Document 1
1
Transitioning to COVID-19 endemicity: Laboratory considerations. 21/09/21
Report prepared by Dr Gary McAuliffe, with contributions from Dr Susan Morpeth, Dr Michael
Addidle and the New Zealand Microbiology Network
Executive summary
• In order to control COVID-19 transmission testing strategies need to adapt to meet changing
needs.
• Test performance for detection of infection becomes less critical than ability to detect
infectiousness, speed and availability of tests, increasing the applicability of rapid antigen
tests, self-testing methods and saliva PCR testing.
• An overarching strategy is needed which indicates why and how different tests would be
used and what the actions would be for positive test results under different scenarios as we
transition to endemic COVID-19; as well as the trigger points for each phase, in order to
build appropriate resources across the stakeholders involved.
• NZ’s current testing capacity is a third of the equivalent per-population testing being carried
out in Denmark, a country with high vaccination rates and endemic COVID-19.
• Upscaling of PCR test capacity and introduction of rapid antigen testing will be needed to
bridge this gap.
• This work needs to be done now with urgency to prepare for endemic COVID-19 and will
require significant investment in equipment and staff, pilot studies, and operational
planning.
Background
Several countries previously pursuing elimination approach e.g. Singapore, Australia have signalled
their intentions to move to COVID endemic or resilience phases once their vaccinated population
reaches a certain threshold. In Australia this signal has come prior to reaching target population
vaccination due to lockdown fatigue and inability to achieve elimination. Other countries pursuing
mitigation strategies e.g. the UK, Denmark have signalled a shift in public health approach with
increasing levels of immunity due to vaccination and prior infection. These changes in approach have
consequences for testing strategy and required laboratory capacity, in both the transition phase, and
the endemic phase. However, it is clear that in order to avoid overwhelming healthcare, testing will
under the Offical Information Act 1982
remain a key pillar of the response. We explore this and what that would require using examples
from overseas including New South Wales, Denmark, Singapore and the United Kingdom.
Released
1
Document 1
2
Table. Current COVID-19 cases, vaccination and testing situations in NZ, NSW and Denmark, current
as of 21/9/21
NZ current
New South
Denmark
Singapore
Wales
Population:
5,122,600
8,200,000
5,816,000
5,690,000
Fully
1,618,673
3,482,945
4,210,892
4,455,536
vaccinated:
(32% total
(43% total
(72% total
(78% total
population)
population)
population)
population)
Cases: (7 day
21
1194
850
1178
average)
Antigen
0
Not
82,212
Not
tests/day (7
publically
publically
day average)
available
available
PCR tests/day
12,971
143,235
70,723
67,100
(7 day
average)
Total
12,971
143,235
152935
67,100
tests/day
(0.25%
(1.7%
(2.6%
(1.2%
popn/day)
popn/day)
popn/day
popn/day)
Tests per case 597
120
180
56.9
https://www.moh.gov.sg/news-highlights/details/update-on-local-covid-19-situation-(21-sep-2021);
https://www.sst.dk/en/english/corona-eng/status-of-the-epidemic/covid-19-updates-statistics-and-
charts; https://www.health.nsw.gov.au/Infectious/covid-19/Pages/default.aspx;
https://www.health.govt.nz/our-work/diseases-and-conditions/covid-19-novel-coronavirus/covid-
19-data-and-statistics/testing-covid-19
Changes to testing strategy
Based on overseas experience (see appendices) elements of NZ’s testing strategy will change; these
include a lesser focus on the sensitivity of the test to pick up all individuals with COVID-19, compared
with factors such as tolerability, rapidity and accessibility e.g. rapid antigen testing, saliva PCR
testing, self-testing.
The European CDC described testing strategies and objectives for COVID-19 in September 2020
which are important considerations as public health strategy changes from an elimination approach,
under the Offical Information Act 1982
key relevant points over and above NZ’s current testing focuses are:
All people with COVID-19 symptoms should be tested as soon as possible which requires easy access
to testing for all, including non-residents. Test turnaround time should be minimised, people testing
positive should isolate and timely contact tracing should be carried out, ensuring that all close
contacts are tested, irrespective of symptoms.
