 Memo
Date:
Memo
Date:
22 October 2021
To:
s 9(2)(g)(ii)
Manager, Clinical Risk Management, Medsafe
From:
s 9(2)(g)(ii)
(Advisor, Pharmacovigilance, CVIP)
Original memo prepared by s 9(2)(g)(ii)
(Advisor, Pharmacovigilance,
Medsafe)
Subject:
Comirnaty vaccine and menstrual disorders/unexpected vaginal bleeding –
UPDATE to 23 June 2021 memo
Incident ID:
N/A
Lotus Notes Location: N/A
For your:
Action: [√] Decision: [√] Information: [√]
Page 1 of 20
TABLE OF CONTENTS Description .................................................................................................................................................................................... 3
Nature of the Safety Concern ................................................................................................................................................ 3
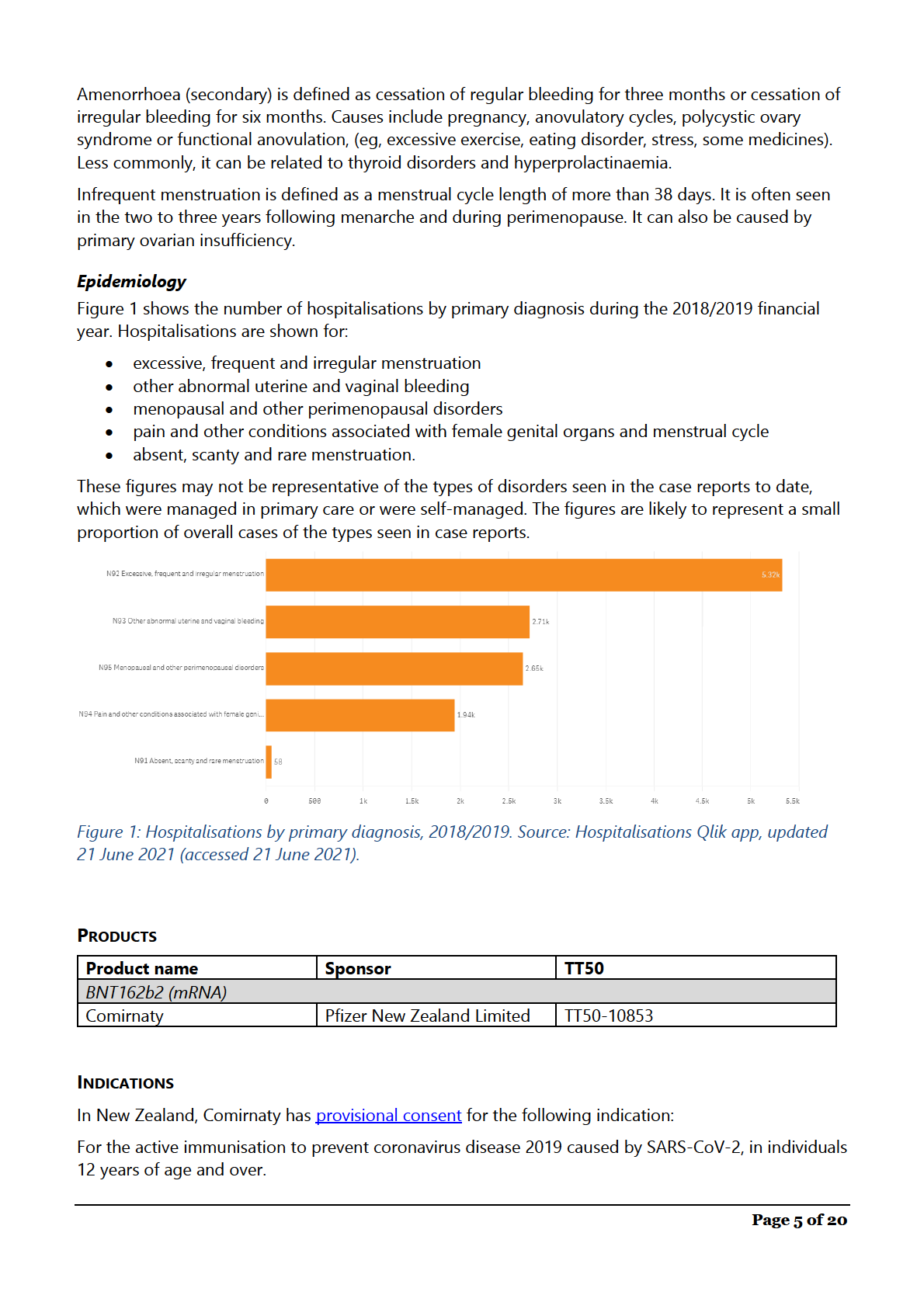
Abnormal vaginal bleeding [1] ......................................................................................................................................... 3
Diagnosis and management [1] ....................................................................................................................................... 3
Epidemiology ........................................................................................................................................................................... 5
Products ......................................................................................................................................................................................... 5
Indications ..................................................................................................................................................................................... 5
Usage Data .................................................................................................................................................................................... 6
Historical Information ............................................................................................................................................................... 7
Source of Safety Concern ........................................................................................................................................................ 7
Review of the available information .................................................................................................................................... 8
Pfizer/BioNTech Comirnaty PSUR & SMSR .................................................................................................................. 8
Review of clinical study data ......................................................................................................................................... 9
Post-marketing reports ................................................................................................................................................... 9
Observed to Expected analyses ...................................................................................................................................... 12
MHRA weekly summary of Yellow Card reporting – United Kingdom ............................................................ 13
European Medicines Agency (EMA) – European Union ........................................................................................ 14
Therapeutic Goods Administration (TGA) – Australia ............................................................................................ 14
Discussion in the literature ............................................................................................................................................... 15
