 CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
ting
Released
under
Smokefree Environments and
Regulated Pr
the oducts (Vaping)
Amendment
Official Bill
Departmental Report
Information
Prepared by the Ministry of Health
Act
May 2020
1982
Released
under
the Official
Information
Act 1982
link to page 7 link to page 7 link to page 8 link to page 8 link to page 8 link to page 9 link to page 9 link to page 9 link to page 9 link to page 9 link to page 9 link to page 10 link to page 10 link to page 10 link to page 10 link to page 11 link to page 11 link to page 12 link to page 14 link to page 14 link to page 14 link to page 17 link to page 17 link to page 18 link to page 18 link to page 19 link to page 19 link to page 21 link to page 21 link to page 22 link to page 25 link to page 25 link to page 34 link to page 34
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Contents
Introduction ............................................................................................................................... 1
About the Bil ............................................................................................................................. 1
Released
Section 1: Summary of submissions .......................................................................................... 2
High-interest topics ............................................................................................................................. 2
Flavours ........................................................................................................................................... 2
Promotion and advertising ............................................................................................................. 3
Sales to under 18-year-olds ............................................................................................................ 3
Vape-free workplaces, early childhood centres and schools ......................................................... 3
under
Standardised packaging .................................................................................................................. 3
Nicotine levels ................................................................................................................................. 3
Evidence on vaping ......................................................................................................................... 3
The diversity of perspectives presented ............................................................................................. 4
the
Māori health sector organisations .................................................................................................. 4
The wider health sector .................................................................................................................. 4
Official
Vaping consumers ........................................................................................................................... 4
Small retailers ................................................................................................................................. 5
Vape retailers .................................................................................................................................. 5
Section 2: Summary of recommendations ................................................................................ 6
Information
Section 3: Clause-by-clause analysis .......................................................................................... 8
Commencement dates ........................................................................................................................ 8
Clause 2: Commencement .............................................................................................................. 8
Definitions ......................................................................................................................................... 11
Clause 5: Section 2 amended (Interpretation) .............................................................................. 11
Purpose of this Act ............................................................................................................................ 12
Clause 6: New section 3A: Purposes of this Act ............................................................................ 12
Act
Vaping prohibited in legislated smokefree areas ............................................................................. 13
Clauses 9–20: Various amendments prohibiting vaping in legislated smokefree areas .............. 13
1982
Specialist vape retailers .................................................................................................................... 15
Clause 21: New section 14: Vaping in approved specialist vape retail premises ......................... 15
Clause 21: New section 14A: Application for approval as a specialist vape retailer .................... 16
Restrictions on advertising regulated product (and exemptions) .................................................... 19
Clause 26: New sections 23, 24 and 25 ......................................................................................... 19
Restrictions on sponsorship .............................................................................................................. 28
Clause 26: New sections 28, 29 and 30 – Restrictions on sponsorship and related activity ........ 28
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report i
link to page 35 link to page 35 link to page 37 link to page 37 link to page 37 link to page 38 link to page 38 link to page 38 link to page 43 link to page 43 link to page 43 link to page 45 link to page 45 link to page 48 link to page 52 link to page 52 link to page 53 link to page 53 link to page 53 link to page 54 link to page 54 link to page 55 link to page 55 link to page 56 link to page 56 link to page 58 link to page 58 link to page 59 link to page 60 link to page 60 link to page 60 link to page 61 link to page 61 link to page 77 link to page 79 link to page 81
Distribution, inducements and rewards, etc .................................................................................... 29
Clause 26: New sections 32, 33 and 34 – free distribution, discounted products and rewards .. 29
Information and warnings at point-of-sale and on Internet ............................................................. 31
Clause 26: New sections 37 and 38 – Internet and point-of-sale health information or warnings
Released
...................................................................................................................................................... 31
Prohibition on sale and distribution to people under 18 years of age ............................................. 32
Clause 26: New sections 39, 40 and 44: Sale, distribution and supply to people younger than 18
years of age prohibited (and repeat offenders) ........................................................................... 32
Products label ed for chewing or other oral use .............................................................................. 37
Clause 26: New section 53: Regulated products cannot be advertised or labelled, etc, as suitable
for chewing ................................................................................................................................... 37
under
Obligations on retailers, including notifications and flavour restrictions ......................................... 39
Clause 26: New section 63 ............................................................................................................ 39
Powers of the Director-General of Health ........................................................................................ 42
the
Prohibited ingredients and colouring substances ............................................................................ 46
Clause 26: New section 66: Substances that notifiable products must not contain .................... 46
Establishment of notification database ............................................................................................ 47
Official
Clause 26: New section 73: Establishment of database and confidentiality of certain information
...................................................................................................................................................... 47
Regulations........................................................................................................................................ 48
Clause 26: New sections 75–80 .................................................................................................... 48
Information
Infringement offences ....................................................................................................................... 49
Clause 26: New sections 81–84 .................................................................................................... 49
Enforcement officers ........................................................................................................................ 50
Clause 26: New sections 85–93 .................................................................................................... 50
Annual returns and reports ............................................................................................................... 52
Clause 26: New section 94 ............................................................................................................ 52
Section 4: Minor and technical changes .................................................................................. 53
Act
Section 5: Out-of-scope comments noted in submissions ...................................................... 54
Comments on tobacco and tobacco products .................................................................................. 54
1982
Vaping products should be regulated under the Medicines Act ...................................................... 54
Appendices ............................................................................................................................... 55
Appendix 1:
Submitters’ details ......................................................................................... 55
Appendix 2:
The Ministry’s views on the risks and benefits of vaping products ............... 71
Appendix 3:
Youth vaping (14- to 24-year-olds) ................................................................ 73
ASH Year 10 Snapshot survey ....................................................................................................... 75
ii Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
link to page 83 link to page 84 link to page 85 link to page 85
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Youth Insights Survey .................................................................................................................... 77
Health and Lifestyle Survey ........................................................................................................... 78
New Zealand Health Survey .......................................................................................................... 79
Youth 19 Rangatahi Smart Survey................................................................................................. 79
Released
under
the Official
Information
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report iii
Released
under
the Official
Information
Act 1982
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Introduction
The Smokefree Environments and Regulated Products (Vaping) Amendment Bil (the Bil ) was
introduced on 24 February 2020 and completed its first reading on 11 March 2020, at which time, it
was referred to the Health Select Committee (the Committee) for consideration. Public submissions
Released
on the Bil closed on 1 April 2020.