Healthcare and social care settings require intensive testing when there is documented community
Released
transmission. Periodic and comprehensive testing of all staff and residents/patients is recommended
to prevent nosocomial transmission. Furthermore, all patients/residents should be tested upon or
immediately prior to admission.
2
Document 1
3
Clusters or outbreaks may occur in certain settings, such as workplaces, healthcare facilities,
educational facilities, prisons, and migrant detention centres. Testing policies and systems should be
in place for rapid detection and control to protect the relevant populations in these settings and to
protect the community from amplified transmission.
In the face of increasing COVID-19 prevalence, addressing these elements requires increased
laboratory capacity for PCR testing, particularly for testing symptomatic individuals, and routine
surveillance testing of patients and hospital staff. PCR will also be required to confirm positive RAT
results and for testing of contacts of cases. Further details are available in the appendix.
Laboratory capacity requirements
In the transition phase and early to mid-endemic phase, sustained high rates of laboratory based
PCR testing will be needed in order to support mitigation of transmission.
In New South Wales currently, 143,235 tests have been performed on average each day for the last
seven days. The equivalent, relative to the NZ population, would be 88,805 tests per day. When
compared with the 49,736 performed on the largest day by NZ labs in the current outbreak, this
indicates a significant gap in capacity. Test capacity requirements should be modelled based on this
scenario as an indicator of required surge capacity, particularly during transition and early endemic
phase. Additional modelling, including the impact of healthcare worker screening on laboratory
capacity and ability to maintain the workforce is also necessary.
Thinking beyond a transition period, in Denmark, the equivalent number of PCR tests that would
need to be performed sustainably in NZ would be 62,236 tests.
Table. NZ current and future projected testing requirements
NZ current
NZ testing capacity
NZ testing
NZ testing
requirements
Capacity
capacity
equivalent to NSW
requirements
requirements
equivalent to
equivalent to
Denmark
Singapore
Baseline
62% of NSW testing 88% of Denmark
90% of
testing
Singapore
testing
under the Offical Information Act 1982
Antigen tests/day
0
Not recorded
72,346
Not recorded
(7 day average)
PCR tests/day (7
12,971
88,805
62,236
60,390
day average)
Total tests/day
12,971
88,805
134,582
60,390
(maximum daily
tests performed=
49,736)
Released
Consideration needs to be given to the capacity of the different NZ regions, as Auckland has
performed 56% of the country’s tests but has also relied on sending tests elsewhere. With endemic
COVID-19 this may not be possible as the regions will need to support their own public health
3
Document 1
4
interventions, necessitating higher capacity in the Auckland region e.g. 36-52,000 tests/day based on
the case scenarios described. The regions themselves have not yet been pressure tested.
We also anticipate current laboratory capacity will need to transition to the ability to incorporate
saliva-based PCR testing as a higher proportion of workload. Whilst this work is ongoing, and
generally capacity is increasing, faster progress and strategic leadership is needed in this area.
What resources will laboratories need to upscale and maintain PCR testing?
Several laboratories are currently looking at this. In order to upscale and maintain ability to test the
projected high numbers of PCR tests the key elements are:
Staff. A sustainable workforce, taking into account the peaks and troughs of test volumes. A mobile
molecular workforce that can ebb and flow into other areas of the lab as required. Requires
investment beyond the short term requirements.
Space. Many laboratories are unable to expand and work safely within their existing environments,
and assistance to rapidly source and outfit new suitable areas.
Equipment. Automating as much of the process as possible. These platforms are expensive but are
necessary in order to upscale and maintain capacity. Further PCR machines may be required in larger
laboratory hubs and smaller laboratories in the regions e.g. Northland, Taranaki to reduce
turnaround time to results. Diversity of platforms has been a strength, due to supply constraints,
equipment downtime etc. and needs to be an ongoing consideration.