VigiLyze data ......................................................................................................................................................................... 17
Public Interest ............................................................................................................................................................................ 18
Conclusions and Proposed Actions ................................................................................................................................... 19
Recommendations ................................................................................................................................................................... 19
References ................................................................................................................................................................................... 20
Page 2 of 20
DESCRIPTION
There have been reports of menstrual disorders, and other related disorders such as unexpected
vaginal bleeding, as an adverse effect following Comirnaty administration in New Zealand.
This memo reviews the information currently available on this issue and considers whether any further
action is required. This memo has been updated with the latest information since the initial memo on
the 23 June 2021.
Updated sections are:
• Usage Data
• Historical Information
• Source of Safety Concern
• Review of the available information
• Discussion in the literature
• Public Interest
• Conclusions and Proposed Actions
NATURE OF THE SAFETY CONCERN
Abnormal vaginal bleeding [1]
Per vaginum (PV) bleeding often originates from the uterus, but bleeding from the vulva, vagina or
cervix can also occur.
Heavy, irregular or missed periods can be normal for some people, or can be related to lifestyle
factors such as stress, weight loss, excessive exercise, being overweight and contraceptive use [2].
Abnormal bleeding frequency is considered to be a cycle shorter than 24 days or longer than 38 days.
Prolonged duration is longer than eight days. Irregular menstruation is a cycle length that varies by
more than 8-10 days. Flow volume is subjective.
Possible medical causes of uterine bleeding include anovulatory cycles, pregnancy, menopause,
structural abnormalities, bleeding disorders and malignancy. Possible causes of lower genital tract
bleeding include infection, trauma, urogenital atrophy or malignancy.
Diagnosis and management [1]
Differential diagnosis is guided by bleeding type:
• heavy menstrual bleeding
• intermenstrual or unscheduled/breakthrough bleeding
• post-coital bleeding (not discussed here)
• post-menopausal bleeding
• absence of bleeding.
The type of bleeding can help to identify the most likely causes. The first step is usually to exclude
pregnancy, unless the patient is post-menopausal.
History-taking should include age, menstrual bleeding patterns, characteristics and timing of bleeding,
associated symptoms, medicines use, sexual health history, obstetric history, surgical history, and
symptoms arising from systemic disease.
Medicines that may be associated with PV bleeding include hormonal contraception, menopausal
hormone therapy, anticoagulants, tamoxifen, antipsychotics and some herbal products.
Page 3 of 20
Heavy menstrual bleeding
Heavy menstrual bleeding is usually defined as a bleeding volume that interferes with quality of life, as
measurement of actual bleeding volume is usually impractical.
Heavy menstrual bleeding can be related to uterine structure and this becomes more common with
increasing age. For example, fibroids, polyps, adenomyosis, and endometrial cancer or hyperplasia.