This report summarises the submissions received by the Committee and recommends whether the
Bil should be amended accordingly.
Our recommendations on amendments to the Bil are subject to the Parliamentary Counsel’s
discretion concerning how best to express each recommendation in legislation. In addition, the
Parliamentary Counsel may recommend further amendments to the Bill that are:
under
• a consequence of implementing a recommendation made by the Ministry of Health (the
Ministry)
• necessary for the overall coherence of the legislation
• required editorial changes (eg, punctuation, spelling and typographical errors).
the
About the Bil
Official
The Bil amends the Smoke-free Environments Act 1990 (the Act) (which currently applies to tobacco
smoking products and herbal smoking products) to include provision for regulating smokeless tobacco
devices and vaping products. Each of these products is covered under the new term ‘regulated
product’, which the Bil introduces to incorporate the broader scope of products regulated under the
Act.
Information
The Bil aims to take a balanced approach to the regulation of vaping and smokeless tobacco products.
The Bill reflects concerns regarding children’s and young people’s access to and use of vaping and
smokeless tobacco products and aims to protect these groups from any risks associated with such
products. The Bil does this by extending many of the existing provisions of the Act to vaping products
and heated tobacco devices, including the prohibitions on promoting and advertising smoking and
tobacco products and smoking in indoor workplaces, early childhood centres and schools.
At the same time, the Bil acknowledges that vaping products are much less harmful than smoking and
that they may help some people to quit smoking. In doing so, the Bil provides exemptions for vaping
products (and to a lesser extent smokeless tobacco products) to some of the Act’s promotion and
Act
advertising restrictions to support smokers’ access to these products and to information and advice
that may help smokers switch successfully from smoking to vaping.
1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 1
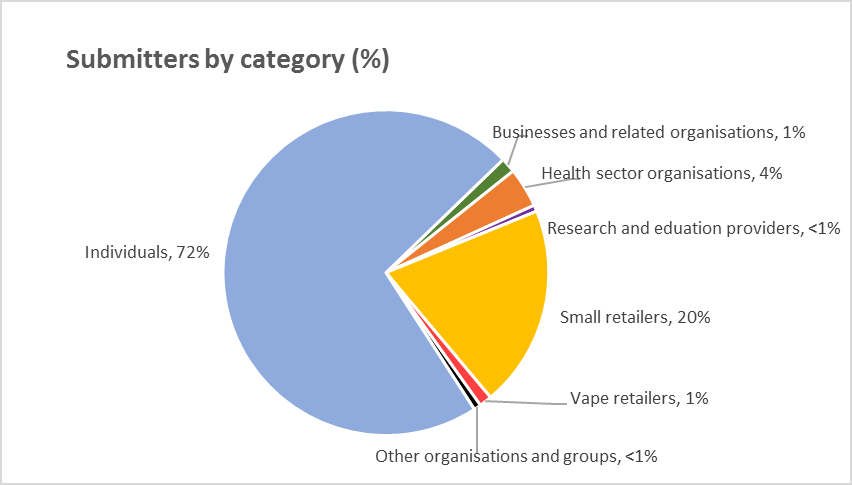
Section 1: Summary of submissions
Over 1,200 submissions were made by a wide range of submitters
The Committee received a total of 1,271 written submissions on the Bill and heard 84 oral
submissions. The list of submitters is attached as Appendix 1.
Released
The largest group of submitters was individuals (914 written submissions), with over half identifying
as vaping consumers. Many responded using a third-party questionnaire that sought responses to
set questions.
Small general retailers were the next largest submitter group, with 255 written submissions. Most
were from dairies and grocery stores, while some were from liquor stores and service stations.
under
Figure 1: Submitters by category
the Official
Information
High-interest topics
Submitters made comments across most areas of the Bil , but some topics were of high interest, with
Act
at least half of all submitters commenting on these areas. These topics of high interest are discussed
in more detail below.
1982
Flavours
The availability and range of flavours was the topic of most interest to submitters, especially small
retailers who mostly disagreed with the proposed restrictions on the flavours they would be able to
sell.
Individual submitters, especially those identifying as vape consumers, were largely concerned about
the impact that limiting flavours in generic retail stores (eg, dairies, supermarkets and service stations)
would have on smokers wishing to transition or maintain their transition to vaping products.
2 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Promotion and advertising
There was significant feedback on the promotion and advertising proposals in the Bill. Submitters had
a wide range of views about the promotion and advertising proposals, with the majority supporting
some sort of promotion or advertising with varying levels of restriction.
Released
Sales to under 18-year-olds
Most submitters from all categories agreed that the sale of vaping products should not be permitted
to anyone under 18 years of age.
Vape-free workplaces, early childhood centres and schools
under
Over half of the submitters that commented on the provisions prohibiting vaping in legislated
smokefree areas generally agreed with the prohibitions, while about one-third disagreed. Other
submitters supported an approach where vaping would be prohibited in some areas and allowed in
others.
the
Standardised packaging
Official
There was an even split between submitters who agreed that packaging should be standardised and
those who disagreed.
Nicotine levels
Information
Over half of the submitters commented on the setting of maximum nicotine levels in vaping products,
although this is a matter for the regulations, rather than the Bill. Many of those who agreed with
setting maximum levels made specific suggestions. The Ministry wil consult on maximum nicotine
levels during the development of the regulations.
Evidence on vaping
Submitters presented a wide range of evidence to support their submissions. The evidence was often
conflicting, supporting arguments for both a tighter and a more relaxed regulatory approach than that
Act
set out in the Bill.
Appendix 2 outlines the Ministry’s views on the risks and benefits of reduced-harm tobacco and vaping
1982
products.