Supply chain. National oversight to help with negotiations with suppliers/mitigate stock shortages,
watch global supply for key reagents
We recommend all labs work regionally and nationally to scope their requirements based on the
estimates above, with clear accountability lines for resourcing and investment e.g. from the Ministry
of Health or District Health Boards/Health NZ.
Rapid antigen testing
The elimination approach in NZ to date has necessitated the exclusive use of high sensitivity PCR
laboratory-based assays in order to minimise the risk of false negative results (See NZMN position
under the Offical Information Act 1982
statement on RATs). A COVID-endemic setting will likely require the use of testing modalities outside
of laboratories e.g. rapid antigen testing (RAT), in order to help mitigate the impact of COVID-19 on
healthcare and the wider activities of the population.
There are several different potential applications of RATs (see appendices for specific examples):
• Workplace surveillance, particularly in “higher risk” workplaces, e.g. dine-in food and
beverage establishments, personal care services, and gym and fitness studios, amongst
Released
others.
• Surveillance in educational settings
• Pre-event surveillance testing
• Surveillance prior to crossing state boundaries
• Testing of “low risk” COVID contacts
4
Document 1
5
• Rapid diagnosis of symptomatic people. Would require to be done in parallel with PCR.
Could be used in hospital admission setting or where investigating outbreaks in institutions
such as remand facilities and aged residential care
When used for surveillance purposes, using RATs at higher frequency, e.g. 2-3 times per week, can
counteract their reduced sensitivity compared to PCR testing.
We anticipate RATs will be needed to combat staff shortages, both in healthcare and the wider
workforce by testing before coming to work to avoid stand-down amongst other workers.
It is notable that in the UK only 14% of 691 million RATs have been registered as used by 26th May
2021 and it is therefore difficult to know how they have been used and what the impact has been. It
is therefore important to have a system for recording results at a national level for visibility and
impact assessment. There is also a need to support access to testing in vulnerable groups. Language
barriers and health literacy are significant risks with at home testing.
Another challenge is integrating RATs into policy within and outside the healthcare setting.
Maintaining quality, recording results, and where funding for these tests will come from all need
consideration. Whilst testing of prisoners and those in social care settings may fall under the
mandate of public policy, does testing of workers outside these spheres fall on the businesses
themselves under a mandate from central government?
Whilst NZ labs should initially be involved in RAT piloting and rollout, consideration needs to be
given how these will be governed and monitored when widespread as this will fall outside laboratory
capability and remit.
Other laboratory considerations
Whole genome sequencing technology should be more widely available for surveillance of variants
and supporting contact tracing efforts using low complexity platforms in regional hubs in
conjunction with ESR’s bioinformatics pipeline.
Immunity testing may play an important part in determining risk of outbreaks in certain geographical
areas and for certain individuals exposed to the virus.
under the Offical Information Act 1982
With border reopening in the future, influenza and RSV testing should be available to detect and
differentiate COVID-19, RSV and influenza infections.
Information technology is critical for registering, tracking and reporting results for COVID-19 and we
need to build on existing capabilities to ensure a robust national system for this which includes RATs.
Conclusions/strategic needs
Released
There is a need for an overarching strategy across labs, health, and government to incorporate
testing through healthcare and wider society.
A mixture of PCR and RAT will be required for endemic COVID-19. Fast results (and action based on
the results) and low resource impact become more important than sensitivity; self-testing and saliva
5
Document 1
6
based testing should be available, though for scalability across the country nasopharyngeal swab
testing remains an important modality.
Implementation planning for implementing RATs is required for intended use scenarios including
frequent testing in healthcare workers, schools, work places, mass events. These require piloting
now as time is not on our side.
PCR testing capacity needs further upscaling—ability to surge comfortably to 90,000 tests per day
(50,000 in Auckland region) which requires investment in space, staff, robotics and local, regional
and national coordination.
Whole genome sequencing capability should be increased across the laboratory network.
Summary
We should urgently plan for an alternative testing strategy for NZ in order to have the key elements
available should widespread community transmission arise. This is one part of an overarching
national plan that is needed addressing healthcare, social care, workforce, and productivity.