Non-structural causes of heavy menstrual bleeding include:
• medicines (e.g. copper IUD, tamoxifen, depot medroxyprogesterone acetate, menopausal
hormone therapy, anticoagulants, aspirin, some herbal supplements)
• ovulatory dysfunction (e.g. psychological stress, weight change, excessive exercise, polycystic
ovary syndrome, thyroid disease)
• coagulation disorders
• endometrial disorders.
Investigations can include pregnancy tests, complete blood counts, thyroid-stimulating hormone,
coagulation and liver function tests, Pipelle biopsy and pelvic ultrasound.
Treatment is influenced by the cause of bleeding, need for contraception, any contraindications to
oestrogen or progestogen use, and patient preference. Treatments include:
• hormonal treatments (Mirena IUD, combined oral contraception, progestogen-only
contraceptives, cyclical progestogens)
• non-hormonal treatments (tranexamic acid, mefenamic acid)
• surgery can be considered if pharmacological treatments are ineffective.
Intermenstrual or unscheduled bleeding
Intermenstrual bleeding is any cyclic or random bleeding between menstrual periods. Unscheduled or
breakthrough bleeding occurs between withdrawal bleeds for women taking hormonal contraception
or menopausal hormone therapy.
Common causes include ovulation, sexually transmitted infections (STI), polyps, progestogen-only
contraceptives, endometrial malignancy or hyperplasia and Caesarean scar defect.
Investigations include pregnancy testing, STI testing, cervical smear testing and pelvic ultrasound, with
further investigation and appropriate treatment if results are abnormal. Some women may experience
light spotting or bleeding while ovulating. If investigations are normal, no further intervention may be
required.
Post-menopausal bleeding
Post-menopausal bleeding is defined as occurring after more than 12 months of menopausal
amenorrhoea.
The most common causes are endometrial or vaginal atrophy, menopausal hormone therapy and
polyps, endometrial hyperplasia or cancer and cervical cancer.
Investigations may include cervical smear testing, STI testing, Pipelle biopsy, pelvic ultrasound and
hysteroscopy.
Infrequent or absent menstrual cycles
Page 4 of 20
The use of this vaccine should be in accordance with official recommendations.
USAGE DATA
The COVID-19 vaccine is only approved for those aged 12 years and older.
The New Zealand immunisation programme started on 20 February 2021 with border and MIQ
workers and the people they live with. From March, this extended to high-risk frontline workers and
people living in high-risk places. On 21 June 2021, an extension of indication allowed use in
adolescents aged 12 to 15 years of age.
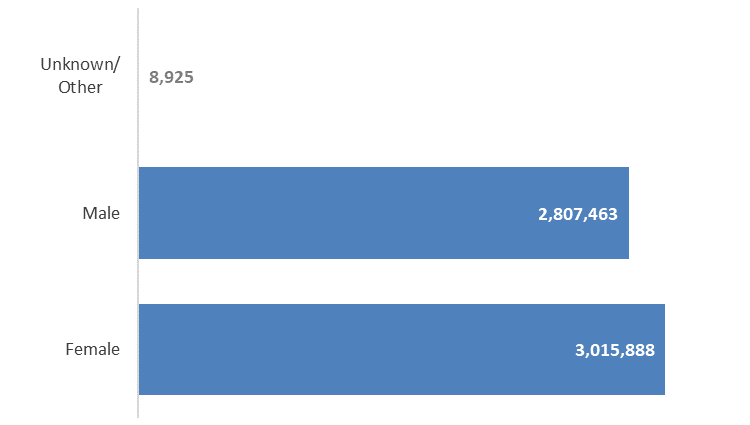
From 20 February 2021 up to and including 10 October 2021, 5,832,276 vaccine doses have been
administered. 3,015,888 doses have been administered to females as shown in Figure 2. For the
number of vaccinations administered in females broken down by ten year age group see Figure 3.