Appendix 3 outlines the data on youth vaping in New Zealand (ie, in the 14- to 24-year-old age group).
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 3
link to page 10
The diversity of perspectives presented
The Committee received a diverse range of perspectives on the Bil from different groups of
submitters, including Māori health sector organisations, the wider health sector, vaping consumers,
small retailers and vape retailers.
Released
Māori health sector organisations
Māori health sector organisations welcomed the Bil and the regulation of vaping products. All
shared concerns regarding tamariki and rangatahi being exposed to and taking up vaping, and all
supported legislation that would discourage this. Just over half of these submitters advocated for
more stringent measures to achieve this, raising concerns that vaping is contributing to the inter-
generational impacts of nicotine dependence on whānau, hapū and iwi.
under
Many of these submitters commented on the colonial context of tobacco; its introduction into New
Zealand, and the impact on Māori, who were tobacco-free before colonisation. Submitters highlighted
the disproportionately high rates of smoking and burden of tobacco-related disease and mortality
among Māori.
the
Some submitters referenced the 2010 Māori Affairs Select Committee’s
Inquiry into the Tobacco
Industry in Aotearoa and the Consequences of Tobacco Use for Māori1 and the recommendations
made in that report, along with the Government’s commitment to achieving the Smokefree Aotearoa
Official
2025 goal.
The wider health sector
Health sector organisations, more generally, expressed a similar range of perspectives to the Māori
Information
health sector, with some seeking a more liberal regulatory approach, while others wanted tighter
restrictions.
An area of difference for the health sector, amongst those with more conservative views, related to
being more restrictive at retail. Suggestions included not allowing retailers such as diaries to sell
devices; limiting sales of all regulated products to specialist vape stores, pharmacies, and distribution
via publicly-funded stop-smoking services; and implementing a licensing system for all retailers of all
regulated products.
Act
Vaping consumers
Many submitters who identified as vapers shared their personal stories on the positive impact that
1982
switching from tobacco smoking to vaping had had, and continues to have, on their health, their
finances and other aspects of their lives.
1 New Zealand Parliament. 2010.
Inquiry into the Tobacco Industry in Aotearoa and the Consequences of
Tobacco Use for Māori. Report of the Māori Affairs Committee. Wellington: House of Representatives. URL:
www.parliament.nz/resource/en-NZ/49DBSCH_SCR4900_1/2fc4d36b0fbdfed73f3b4694e084a5935cf967bb
(accessed 10 May 2020).
4 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Smal retailers
Many small retailers expressed concerns that the restrictions on flavours would damage their
businesses. Many of these submitters said that they had helped smoking customers switch to vaping
by supplying a range of flavours. They also expressed concerns that they would be prohibited from
communicating with their customers.
Released
Vape retailers
Vape retailers welcomed regulation, and many pointed out that they had been self-regulating for
several years in the absence of legislation.
Many shared small retailers’ concerns that the range of flavours would be limited in generic stores.
under
There was a general concern about the costs associated with the regulatory requirements, the lack
of detail on the regulations and the short commencement timeframes.
A number of these submitters provided detailed suggestions that wil be useful in developing the
product safety regulations.
the Official
Information
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 5
Section 2: Summary of recommendations
Table 1: Summary of recommended changes to the Bil
Topic
Clause
Recommendations
Commencement
Clause 2
We recommend that:
Released
• the notification regime comes into effect 12 months after Royal assent,
through amending commencement and transitional provisions
• the Bill be amended to give schools and early childhood education and care
centres a six-month lead-in time to change their notices from prohibiting
smoking to prohibiting smoking and vaping.
Interpretation
Clause 5
We recommend that the Bill be amended to exclude the use of heated tobacco
products in specialist vape stores, as was the policy intent.
Purpose
Clause 6,
We recommend that the Bill be amended to include an additional purpose
new section along the lines of: ‘to support smokers to switch to significantly less harmful
under
3A
alternatives’.
Application for
Clause 26,
We recommend that the Bill be amended so that existing vape stores that have
approval as
new section more than 50 percent of their sales from vaping products can trade as such
specialist vape
14A
under new section 14A for a transitional period of 12 months.
retailer
the
Specified
Clause 26,
We recommend that the Bill be amended to:
publications
new section • amend new section 24(1)(g) along the fol owing lines:
exempt from
24
(i) the display, in accordance with regulations, of vaping products within
Official
advertising
any retail premises specified in regulations or on any Internet site
prohibition
specified in regulations; and
(ii) the provision, in accordance with regulations, of information (in any
medium) relating to vaping products within those premises or on that
Internet site
• add regulation-making powers for new section 24(1)(g)(i) and (ii), as was
intended
Information
• delete new section 24(1)(h) (advice and recommendations that specialist
vape retailers can give to customers)
• provide for the following additional exemptions in new section 24:
o
publication, dissemination and discussion related to research into
vaping and smokeless tobacco products, or ways of encouraging
smokers to switch to reduced-harm products
o
publication of non-sponsored media articles/blogs etc
encouraging the use of reduced-harm products
o
manufacturers and importers to provide retailers with
information about the use of vaping and smokeless tobacco
products, in accordance with regulations (with a corresponding
Act
regulation-making power)
o
specialist vape retailers to communicate with their existing
customers about vaping products, in accordance with regulations
(with a corresponding regulation-making power)
1982
• exempt communications in specified circumstances described in
regulations in relation to vaping and smokeless tobacco products (with a
corresponding regulation-making power)
• amend new section 24(1)(f) to limit the application of the exemption to
publicly funded healthcare services only and delete the word ‘approved’
and replace it with ‘issued’
• clarify new section 24(1)(i) to ensure that advice can also be given to
groups of individuals by qualified health professionals.
6 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Topic
Clause
Recommendations
Oral nicotine
Clause 26,
We recommend that:
products
new section • the Bil be amended to regulate oral nicotine products (other than products
53
that have received consent for distribution under section 20, or provisional
consent under section 23, of the Medicines Act 1981) under clause 53 of
the Bill
Released
• a consequential amendment to the Medicines Regulations be made to
clarify that all non-oral nicotine-containing products are medicines.