Investment in the elements required to increase and sustain additional laboratory capacity is
needed. A mixture of PCR and RAT will be required and RAT testing should be piloted in use case
scenarios now. The funding, recording, governance of any RAT based testing outside traditional
healthcare settings e.g. at mass events, workplaces requires consideration.
under the Offical Information Act 1982
Released
6
Document 1
7
Appendices
Appendix 1.
Principles of testing strategies (from ECDC COVID-19 testing strategies and objectives. 15 Sep 2020)
https://www.ecdc.europa.eu/en/covid-19/surveillance/testing-strategies
• ECDC proposes five main objectives for testing:
o in order to control transmission;
o reliably monitor SARS-CoV-2 transmission rates and severity;
o mitigate the impact of COVID-19 in healthcare and social care settings;
o detect clusters or outbreaks in specific settings and
o maintain sustained control of COVID-19 once achieved.
• Testing strategies should be flexible and rapidly adaptable to change, depending on the local
epidemiology, transmission, population dynamics and resources.
• Ideally, all people with COVID-19 symptoms should be tested as soon as possible after
symptom onset.
• This requires easy access to testing for all, including non-residents. Test turnaround time
should be minimised, people testing positive should isolate and timely contact tracing
should be carried out, ensuring that all close contacts are tested, irrespective of symptoms.
• All patients with acute respiratory symptoms in hospitals and other healthcare settings, and
all specimens from sentinel primary care surveillance should be tested for both SARS-CoV-2
and influenza during the influenza season to monitor incidence and trends over time.
• Healthcare and social care settings require intensive testing when there is documented
community transmission. Periodic and comprehensive testing of all staff and
residents/patients is recommended to prevent nosocomial transmission. Furthermore, all
patients/residents should be tested upon or immediately prior to admission.
• Clusters or outbreaks may occur in certain settings, such as workplaces, educational
facilities, prisons, and migrant detention centres. Testing policies and systems should be in
place for rapid detection and control to protect the relevant populations in these settings
and to protect the community from amplified transmission.
• Countries experiencing high SARS-CoV-2 transmission in a local community should consider
testing the whole population of the affected area. This would enable identification of
infectious COVID-19 cases and allow for their prompt isolation to interrupt chains of
under the Offical Information Act 1982
transmission. Depending on the epidemiological situation, size and population density of the
affected area, such an approach could be less disruptive for society than having to introduce
and ensure compliance with more stringent public health measures.
• To prevent re-introduction, countries or sub-national areas that have achieved sustained
control of SARSCoV-2 circulation should, in addition to quarantine measures, consider
targeted testing and follow-up of individuals coming from other areas within the within the
same country, or from other countries that have not yet achieved sustained control of the
Released
virus.
Appendix 2.
Case study: New South Wales
7
Document 1
8
New South Wales; transitioning to endemic COVID in context of an outbreak. Currently in
suppression phase. Likely represents surge capacity needed in short term for NZ transition.
High levels of PCR testing on a per population basis compared with NZ.
Additional use of RATs e.g. in unvaccinated aged care sector workers:
https://www.health.gov.au/news/announcements/rapid-antigen-testing-kits-for-high-risk-nsw-local-
government-areas
Table: Vaccination, cases and tests in New South Wales
New South Wales
Population:
8,200,000
Fully vaccinated:
3,482,945 (42.5% total
population)
Cases: (7 day average)
1194
Antigen tests/day (7
Not publically available
day average)
PCR tests/day (7 day
143,235
average)
Total tests/day
143,235 (1.7%
popn/day)
Tests per case
120
Appendix 3.
Case study: Denmark
Denmark has transitioned to endemic COVID
Use of RATs appears to be widespread for asymptomatic testing, particularly in those attending
workplaces. PCR appears to be used for symptomatic individuals, those who are close contacts of
cases, and those with a positive RAT.