Figure 2: Vaccine doses administered by gender, 20 February 2021 to 10 October 2021. Source: COVID-
19 Vaccination Events Qlik app, updated 11 October 2021 (accessed 11 October 2021).
Page 6 of 20
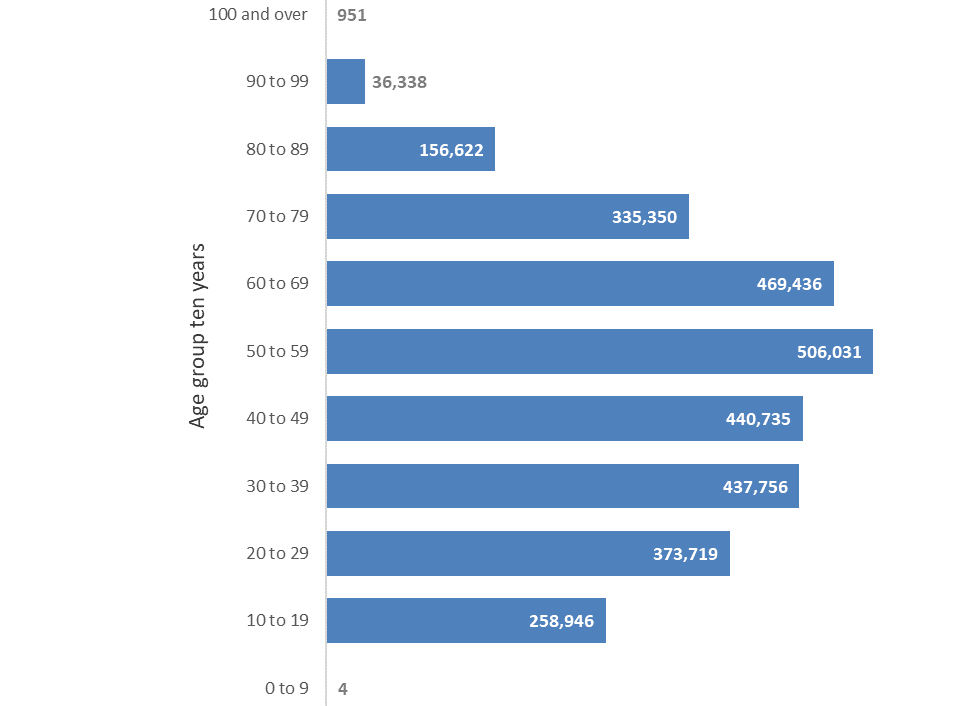 Figure 3: Vaccine doses administered to females by ten year age band, 20 February 2021 to 10 October
Figure 3: Vaccine doses administered to females by ten year age band, 20 February 2021 to 10 October
2021. Source: COVID-19 Vaccination Events Qlik app, updated 11 October 2021 (accessed 11 October
2021).
HISTORICAL INFORMATION
Menstrual disorder and similar terms are not listed in the New Zealand Comirnaty data sheet. This
concern was previously reviewed by Medsafe on 23 June 2021 and was presented to the COVID-19
Independent Safety Monitoring Board (CV-ISMB) on 24 June 2021. At that time the evidence did not
suggest an increased risk of menstrual disturbances or unexpected vaginal bleeding following
vaccination with Comirnaty, and a recommendation was made to continue to monitor this topic
through routine pharmacovigilance. A short statement outlining this was published in Medsafe Safety
Report #14 on 30 June 2021.
SOURCE OF SAFETY CONCERN
The source of this safety concern is spontaneous adverse reaction reports received in New Zealand.
Since this topic was originally reviewed on 23 June 2021, there has been an increase in the number of
reports to the Centre for Adverse Reactions Monitoring (CARM). As at 7 October 2021 there have
been 503 reports of menstrual disturbances or unexpected vaginal bleeding.
The majority of reports occurred after dose 1 (77%). The average age of reporters is 37 years (range 14
to 79 years).
The most commonly reported manifestations are:
• Heavier than usual menstrual bleeding (137 reports)
Page 7 of 20
• Unexpected vaginal bleeding or spotting soon after vaccination (260 reports). This includes
people who experienced menstrual bleeding earlier than expected, intermenstrual bleeding,
breakthrough bleeding on oral contraception, or bleeding in people who are normally
amenorrhoeic on various forms of contraception.
• Late or missed periods (92 reports)
• More painful than usual menstruation (69 reports)
• Irregular cycles since vaccination (12 reports).
• Post-menopausal bleeding (45 reports).
Note there may be some overlap, as some people reported multiple manifestations (for example,
heavy prolonged period). Other reports included lighter or shorter than usual menstrual periods,
irregular periods or cycles, and unexpected return of menstruation post-partum.