Powers of the
Clause 26,
We recommend that the Bill be amended to:
Director-General
new sections • empower the Director-General of Health to set up technical advisory
of Health
67–72
committees to assist in decision-making and exercise of powers under the
Bil once enacted
• provide the notifier a reasonable opportunity to be heard before the
Director-General of Health can suspend or cancel a product notification of
a notifiable product
under • provide a notifier with a right of appeal against a decision to suspend or
cancel a product notification of a notifiable product similar to the appeal
right in section 11 of the Psychoactive Substances Act 2013 (ie, a right of
appeal to an appeals committee of three members appointed by the
Minister of Health, and a further right of appeal on questions of law to the
High Court)
the
• require the Director-General of Health to be satisfied on reasonable
grounds that the product presents an unacceptable safety risk before
making a recall order.
Official
Prohibited
Clause 26,
We recommend that new section 67 be amended to enable the Director-
ingredients
new section General of Health to set maximum limits for ingredients, in addition to outright
66
prohibiting ingredients.
Minor or
Various
We recommend the following minor and technical changes be made to the Bill.
technical
• The fol owing definitions in section 2(1) and (2) of the current Act should
Information
be amended to include al regulated products (they currently just apply to
tobacco):
o
Automatic vending machine
o
Distributor
o
‘Of the same kind’.
• The Bill should be amended to ensure that a vaping substance does not
include medicinal cannabis or a cannabidiol (CBD) product (as these are
regulated under the Medicines Act 1981 and the Misuse of Drugs Act
1975).
• The Bill should be amended to enable the current regulations to continue
to apply to tobacco products and, where applicable, herbal smoking
Act
products, until a new set of regulations applying to regulated products
comes into force, as was the policy intent.
1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 7
Section 3: Clause-by-clause analysis
Commencement dates
Clause 2: Commencement
Released
This clause provides that the Bill comes into force on the day after it receives Royal assent, with a small
number of exceptions.
• New section 36 (which restricts the visibility of regulated products) comes into force one month
after the date the Act receives Royal assent.
• The following provisions come into force six months after the date the Act receives Royal assent.
- New sections 59–62, which require a manufacturer or importer of a vaping product or
smokeless tobacco product to notify the product before sale in New Zealand
under
- New section 63(2), which links to new schedule 2 and restricts the flavours that may be
contained in vaping products sold by generic retailers
- New section 73, which requires the Director-General of Health (the Director-General) to
establish a database for the purpose of the new Part 4.
the
Submissions
General comments
Official
Several submissions mentioned that, in the wake of the COVID-19 response, the Committee process
is being rushed and there needs to be ful stakeholder inclusion and consultation. These submitters
recommended that the commencement date for the provisions that come into force six months after
Royal assent be changed to 12 months. One submitter suggested 12–18 months from Royal assent
would be needed to al ow for consultation on the regulations.
Information
Some submitters, however, asked the Committee and officials to ensure that the Bil moves as fast as
possible through the House (and by implication to commencement).
Notification requirements
Some submitters commented that, while they supported regulating vaping products, the
commencement period of six months for new sections 59–60 (which require a manufacturer to notify
the product before sale in New Zealand) is too short, and this period should be changed to 12 months.
In particular, these submitters were concerned that they had not yet seen any regulations specifying
Act
the notification requirements, including the product standards and fees. They made the point that the
legislation will have a significant regulatory impact, particularly on small businesses, and that industry
needs more time for implementation and to ensure compliance.
1982
Standardised packaging
Submitters were concerned that the period provided for meeting packaging requirements wil not be
adequate as it will not allow enough time for supply chains, manufacturers and retailers to change
label descriptions and packaging.
Submitters commented that there is no line of sight as to what will be in the regulations.
8 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Comment
We agree that some aspects of the Bil should come into force later than is currently provided for in
the Bill. We recommend delaying the date at which compliance is required, as set out below. This will
ensure that provisions associated with some of the key purposes of the Bil , such as preventing the
normalisation of vaping and minimising harm to young people, are in effect as soon as possible, while
Released
still allowing time for full consultation on the regulatory proposals and for industry implementation.
Notification
We recommend the notification regime come into effect 12 months after Royal assent, by amending
commencement and transitional provisions.
under
Standardised packaging
We consider that new sections 49 and 50 (standardised packaging, including an offence provision)
should continue to come into force on the day after Royal assent because, in practical terms, this
would have no effect until regulations are in force. Transitional times would be built into the
regulations to ensure time for industry implementation, as was the case when standardised packaging
the
for tobacco products was implemented. We intend to consult on appropriate commencement
timeframes for the packaging regulations when we consult on the substantive requirements.
Official
Existing standardised packaging regulations would continue for tobacco products during the
transitional period (but these do not apply to vaping products or heated tobacco devices).
Notices at schools and early childhood education and care centres
The Bil requires managers of schools and early childhood education and care centres to take al
Information
reasonably practicable steps to ensure that a notice stating that vaping is forbidden is prominently
displayed at or immediately inside every entrance to the premises and every outer entrance to every
building or enclosed area forming part of the premises.
At present, schools and early childhood education and care centres have notices saying that smoking
is forbidden (section 7A of the current Act). Schools and early childhood care centres will need to
change these notices to include vaping, but there is currently no lead-in time to al ow them to do this.
The Ministry will work with the Health Promotion Agency (HPA) to support schools and early childhood
education and care centres to replace their notices, at no cost to schools. However, given the COVID-
Act
19 response implications for education providers, we recommend that schools and early childhood
education and care centres be given a six-month lead-in time to change their notices from prohibiting
smoking to prohibiting smoking and vaping (notice requirements relating to smoking wil remain in
1982
force in the interim).
Recommendations
We recommend that:
• the notification regime comes into effect 12 months after Royal assent, through amending
commencement and transitional provisions
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 9
• the Bill be amended to give schools and early childhood education and care centres a six-month
lead-in time to change their notices from prohibiting smoking to prohibiting smoking and
vaping.
