIONS
Act
136520
MAR2020 SUICIDE
*
PALIPERIDONE
53
F
Died - cause unclassifiable
Unknown
138733
OCT2020
NEOPLASM MALIGNANT
*
CHLORPROMAZINE
M
Died - cause unclassifiable
*
PARALDEHYDE
Unknown
137384
JUN2020 SUICIDE
*
QUETIAPINE
59
M
Died - unrelated to drug
BLOOD DISORDER
Maori
137668
JUL2020
THERAPEUTIC RESPONSE DECREASED
*
VENLAFAXINE
45
M
Died - unrelated to drug
Information
BISOPROLOL
European
Miscellaneous
s 9(2)(a)
137735
JUL2020 INTESTINAL
NECROSIS
* DABIGATRAN
76
M
Died - cause unclassifiable
Official
*****
*
IDARUCIZUMAB
Unknown
137578
JUL2020 FEELING
UNWELL
*
M&P VITAL ALL-IN-ONE
78
F
Died - cause unclassifiable
SUDDEN DEATH
European
the
under
MARC September 2021 – CARM Quarterly Report
55
Released
1982
Act
Information
Official
the
under
Released

1982
Act
Information
Official
the
under
Released

1982
Act
Information
Official
the
under
Released
1982
Act
Information
Official
the
under
Released
1982
Act
Information
Official
the
under
Released
1982
Act
Information
Official
the
under
Released
1982
Act
Information
Official
the
under
Released

1982
Act
Information
Official
the
under
Released
s 9(2)(a)
s 9(2)(a)
s 9(2)(a)
145365
OCT2022 SUICIDE
* CLOZAPINE
23
Female
1982
LAXSOL
CODEINE
FLUOXETINE
ZOPICLONE
Act
145367
OCT2022 DEMENTIA
* CLOZAPINE
80
Male
HYOSCINE BUTYLBROMIDE
LEVODOPA/CARBIDOPA (NOS)
HYDROXOCOBALAMIN
LACTULOSE
145369
OCT2022 PNEUMONIA
* CLOZAPINE
58
Male
DRUG LEVEL INCREASED
PULMONARY CARCINOMA
POST-OPERATIVE COMPLICATION
NOS
145415
OCT2022 METABOLIC SYNDROME
* CLOZAPINE
60
Female
Information
145416
OCT2022 LOWER RESP TRACT INFECTION
* CLOZAPINE
71
Male
RESPIRATORY FAILURE
METFORMIN
SIMVASTATIN
OMEPRAZOLE
ARIPIPRAZOLE
145420
OCT2022 CARCINOMA SQUAMOUS
* CLOZAPINE
76
Male
Official
METASTASES NOS
145421
OCT2022 LYMPHOMA MALIGNANT
* CLOZAPINE
67
Female
the
145423
OCT2022 CARDIAC ARREST
* CLOZAPINE
41
Male
145459
OCT2022 DEMENTIA
* CLOZAPINE
78
Female
CACHEXIA
MORPHINE SULPHATE
CILAZAPRIL
THYROXINE
under OMEPRAZOLE
145465
OCT2022 DEMENTIA
* CLOZAPINE
74
Male
MADOPAR
CLONAZEPAM
CELECOXIB
RIVASTIGMINE
MARC June 2023 – CARM Quarterly Report
55
Released
1982
s 9(2)(a)
145467
OCT2022 ASPIRATION PNEUMONITIS
* CLOZAPINE
s 9(2)(a)
77
Female s 9(2)(a)
145546
NOV2022 CARDIAC FAILURE
* CLOZAPINE
55
Male
Act
RISPERIDONE
GLICLAZIDE
VALPROATE SODIUM
VILDAGLIPTIN
145547
NOV2022 MYOCARDITIS
* CLOZAPINE
74
Male
CARDIOMYOPATHY
CARDIAC FAILURE
145548
NOV2022 VOMITING
* CLOZAPINE
71
Male
ASPIRATION
LEVODOPA/CARBIDOPA (200/50)
PNEUMONIA
ENTACAPONE
DROWSINESS
VENLAFAXINE
GABAPENTIN
Information
145549
NOV2022 PANCREAS NEOPLASM MALIGNANT * CLOZAPINE
50
Male
CACHEXIA
CLONAZEPAM
ANAEMIA
SIMVASTATIN
LACTULOSE
145550
NOV2022 CARDIAC FAILURE
* CLOZAPINE
45
Male
INSULIN GLARGINE
INSULIN ASPART
Official
145838
DEC2022 CARCINOMA COLON
* CLOZAPINE
63
Male
LAXSOL
VITAMIN D
the
145842
DEC2022 RENAL FAILURE CHRONIC
* CLOZAPINE
59
Male
145900
DEC2022 UNCONSCIOUSNESS
* CLOZAPINE
60
Male
COLLAPSE CIRCULATORY
under
HEART DISORDER
145902
DEC2022 MELANOMA MALIGNANT
* CLOZAPINE
81
Male
SERTRALINE
LORAZEPAM
LAXSOL
MARC June 2023 – CARM Quarterly Report
56
Released

1982
Act
Information
Official
the
under
Released