Cases of note include:
s 9(2)(a)
REVIEW OF THE AVAILABLE INFORMATION
s 9(2)(ba)(i)
Page 8 of 20
s 9(2)(ba)(i)
Page 9 of 20
s 9(2)(ba)(i)
Page 10 of 20
s 9(2)(ba)(i)
Page 11 of 20
s 9(2)(ba)(i)
Page 12 of 20
s 9(2)(ba)(i)
MHRA weekly summary of Yellow Card reporting – United Kingdom
The MHRA has issued a brief statement on menstrual disorders and unexpected vaginal bleeding, as
part of the Coronavirus vaccine - weekly summary of Yellow Card reporting (see Figure 4). The report
covers the period 9 December 2020 to 29 September 2021.
The MHRA did not consider that there appeared to be an increased risk after vaccination, and that
numbers of reports were low considering the number of females who have received the vaccine and
how common menstrual disorders are generally [5].
Page 13 of 20
 Figure 4: MHRA statement on menstrual disorders and unexpected vaginal bleeding 7 October 2021 [3]
European Medicines Agency (EMA) – European Union
Figure 4: MHRA statement on menstrual disorders and unexpected vaginal bleeding 7 October 2021 [3]
European Medicines Agency (EMA) – European Union
The EMA publishes COVID-19 vaccine safety updates for Comirnaty following the outcome of
assessments carried out by the EMA’s Pharmacovigilance Risk Assessment Committee (PRAC). In their
meeting dated 5 August 2021, the PRAC discussed reported cases of menstrual disorders occurring
after COVID-19 vaccinations.
The PRAC concluded that “No causal association between COVID-19 vaccines and menstrual disorders
has been established so far.
Menstrual disorders are very common and can occur without an underlying medical condition. Causes
can range from stress and tiredness to underlying medical conditions, such as fibroids and
endometriosis. Women experiencing unexpected vaginal bleeding (e.g. in postmenopausal women) or
who are concerned about prolonged or severe menstrual disturbances may want to seek medical
advice.
The marketing authorisation holders for all COVID-19 vaccines approved in the EU have been asked to
provide further data as part of the monthly summary safety reports. The PRAC will review all available
evidence, including reports of suspected adverse events and scientific literature and will continue
monitoring the issue.”
Therapeutic Goods Administration (TGA) – Australia
The TGA is continually monitoring the safety of the COVID-19 vaccines and in their weekly safety
report dates 26 Aug 2021, menstrual disorders was mentioned.
The statement reads “The TGA recently carried out an investigation of menstrual problems (also
known as period problems) and unexpected vaginal bleeding suspected to be related to COVID-19
Page 14 of 20
vaccination. This was in response to increased reporting of these events in Australia and
internationally. The investigation did not find evidence for a link between vaccination and menstrual
problems, but we continue to monitor this issue as part of our routine vaccine surveillance activities.
The TGA’s investigation involved assessing rates of reporting within Australia and overseas, reviewing
findings from other regulators and examining the medical literature. To 22 August 2021, we have
received 322 reports detailing a menstrual disorder or unexpected vaginal bleeding following the
Comirnaty (Pfizer) vaccine, 157 reports following the Vaxzevria (AstraZeneca) vaccine and 2 reports
where the vaccine brand was not specified. The most commonly reported symptoms were heavy
periods, irregular bleeding, bleeding between periods and painful periods. Vaginal bleeding in
postmenopausal women has also been reported.
Evidence from this review does not support a link between vaccination and menstrual problems. The
Medicines and Healthcare products Regulatory Agency in the UK and the European Medicines
Agency’s Pharmacovigilance Risk Assessment Committee came to similar conclusions following their
own rigorous investigations.
Abnormal menstrual bleeding is very common regardless of vaccination and affects up to a quarter of
women during their reproductive years. Many factors can cause period problems including stress,
weight gain or loss and underlying conditions like polycystic ovary syndrome and uterine fibroids.
Changes to the menstrual cycle have also been reported after infection with COVID-19 and in people
with ‘long COVID’.
Women who experience changes to their periods that are unusual for them and persist over time
should talk to their doctors. Any vaginal bleeding after menopause is considered abnormal and
warrants medical attention.”
DISCUSSION IN THE LITERATURE
Letter to the editor (BMJ) –18 April 2021. Merchant, H. “CoViD-19 post-vaccine menorrhagia,
metrorrhagia or postmenopausal bleeding and potential risk of vaccine-induced thrombocytopenia in
women” [6]
This letter was submitted by the Subject Leader in Pharmacy at the University of Huddersfield, United
Kingdom in response to an opinion piece titled ‘Thrombosis after covid-19 vaccination: these rare
events must not derail vaccination efforts’ [7].