Released
under
the Official
Information
Act 1982
10 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Definitions
Clause 5: Section 2 amended (Interpretation)
This clause amends the interpretation section to insert new terms, enable current terms to apply to
regulated products and replace the definition of open area.
Released
Submissions
Fewer than 10 submitters commented directly on this clause. A few of these submitters suggested
that the definition of ‘vaping’ should not include the use of heated tobacco products, although their
rationales differed, for example:
• ‘nicotine vaping products intended to support quitting should be clearly differentiated from
under
tobacco vaping products’
• ‘this may allow the use of heated tobacco products in specialist vape stores (clause 9, new section
14(1) refers)’.
A smal number of submitters considered that the definition of ‘regulated product’ was not broad
the
enough and should be extended to include tobacco product accessories (eg, cigarette papers) or
nicotine products that do not contain tobacco leaf (eg, oral nicotine pouches and drops) as it is difficult
to enforce the Act for these products.
Official
A few submitters suggested changes to allow the meaning of ‘regulated product’ to be expanded over
time (eg, by regulations) to future-proof against unforeseen product developments.
Comment
Information
We note that the exemption for vaping in approved premises of specialist vape retailers was intended
to apply only to vaping using a vaping device. It was not intended to apply to vaping through use of
heated tobacco products. We recommend correcting this oversight.
The Ministry does not agree that the Bil should enable the definition of a regulated product to be
expanded by regulations. This definition is central to the regulatory framework and properly belongs
in the primary statute.
We do not consider it necessary to expand the scope of the definition of ‘tobacco product’ to cover
al accessories. We acknowledge that there has been some uncertainty about what constitutes a
Act
tobacco product advertisement, with conflicting court decisions. This has, however, been addressed
in the Bill by amending the offence provision for advertising (new section 23) to prohibit publishing a
‘regulated product advertisement’ rather than the current section 36, which prohibits publishing ‘any
1982
advertisement for a tobacco product’.
Recommendation
We recommend that the Bil be amended to exclude the use of heated tobacco products in specialist
vape stores, as was the policy intent.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 11
Purpose of this Act
Clause 6: New section 3A: Purposes of this Act
This clause amends the purposes of the Act to include:
Released
• prevention of the normalisation of vaping
• regulation of the safety of vaping products and smokeless tobacco products.
Submissions
Five submitters commented explicitly on this clause. Of these, two expressed general support for the
changes to the purposes of the Act. One submitter recommended that the Act’s statement of purpose
acknowledge that vaping and smokeless tobacco products are less harmful than smoking and that they
under
may help people to quit smoking.
Two submitters did not support the inclusion of the purpose ‘to prevent the normalisation of vaping’,
as they thought this was inappropriate given the public health interest in shifting people from smoking
to vaping. One of the submitters described how de-normalisation strategies have contributed to
the
stigmatisation of the smoker, contributing to feelings of shame and isolation from support services,
and was concerned that this would happen with vaping.
Alternative suggestions for the purposes were:
Official
• to regulate vaping, tobacco heating devices and al non-combustible oral nicotine and tobacco
products not covered by the Medicines Act 1981 to provide consumers with confidence that the
products have been manufactured in accordance with minimum quality standards
• to restrict the advertising and sale of al products covered by the Act to people aged 18 years and
over.
Information
Comment
The Bil seeks to strike a balance between preventing the uptake of vaping among young people and
supporting smokers to switch to a significantly less harmful product. The Ministry considers that these
complementary purposes both need to be expressed in the Bill.
We do agree, however, that the Bill lacks any expression in its purpose statements of its support for
smokers to switch to significantly less harmful alternatives. We recommend that an additional purpose
Act
be added to the Bill to this effect.
1982
Recommendation
We recommend that the Bil be amended to include an additional purpose along the lines of: ‘to
support smokers to switch to significantly less harmful alternatives’.
12 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Vaping prohibited in legislated smokefree areas
Clauses 9–20: Various amendments prohibiting vaping in legislated
smokefree areas
Released
Clauses 9–20 extend the existing restrictions on smoking in legislated smokefree areas to include
vaping and the use of heated tobacco products.
Submissions
Over 800 submitters commented on vaping in legislated smokefree areas. Over half of these
submitters generally agreed with the proposed prohibitions, and about one-third disagreed. The
remaining submitters supported an approach where vaping would be prohibited in some areas, such
under
as where children congregate, and allowed in other areas, such as R18 venues.
Reasons for supporting the prohibition on vaping in smokefree areas included:
• reducing the likelihood of vaping being normalised
the
• signalling that second-hand vapour is not harmless
• making enforcement easier
• more general concerns about vaping odour and clouds.
Official
Those who disagreed with the proposed prohibitions said that there is no evidence of harm to justify
a blanket ban and that vaping is not as offensive as smoking.
Reasons given for a mixed approach were that vaping has lower health risks for users and bystanders
compared with smoking and vaping should be allowed in some smokefree areas to encourage smokers
to switch to vaping.
Information
Many submitters supported a prohibition on vaping in areas that children frequent, such as
playgrounds, sportsgrounds, beaches and parks because of the risk of normalising vaping.
Some submitters recommended looser restrictions on the prohibition of vaping in certain
environments. For example, they supported employer or owner discretion in R18 venues and some
workplaces, especially those employing manual labourers. Some submitters supported the United
Kingdom’s approach, which allows owners and managers to determine a suitable policy for their
premise.
Several submitters considered that vaping should be prohibited in the outdoor smokefree areas that
Act
are designated by councils because this would help provide consistency and enable easier
enforcement.
1982
Exemptions al owing vaping in workplace vehicles and dedicated rooms in hospitals, etc.
Around 15 submitters commented on the exemptions that would allow vaping in workplace vehicles
and dedicated rooms within hospitals and residential care institutions. Those who disagreed with
these exemptions argued that vaping in these enclosed spaces increases the risk of exposure to toxins.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 13
A few submitters considered that vaping should be al owed in the broader areas of a hospital or health
care institution (ie, not just in dedicated rooms), where smoking cessation may be encouraged as part
of treatment (eg, inpatient mental health facilities).
