Worksheet for AEFI causality assessment
Mar 2019
Patient ID/ Name :
DoB/ Age:
Sex: Male/ Female
Step 1 (Eligibility)
Name one of the vaccines administered before
this event
What is the Valid Diagnosis?
Does the diagnosis meet a case definition?
Create your quest
ion on causality here
Has the ____________ vaccine / vaccination caused ________________________________(The event for review in step 2 - valid diagnosis)
Is this case eligible for causality assessment? Yes/ No; If, “Yes”, proceed to step 2
Step 2 (Event Checklist) ✓
(check) all boxes that apply
I. Is there strong evidence for other causes? Y N UK NA Remarks
1. In this patient, does the medical history , clinical ex amination and/ or inv estigations,
confirm
another cause for the ev ent?
II. Is there a known causal association with the vaccine or vaccination?
Vaccine product
1. Is there ev idence in published peer rev iew ed literature that this v accine may cause such an
ev ent if administered correctly ?
2. Is there a biological plausibility that this v accine could cause such an ev ent?
3. In this patient, did a specific test demonstrate the causal role of the v accine ?
Vaccine quality
4. Could the v accine giv en to this patient hav e a quality defect or is substandard or falsified?
Immunization error
5. In this patient, w as there an error in prescribing or non-adherence to recommendations for
use of the v accine (e.g. use bey ond the ex piry date, w rong recipient etc.)?
6. In this patient, w as the v accine (or diluent) administered in an unsterile manner?
7. In this patient, w as the v accine's phy sical condition (e.g. colour, turbidity , presence of foreign
substances etc.) abnormal w hen administered?
8. When this patient w as v accinated, w as there an error in v accine constitution/preparation by
the v accinator (e.g. w rong product, w rong diluent, improper mix ing, improper sy ringe filling etc.)?
9. In this patient, w as there an error in v accine handling (e.g. a break in the cold chain during
transport, storage and/or immunization session etc)?
10. In this patient, w as the v accine administered incorrectly (e.g. w rong dose, site or route of