The letter stated that there have been many reports of post-vaccination menstrual irregularities
submitted in the UK, with twice as many reports submitted for the Vaxzevria (AstraZeneca) than
Comirnaty. The writer notes that the number or reports is likely to be an underestimate as many
events may not be reported.
The writer states that there have been reports of haemorrhage, blood clots and thrombocytopaenia
following COVID-19 vaccination. The European Summary of Product Characteristics (SmPC) for
Vaxzevria has been updated to include thrombocytopaenia as an adverse reaction, with the frequency
‘common’.
The writer believes that vaccine-induced thrombocytopenia may be an explanation for reports of
heavy menstrual bleeding.
Comment: In the UK, more doses of Vaxzevria have been administered (approximately 42.3 million
doses) than Comirnaty (approximately 26.4 million doses) [3]. This may explain the difference in the
numbers of reports.
Page 15 of 20
Expert opinion (Journal of Pediatric and Adolescent Gynecology) – August 2021. NASPAG.
“NASPAG Position Statement on COVID-19 Vaccines and Gynecologic Concerns in Adolescents and
Young Adults” [7]
The North American Society for Pediatric and Adolescent Gynecology (NASPAG) has produced a
Position Statement relating to COVID-19 vaccine use in adolescents and young adults, based on
available data and expert opinion.
They noted anecdotal and media reports of irregular menstrual cycles, including skipped periods,
prolonged periods, and spotting after COVID-19 vaccines, and similar reports of changes in menstrual
cycles after COVID-19 infection. Authors stated that irregular periods are a common part of
adolescence and are part of normal pubertal development. Additionally, infections, immune reactions,
and fevers are understood to cause short-term, self-limited changes in cycles. They recommended
eligible adolescents are vaccinated regardless of stage of puberty. They also noted there was no
scientific data demonstrating a link between COVID-19 vaccine and fertility.
Editorial (BMJ, Clinical Research Ed.) – 16 September 2021. Male, V. “Menstrual changes after
covid-19 vaccination” [8]
This editorial was written by Dr Victoria Male, a lecturer in reproductive immunology at the Imperial
College London. She stated that primary care clinicians and reproductive health professionals are
increasingly approached by people experiencing these symptoms after COVID-19 vaccination, and
that 30,000 reports of these events had been submitted to the MHRA. She acknowledged that the
menstrual cycle can be affected by other immune stimuli such as infections or other vaccines (e.g. HPV
vaccine) and outlined possible mechanisms, including immunological influences on hormones or on
the cells lining the uterus, and that these changes are usually short-lived.
The writer called for menstrual changes to be thoroughly investigated as concerns about potential
impacts on fertility are driving vaccine hesitancy amongst young women.
Comment: From New Zealand data on the Gardasil vaccine, there has been infrequent reporting of
menstrual disorders/abnormalities following vaccination. Additionally, it is not listed in the data sheet.
The study cited by the author is a questionnaire of young people (9-15 year olds) and there is likely to be
significant confounding.
PREPRINT: Mixed methods survey (medRxiv) – 12 October 2021. Lee, K. M. et al. "Characterizing
menstrual bleeding changes occurring after SARS-CoV-2 vaccination" [9]
Lee et al., performed an exploratory survey mixed methods survey in current and formerly
menstruating adults in the USA. The survey was advertised on social media. All participants were fully
vaccinated (at least fourteen days after all required doses) and had not had COVID-19 infection
(diagnosed or suspected). 39,129 participants aged between 18 to 80 years old (mean age=34.2 years,
SD=9.2) were included in the analysis. Respondents in this sample were vaccinated with Pfizer
(N=21,620), Moderna, (N=13,001), AstraZeneca (N=751), Johnson & Johnson (N=3,469), Novavax
(N=61), other (N=204), or unknown (N=23) vaccines.
The current analysis focussed on two subgroups identified in the first 3 months of data collection:
• Bleeding flow/heaviness in people who menstruate regularly – pre-menopausal (ages 18-45
years) (N=21,380)
• Breakthrough bleeding in people who do not regularly menstruate – pre-menopausal (ages
18-45 years) on long-term hormone treatments that suppress menstruation (N=270 on
Page 16 of 20
gender-affirming treatment; N=1545 on long-acting reversible contraception (LARC)), or post-
menopausal (ages 55-80 years) with no periods for at least 12 months (N=238).