Released
Vaping in vehicles carrying children
Several submitters suggested that the Bil be amended to explicitly prohibit vaping in vehicles
carrying children and young people under 18 years of age.
Comment
The primary rationale for prohibiting vaping in legislated smokefree areas is to minimise the risk that
vaping wil become normalised. The Ministry acknowledges that there is no robust evidence of harm
under
to bystanders from second-hand vapour.
The Act does not prohibit smoking in outdoor areas. These decisions are made by territorial
authorities. We consider that it would be disproportionate to prohibit vaping in outdoor areas when
this is not the case for smoking, which is significantly more harmful to users.
the
We do not support prohibiting vaping in work vehicles or hospitals and residential care institutions
under the narrow circumstances prescribed in the Act. It would be disproportionate to al ow smoking,
which is much more harmful, but prohibit vaping in these
Official circumstances.
The Smoke-free Environments (Prohibiting Smoking in Motor Vehicles Carrying Children) Amendment
Bill is currently before the House awaiting its third reading. Depending on the progress of that bil , the
Ministry will work with the Parliamentary Counsel Office to ensure that vaping in vehicles carrying
children is also prohibited.
Information
Recommendation
No change.
Act 1982
14 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Specialist vape retailers
Clause 21: New section 14: Vaping in approved specialist vape retail
premises
Released
New section 14 exempts vaping in approved specialist vape retailer premises from the smokefree
environments restrictions in Part 1 of the Bill. Specialist vape retailers must take all practicable steps
to prevent people under the age of 18 years from entering the premises.
Submissions
Around 15 submitters commented on the provisions that allow customers to vape in specialist vape
retailer premises, with most in support. Some submitters provided further suggestions, including
under
restricting the number of people who can vape at a single time, banning large ‘consumer promotional
events’ and only permitting smokers to vape.
A few submitters considered that this exemption should be extended to specialist tobacco retailers so
that they could encourage their smoking customers to switch to vaping products.
the
Most of these submitters supported the prohibition on those under the age of 18 years entering a
specialist vape store. However, some of these submitters considered it impractical, as parents wishing
to enter a vape store would have to leave their children waiting outside the store. They noted that
Official
such requirements do not exist for retailers selling tobacco products.
Comment
The intent of the exemption is to support smokers to switch to vaping by permitting them to sample
Information
different products and to receive instruction and guidance inside specialist vape retailer premises. We
do not support regulating the number of people who can vape within specialist vape retailer premises
as we consider that this can continue to be managed by vape retailers (this provision continues the
status quo for them).
We do not recommend extending this provision to include specialist tobacco retailers. Specialist vape
retailers are required to take all practicable steps to prevent minors from entering the store, which is
a requirement that does not apply to specialist tobacco retailers.
Act
Recommendation
No change.
1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 15
Clause 21: New section 14A: Application for approval as a specialist
vape retailer
New section 14A provides that the Director-General must not give a person approval to be a
specialist vape retailer unless:
Released
• the retail premise is a fixed permanent structure
• at least 85 percent of the person’s total sales from the retail premises are or wil be from the sale
of vaping products
• any requirements of regulations have been met.
Submissions
A specialist vape premise as a fixed permanent structure
under
A few submitters raised concerns about the need for a fixed permanent structure, noting that:
a. small vaping businesses such as mobile vape stores located in rural and small towns cannot afford
to sell from a fixed permanent structure
b. manufacturers that do not operate from a fixed permanent structure would be unable to provide
the
customers with information about vaping products on their website
c. the requirement is ‘inappropriate’ as it is more stringent than what is applied to smoked tobacco
products, which are more harmful
Official
d. the United Kingdom allows sale of al regulated products online and has not seen a significant
uptake in youth vaping.
Some of these submitters made suggestions for change, including that stores with an online-only
presence should be eligible to become specialist vape stores, with one submitter considering that
having age verification systems in place was the key factor. A few of these submitters recommended
Information
removing the fixed permanent structure requirement.
85 percent of sales from vaping products
Around 15 submitters (mainly vape retailers) commented on the requirement that 85 percent of the
total sales of a specialist vape retailer must be from vaping products. Most of these submitters argued
that this requirement was too stringent and that only a very smal numbers of vape retailers would be
able to reach the threshold because many vape stores sell a range of other products.
Suggested alternatives to the 85 percent threshold included giving the Director-General discretion to
Act
approve specialist vape retailers that do not meet the threshold (eg, based on experience and
expertise), lowering the threshold (eg, to 50 percent), having no threshold and licensing all retailers.
One submitter suggested extending the 85 percent requirement to include al regulated products, not
1982
just vaping products.
Other submitters were concerned that specialist vape stores would be able to sell smoked-tobacco
products alongside vaping and smokeless tobacco products.
16 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Changing the retail model (eg, licensing all retailers of vaping products)
Some submitters (mainly health sector organisations) suggested variations on the specialist vape
store model set out in the Bill. For example:
• all retailers who want to sell vaping products should be required to apply for and be granted a
licence, subject to meeting certain conditions
Released
• vaping products (or at least devices) should not be able to be sold in generic stores such as dairies,
supermarkets and service stations. Instead, they should only be sold in specialist vape stores and
pharmacies and distributed through publicly-funded stop-smoking services
• R18 liquor stores should be able to become specialist vape stores (or at least sell the wider range
of flavours).
Comment
under
A specialist vape premise as a fixed permanent structure
We do not support mobile or temporary stores (eg, sales from vehicles, or pop-up stalls at festivals).
These would be difficult to monitor and ensure compliance with the retailer’s obligations (eg, that
only notified products that meet product safety standards may be sold).
Online-only stores could be managed from a compliance perspective, although it may be more difficult
the
to prevent minors from purchasing products from such stores. Some online vape retailers, however,
said that they do have age verification systems in place (or could put it in place) for online sales.