From COVID-19 Surveillance, Danish Health Authority:
https://www.sst.dk/en/english/corona-
eng/status-of-the-epidemic/covid-19-updates-statistics-and-charts
under the Offical Information Act 1982
Testing Guidance from
: https://www.sst.dk/en/English/Corona-eng/Symptoms_tested-positive-or-a-
close-contact/On-being-tested
Table. Vaccination, cases and tests in Denmark
Denmark
Population:
5,816,000
Fully
4,210,892 (72 %
Released
vaccinated:
total population)
Cases: (7 day
850/ day
average)
Antigen
82,212
tests/day (7
8
Document 1
9
day average)
PCR tests/day
70,723
(7 day
average)
Total tests/day 152935= 2.6%
population/day
Tests per
180
case=
Appendix 4.
Case study: COVID Rapid Antigen Test Rollout Singapore
Singapore, with a population of 5.7 million people, are currently in the early transition phase
towards living alongside COVID. This Public Health plan seems to be labelled “
COVID Resilience”.
However, the Singapore government are quite clear about the fact that they are now transitioning to
endemic COVID. Vaccination rates are high within Singapore, with 78.5% of their total population
having been vaccinated against COVID-19 (as of 11th Sept)
Singapore have started rolling ou
t Rapid Antigen Tests (RATs/ARTs) to Households, Workplaces
&ECEs/primary schools. They are also being used as a test option (along with PCR) for unvaccinated
attendees at mass gatherings.
They have also been used in parallel with PCR testing to rapidly screen a prison population following
a positive case.
Most of the Rapid Antigen Testing is performed by self-testing, with the exception being if it is prior
to a formal mass gathering (see below). If the rapid antigen test is positive, the process is to take a
photograph of the RAT result along with identification and call/attend a “Swab and Send Home”
clinic for a confirmatory PCR test. Test data is only collected on positive RAT results, not negative.
They are recommending RATs for
asymptomatic people in the following situations:
Households
Households will get 6 RAT tests issued per household, funded from the government. Any additional
under the Offical Information Act 1982
tests that are required are to be purchased. The indications for the use of these tests in the
household setting are a little unclear. To be used if in a “high-risk situation”, or if there is a possible
contact with a positive COVID case. However, if symptomatic, then patient should
not use a RAT and
should attend
a “Swab and Send Home” (SASH) clinic for PCR testing. Rapid antigen tests are being
rolled out to all households from 28th Aug to 27th Sept 2021.
Workplaces
Released
High risk workplaces are undergoing (asymptomatic) surveillance by Rostered Routine Testing (RRT),
the frequency of which is dictated by the current underlying national case prevalence, either weekly
or fortnightly.
9
Document 1
10
“High risk” workplaces are defined as dine-in food and beverage establishments, personal care
services, and gym and fitness studios.
During the recent increase in cases in Singapore (first two weeks Sept 2021)
, workplace surveillance
has also been extended to retail mall workers, supermarket staff, delivery personnel (including
parcel and food delivery personnel), and public and private transport workers (taxi drivers, private
hire car drivers and all public transport frontline staff)
RATs for workplaces are administered through “Employer Supervised Self Swabbing” (ESSS). The
government are funding this workplace testing until the end of 2021.