Approximately 40% of respondents who were regularly menstruating experienced heavier flow after
Pfizer vaccination. A proportion experienced no change in flow, and the remainder experienced lighter
flow. These proportions were similar in people with and without previously diagnosed reproductive
conditions (such as endometriosis, PCOS, fibroids), and in people who were spontaneously
menstruating versus those on hormonal contraception. 70% of respondents on LARC, 39% of
respondents on gender-affirming treatment, and 66% of respondents who were post-menopausal
experienced breakthrough bleeding after Pfizer vaccination. They found no differences in menstrual
bleeding changes between Pfizer and Moderna.
Comment: This a large self-selected population of people in the USA who responded to a survey posted
on social media. Survey design does not allow for calculation of incidence rates or assessment of
causality, but this survey included a large number of people and gives us a sense of the number of
people affected by menstrual cycle changes.
Online survey (Frontiers in Medicine) – 8 October 2021. Alghamdi, A.N. et al. "BNT162b2 and
ChAdOx1 SARS-CoV-2 Post-vaccination Side-Effects Among Saudi Vaccinees" [10]
Alghamdi et al., conducted an online questionnaire to screen the local, systemic, and allergic post
vaccination reactions for vaccinees who received either one or two doses of the BNT162b2 vaccine or
one dose of the ChAdOx1 vaccine. A link to the online questionnaire was distributed in different social
media including Twitter, Snapchat, and WhatsApp.
2874 participants had received at least one dose of BNT162b2. Of these participants, 95% reported
they were Saudi, 44% were aged between 20-30 years, 71% were female, and 83% indicated they had
no comorbidities. 86% of respondents reported that they experienced side effects after vaccination
with BNT162b2.
Participants reported menstrual cycle abnormalities, including increases in the duration of, or pain
associated with bleeding. There were 18 reports from people after BNT162b2 vaccination (0.7%)
compared to 7 reports from people after ChAdOx1 vaccination (0.5%).
Comment: This a small self-selected population of predominantly young Saudi people who responded to
a survey posted on social media. Survey design does not allow for calculation of incidence rates or
assessment of causality.
s 9(2)(ba)(i)
Page 17 of 20
s 9(2)(ba)(i)
PUBLIC INTEREST
There is significant public and media interest in this topic, with accounts of menstrual irregularities
published on social media platforms.
For example, the Guardian published an article in April about anecdotal accounts of disrupted
menstrual cycles [11]. Dr Kate Clancy, an associate professor at the University of Illinois, and Dr
Katharine Lee, a postdoctoral researcher at Washington University School of Medicine, have started a
survey to explore these accounts. The article emphasised that menstrual changes are reported to be
short-lived and do not appear to affect large numbers of people. Many people experience variations
between menstrual cycles and patterns can be affected by other factors such as stress.
A more recent article published by the BBC in September also reported on anecdotal reports of
menstrual cycle abnormalities following COVID-19 vaccination. [12] The article included statements
from Dr Jo Mountfield, vice president of the Royal College of Obstetricians and Gynaecologists
(RCOG) and Dr Victoria Male, from Imperial College London, emphasising that menstrual disorders are
common and can have many causes; that any changes after COVID-19 vaccination are likely to be
temporary; and that there is no evidence that these temporary changes will have any impact on future
fertility.
Page 18 of 20
Public interest may stimulate reporting of menstrual disturbances and unexpected vaginal bleeding.
Some of the New Zealand case reports referred to hearing about the experiences of other women.
CONCLUSIONS AND PROPOSED ACTIONS
Currently, there is insufficient information to confirm a signal of menstrual disturbances or unexpected
vaginal bleeding with Comirnaty. The Sponsor performed an in-depth analysis of heavy menstrual
bleeding and postmenopausal bleeding, and they did not find a signal.
As at 7 October 2021, CARM had received 503 reports of menstrual disturbances or unexpected
vaginal bleeding with Comirnaty. These included heavy, light, delayed and early menstrual periods,
and worse pain associated with menstruation. Unexpected vaginal bleeding was also reported in
women who usually don’t bleed due to their contraceptive method, and in postmenopausal women.