Official
85 percent of sales from vaping products
The Bill provides exemptions from the general restrictions in the Bill for specialist vape retailers, such
as the prohibition on advertising, the ability to offer free or discounted products and the ability to
vape in-store. A threshold is necessary to distinguish between specialist vape stores, which would be
Information
eligible for these exemptions, and a generic store, which would not be eligible.
It is unclear how many vape stores would be affected, but at least one large chain would be impacted.
If the Committee considers a change is necessary, then lowering the threshold would be the simplest
way to achieve this change.
We do, however, recommend that the Bill be amended so that existing vape stores that have more
than 50 percent of sales from vaping products can trade as specialist vape shops under new section
14A for a transitional period of 12 months. This would allow more time for existing vape retailers to
adjust their business model if they wanted to become specialist vape retailers.
Act
Changing the retail model (eg, licensing all retailers of vaping products)
1982
We do not support a significant re-design of the retail model set out in the Bill. We consider that
allowing all retailers to sell vaping products (subject to a range of restrictions) and establishing
specialist vape retailers that are eligible for exemptions to the more general restrictions strikes a good
balance between protecting young people from accessing vaping products and providing a supportive
environment for smokers who wish to switch to a less harmful alternative.
Extending eligibility to R18 liquor stores would extend the range of outlets that could sell a wide range
of flavours (which appears to be what lies beneath this suggestion from a smal number of submitters),
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 17
but this would also al ow vaping in-store. Our preference is to keep this exemption narrowly focused
on approved specialist vape retailers as set out in the Bill.
Recommendation
Released
We recommend that the Bill be amended so that existing vape stores that have more than
50 percent of their sales from vaping products can trade as such under new section 14A for a
transitional period of 12 months.
under
the Official
Information
Act 1982
18 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Restrictions on advertising regulated product (and
exemptions)
Clause 26: New sections 23, 24 and 25
Released
New section 23 restricts a person from publishing a regulated product advertisement. This section
needs to be read in conjunction with clause 5, which amends section 2 – interpretation to extend the
definition of a tobacco product advertisement to all regulated products.
A ‘regulated product advertisement’ is defined in clause 5 as: any words, whether written, printed or
spoken and any pictorial representation, etc, that are used to:
• encourage the use of a regulated product
• notify the availability of a regulated product
• promote the sale of a regulated product
under
• promote smoking or vaping behaviour.
New section 24 provides for several exemptions from regulated product advertising restrictions in
new section 23, including:
the
• a public health message approved by the Director-General
• the display of vaping products within a retail premises or Internet site in accordance with
regulations
Official
• the provision of information, in accordance with regulations, relating to vaping products
within retail premises or on an Internet site
• the giving of advice and recommendations by a specialist vape retailer about vaping products
to customers who are inside the retailer’s approved vaping premises
• any advice or message given by a suitably qualified health worker to an individual for the
purpose of supporting the individual to switch from smoking to vaping.
Information
New section 25 provides further exemptions from the advertising restrictions for retailers, vending
machines and internet sellers.
Submissions
Over 800 submitters commented on the advertising proposals in the Bill. Submitters had a wide range
of views about the proposals, with the majority supporting some sort of advertising with varying levels
of restriction.
Act
Some submitters thought that al vaping product advertising should be prohibited due to the potential
harm it may cause or that online advertising should be prohibited completely. Conversely, a few
submitters thought that there should be no advertising restriction at all given the relative harm of
1982
vaping compared with tobacco.
Several submitters made comparisons between advertising restrictions for vaping and other ‘adult’
products and services with recognised harms (eg, tobacco, alcohol and gambling) and suggested that
vaping should be aligned with these types of activities. Other submitters used a similar comparison to
suggest that vaping should have lower restrictions, given the relatively lower level of harm when
compared with smoking and alcohol use.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 19
A number of submitters commented that they liked receiving advertising material from vape stores so
that they knew what products were available and at what cost. Other submitters, including several
large vaping retailers, stated that they should be able to advertise to existing customers, for example,
through an email or social media campaign, about a product or support to quit smoking.
A few submitters said that it is important for independent vaping companies to be able to advertise
Released
their products to communicate their differences from big tobacco companies.
Unacceptable limits on freedom of expression
In his report on the Bill under the New Zealand Bill of Rights Act 1990 (BORA), the Attorney-General
found that the Bill is inconsistent with the rights and freedoms affirmed in the BORA. In particular, the
Attorney General considered that the limitations on freedom of expression contained in the Bill
relating to the broad restrictions on advertising were not proportionate and that these provisions
were not justifiable.
under
Several submitters explicitly supported the Attorney General’s view, while others commented more
broadly that any restriction on advertising for regulated products was an unacceptable limit on the
freedom of expression and that it impinged on the right of consumers to receive that information.
the
General retailers’ ability to give advice and recommendations about vaping products
Many submitters felt that all retailers (ie, not just specialist vaping retailers) should be able to give
Official
advice and recommendations about vaping products within their store, particularly to smokers who
were considering a switch to vaping.
Other submitters felt that the proposed restriction on the ability of general retailers to provide advice
and recommendations about vaping products was a missed opportunity for these retailers to support
New Zealand’s Smokefree Aotearoa 2025 goal. Some submitters thought this would particularly
Information
disadvantage adult smokers in rural areas, who do not have access to specialised vape retailers.
Several submitters said that it was important for all retailers to be able to provide guidance on vaping
products for practical reasons. For example, several submitters commented that vaping was different
to smoking in that cigarettes did not require detailed instructions (ie, you put it in your mouth, light it
and inhale), whereas vaping is a relatively new technology, and there are a multitude of different
devices, delivery methods and nicotine levels to choose from.
Several retailers commented that they regularly provided advice and recommendations to their
customers and in doing so had assisted many smokers who were considering a move from smoking to
Act
vaping. Other submitters said that they received ‘bad’ advice from dairies, etc, but got good advice
from ‘specialist’ vape stores.
Other submitters felt that vapers and generic retailers should be able to give advice and
1982
recommendations because they are more informed about the products and how to use them than
health workers, who appear to have very little knowledge of vaping.