ECEs, Primary schools
3 ART test kits are being given to each child attending ECE/primary school on return from the
September school holidays
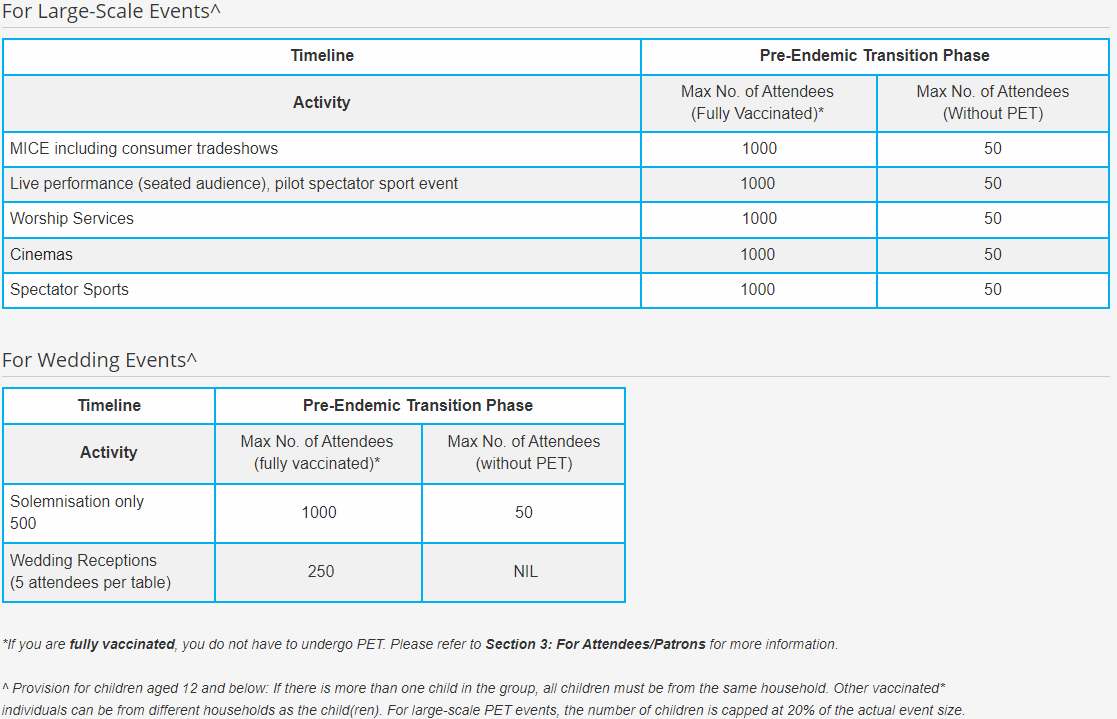
Large Scale Events/Mass Gatherings
Pre- Event Testing (PET) is required, if unvaccinated, and attending a mass gathering. This can be
done by either PCR or RAT at a designated provider site, a maximum of 24 hrs before the event.
PET is not necessary for children or those people who have been fully vaccinated.
See exemplar table below for event types:
under the Offical Information Act 1982
Released
Hospitals
RATs are being used to screen hospital visitors. See the Singapore General Hospit
al guideline as an
example. The surveillance programmes may vary between hospitals.
10
Document 1
11
With regards to hospital staff and patients, surveillance is mostly PCR based, but may include RATs,
and may be supplemented with regular RAT tests during times of increased prevalence. (see
Singapore Hospital guidelines for staff and patients in Appendix A)
Authorised RAT kits
A list of test kits authorised by MOH Singapore can be foun
d here
FAQs
FAQs on RATs/ARTs is available on the Singapore MOH website
Contact Tracing
From 18th September, people who receive a health risk warning or a health risk alert via SMS will be
given instructions by the Singapore MOH on a testing regime that they must follow. This varies
according to the “risk” of the potential contact.
A
health risk warning is issued to those who, based on contact tracing data, have been in close
proximity with a Covid-19 case for an extended period, or are identified as a close contact of a case.
These patients should attend a SASH (Swab And Send Home) clinic for
PCR testing. PCR is done
within 48 hrs of the notification and then at Day 8 from exposure
A
health risk alert is sent to people who, based on contact tracing data, are considered to be at a
lower risk of infection compared with those issued with a health risk warning. These patients require
RAT testing and they can collect the necessary kits (three in total) from pre-designated “vending
machines” free of charge using their identification cards. The RAT tests are performed at Days 3, 5 &
7 from exposure.
People are required by law to comply with these testing and isolation requirements.
https://www.moh.gov.sg/covid-19/general/faqs---border-measures-and-changes-to-stay-home-
notice-requirements/faqs---surgical-health-alerts
Table. Vaccination, cases and tests in Singapore
Singapore
under the Offical Information Act 1982
Population:
5,690,000
Fully vaccinated:
4,455,536
(78% total population)
Cases: (7 day average)
1178
Antigen tests/day (7 day average)
Not publically available
PCR tests/day (7 day average)
67,100
Total tests/day
67,100
(1.2% popn/day)
Released
Tests per case=
56.9
11