Given that several thousand women are hospitalised each year (and likely many more present to
primary care) due to menstrual disorders or other abnormal PV bleeding, the volume of reports
received to date is not unexpected. There is evidence to suggest that menstrual cycle abnormalities or
unexpected vaginal bleeding after COVID-19 vaccination may be a result of immunological influences
on hormones responsible for menstruation or the cells lining the uterus, as is known to occur with
infections, other vaccines, or stress.
Medsafe will continue to monitor this issue through routine pharmacovigilance activities. This includes
monitoring New Zealand case reports, safety reports from the Sponsor, action from other regulators
and information in the literature.
RECOMMENDATIONS
It is recommended that:
1.
This update is presented to the COVID-19 Vaccine ISMB.
Yes
2.
This topic continues to be monitored through routine
Yes
pharmacovigilance.
Page 19 of 20
REFERENCES
[1] bpacnz, “Investigating and managing abnormal vaginal bleeding: an overview,” December 2019.
[Online]. Available: https://bpac.org.nz/2019/bleeding.aspx. [Accessed 21 June 2021].
[2] National Health Service, “Stopped or missed periods,” 2 August 2019. [Online]. Available:
https://www.nhs.uk/conditions/stopped-or-missed-periods/. [Accessed 22 June 2021].
[3] van den Brink, M. J. et al., “Incidence and treatment of heavy menstrual bleeding in general
practice,”
Family Practice, 2017. 34:6(673-678). DOI: https://doi.org/10.1093/fampra/cmx050.
[4] Burbos, N. et al. , “Age-related differential diagnosis of vaginal bleeding in postmenopausal
women: a series of 3047 symptomatic postmenopausal women,”
Menopause International, 2010.
16:1(5-8). doi:10.1258/mi.2010.010005.
[5] Medicines & Healthcare products Regulatory Agency, “Coronavirus vaccine - weekly summary of
Yellow Card reporting,” 7 October 2021. [Online]. Available:
https://www.gov.uk/government/publications/coronavirus-covid-19-vaccine-adverse-
reactions/coronavirus-vaccine-summary-of-yellow-card-reporting#yellow-card-reports.
[Accessed 11 October 2021].
[6] Merchant, H., “Rapid Response: CoViD-19 post-vaccine menorrhagia, metrorrhagia or
postmenopausal bleeding and potential risk of vaccine-induced thrombocytopenia in women
[Letter to the editor],”
BMJ, 2021. 373 (n958). DOI: 10.1136/bmj.n958.
[7] Hunter, P. R., “Thrombosis after covid-19 vaccination,”
BMJ, 2021. 373(n958). doi:
https://doi.org/10.1136/bmj.n958.
[8] Male, V., “Menstrual changes after covid-19 vaccination,”
BMJ (Clinical Research Ed.), 2021.
374(n2211). DOI: https://dx.doi.org/10.1136/bmj.n2211.
[9] Lee, K. M. et al., “Lee, K. M., et al. (2021). "Characterizing menstrual bleeding changes occurring
after SARS-CoV-2 vaccination.",”
medRxiv (PREPRINT), 2021. DOI: 10.1101/2021.10.11.21264863.
[10] Alghamdi, A. N. et al., “BNT162b2 and ChAdOx1 SARS-CoV-2 Post-vaccination Side-Effects
Among Saudi Vaccinees.,”
Frontiers in Medicine, 2021. 8(1796) DOI: 10.3389/fmed.2021.760047.
[11] Davis, N., “Any menstrual changes after Covid jab would be short-lived, experts say,” The
Guardian, 4 June 2021. [Online]. Available:
https://www.theguardian.com/society/2021/jun/04/any-menstrual-changes-covid-jab-would-be-
short-lived-experts. [Accessed 16 June 2021].
[12] BBC News, “Call for investigation of menstrual changes after Covid jabs,” 16 September 2021.
[Online]. Available: https://www.bbc.com/news/health-58573593. [Accessed 20 October 2021].
[13] NASPAG, “NASPAG Position Statement on COVID-19 Vaccines and Gynecologic Concerns in
Adolescents and Young Adults,”
Journal of Pediatric and Adolescent Gynecology, 2021. 34(4): 439-
440 DOI: 10.1016/j.jpag.2021.05.008.
Page 20 of 20