administration; w rong needle size etc.)?
Immunization anxiety (Immunization stress related response - ISRR) 11. In this patient, could this ev ent be a stress response related to immunization (e.g. acute
stress response, v asov agal reaction, hy perv entilation, dissociativ e neurological sy mptom
reaction etc)?
II (time): Was the event in section II within the time window of increased risk (i.e. ‘Yes” response to questions from II 1 to II 11 above)
12. In this patient, did the ev ent occur w ithin a plausible time w indow after v accine
administration?
III. Is there strong evidence against a causal association?
1. Is there a body of published ev idence (sy stematic rev iew s, GACVS rev iews, Cochrane
rev iew s etc.)
against a causal association betw een the v accine and the ev ent?
IV. Other qualifying factors for classification
1. In this patient, did such an ev ent occur in the past after administration of a similar v accine?
2. In this patient did such an ev ent occur in the past independent of v accination?
3. Could the current ev ent hav e occurred in this patient w ithout v accination (background rate)?
4. Did this patient hav e an illness, pre-ex isting condition or risk factor that could hav e contributed
to the ev ent?
5. Was this patient taking any medication prior to the v accination?
6. Was this patient ex posed to a potential factor (other than v accine) prior to the ev ent (e.g.
allergen, drug, herbal product etc.)?
Y: Yes N: No UK: Unknown NA: Not applicable or Not available
1
 Mar 2019
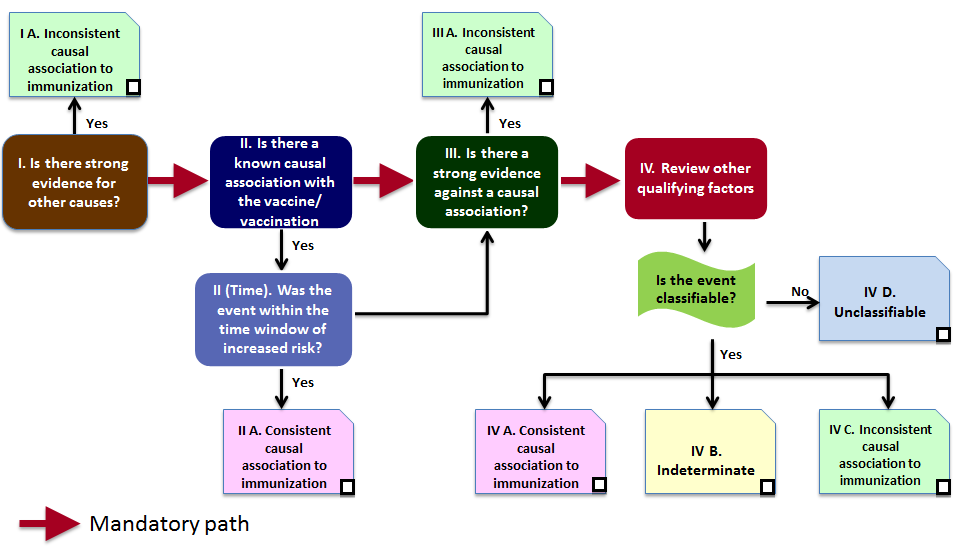
Step 3 (Algorithm) review all steps and
Mar 2019
Step 3 (Algorithm) review all steps and ✓
all the appropriate boxes
Notes for Step 3:
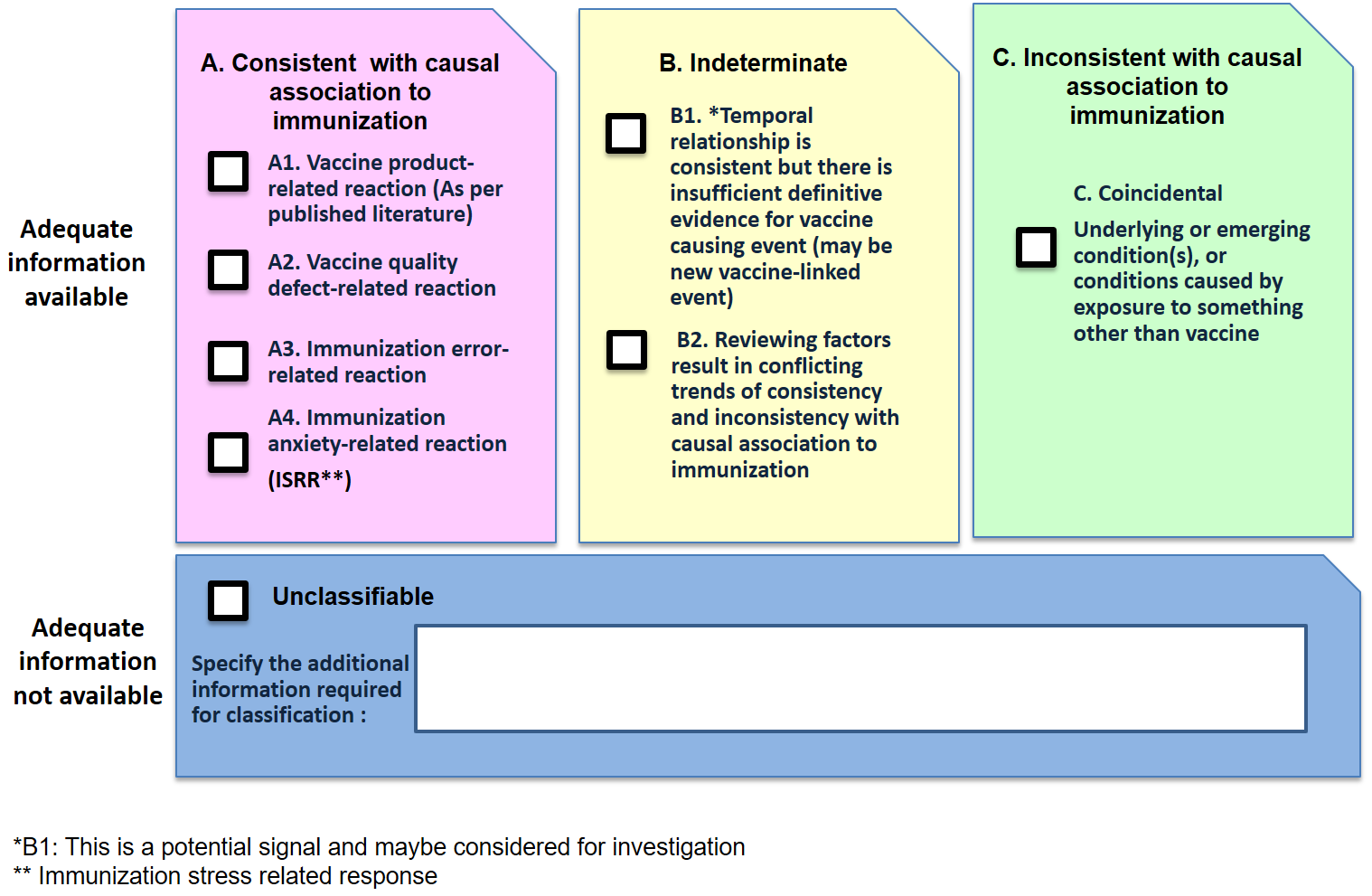
Step 4 (Classification) ✓
all boxes that apply
Summarize the classification logic in the order of priority:
With available evidence, we could conclude that the classification is _____________________________because:
With available evidence, we could
NOT classify the case because:______________________________________
2