The broad scope of the terms ‘advertising’ and ‘publish’
Some submitters were concerned that the definitions of ‘advertising’ and ‘publish’ in the Bill are too
broad and that this could have the unintended consequence of prohibiting:
20 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
• the publication/discussion of research on harm reduction, with respect to smokers moving
to vaping, including on Facebook and Twitter (one submitter argued that this could amount
to the censoring of research)
• research into ways of encouraging uptake of reduced-harm alternatives to smoking
• expert opinions on the use of non-tobacco regulated products
Released
• recommendations that a person should switch to vaping made by parties not specifically
approved by the Director-General, for example a family member or iwi leader
• vaping product manufacturers and importers advertising their products to retailers
• public vaping expos/events for vaping consumers
• email or online direct marketing campaigns by retailers, particularly to existing customers
• discussion within consumer groups, such as online vaping communities
• smoking cessation advice by qualified health professionals.
under
The submitters recommended that the Committee considers exempting these activities from the
advertising restrictions in the Bill.
Advertising targeted at young people and non-smokers
the
Some submitters felt that al advertising should be prohibited so that young people would not be
enticed into vaping and to ensure inequalities are reduced.
Many submitters were concerned that vaping was being marketed aggressively at young people and
Official
that the advertising ‘glamorised’ vaping, for example, through the use of colourful displays and
social media campaigns. Some submitters wanted ‘lifestyle’ advertising prohibited if it encouraged
young people and non-smokers to vape (for example, the use of descriptive terms for vape juice,
such as ‘unicorn puke’ or confectionary flavours that could appeal to young people).
A number of submitters recommended restrictions on advertising on television at times when children
Information
may be watching, for example, only advertising in the evenings as is the case with alcohol.
Some submitters wanted to ensure that restrictions on marketing of all regulated products cover all
media platforms and activities, including social media, product placement and the use of influencers.
Other submitters suggested that there should be restrictions on the frequency of vaping advertising
to limit non-smokers’ exposure to vaping advertising.
Display of vaping products
Act
A number of submitters commented on the display of regulated products. Submitters were fairly
evenly split about whether vaping products should be visible within and from outside stores.
Some submitters said that they did not support the display of vaping products in retail stores at al ,
1982
while others supported allowing vaping products to be displayed within specialist vape retailers but
not generic stores. A number of submitters argued that product displays (both instore and outside)
are a form of point-of-sale marketing that could attract young people.
Adopting an advertising code for regulated products
Several submitters recommended that the Committee consider adopting the United Kingdom’s
advertising code, which minimises appeal to youth and non-smokers. One submitter recommended
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 21
link to page 28
that the Bill include a code for advertising regulated products similar to the one that is in place for
alcohol advertising in New Zealand, or the Therapeutic and Health Advertising Code.
Advertising that promotes vaping as a healthier alternative and public health campaigns approved by
Released
the Director-General
Some submitters felt that advertising for vaping products should be restricted to advertising that is
directed at smokers only, for example, that the advertisement should only be permitted if it promotes
vaping as a healthier alternative to smoking.
Several submitters expressed support for public health campaigns or key messages that are approved
by the Director-General to be exempted from the advertising restrictions. A few submitters stressed
the importance that approved campaigns must be developed and implemented in such a way that
they reduce inequalities.
under
A minority of those who commented considered that there also needs to be a way of conveying the
risks of vaping and discouraging young people’s uptake.
the
Monitoring, enforcement and penalties
One submitter recommended that penalties for breaching advertising restrictions should be
increased, and several submitters commented that breaches of the provisions need to be
Official
appropriately monitored and enforced.
Comment
The starting point in the Bill (new section 23) is that al forms of regulated product advertising are
Information
prohibited. These restrictions have been carried over from the current Act and extended to all
regulated products, including vaping products and smokeless tobacco devices.
The advertising restrictions in the Bill are intentionally broad to reduce the social approval of smoking,
to ensure that vaping is not normalised and to discourage non-smokers (particularly children and
young people) from vaping and using tobacco products.
While advertising is generally prohibited, the Bill recognises that smokers need support and advice to
successful y move away from smoking, and new section 24 provides exemptions for certain types of
advertising to occur. These exemptions are made in recognition that vaping products are a reduced-
Act
harm alternative to combustible tobacco, while balancing this with protections for young people.
1982
Unacceptable limits on freedom of expression
The Ministry has provided advice to the Committee on the Attorney General’s report and views on
this matter.
2 For the reasons set out in that letter, our view is that the Bill’s advertising restrictions are
a justified limit on the right to freedom of expression. This is on public health grounds, given the highly
addictive nature of the product and the unknown long-term health risks.
2 Supplementary advice letter to the Committee dated 9 April 2020.
22 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
General retailers’ ability to give advice and recommendations about vaping products
We consider that the interaction between two of the exemption provisions in section 24: sections
24(1)(g) and section 24(1)(h) requires clarification. These provisions are set out below for ease of
reference.
‘Section 24 (1)[The restrictions on regulated product advertising in section 23] do not apply
Released to: … (g) the following activities:
(i) the display,
in accordance with any regulations, of vaping products within any
retail premises or on any Internet site; and
(ii) the provision,
in accordance with any regulations, of information (in any medium)
relating to vaping products within those premises or on that Internet site:
[emphasis
added]
under
(h) the giving of advice and recommendations by a specialist vape retailer about vaping
products to customers who are inside the retailer’s approved vaping premises.’
When these two provisions are read together, the exemption in new section 24(1)(h) (which applies
only to specialist vape retailers) is arguably broader than the exemption in new section 24(1)(g)(ii),
which applies to all retailers, including
the specialist vape retailers.
It is intended that the giving of advice and recommendations by a specialist vape retailer about vaping
products should not be unregulated. The Ministry therefore recommends that paragraph (h) be
Official
deleted and paragraph (g) should be relied on instead. We recommend that new section 24(1)(g) be
tightened along the fol owing lines:
‘(g) the following activities:
(i)
the display, in accordance with regulations, of vaping products within any
retail premises specified in regulations or on any Internet site specified in
Information
regulations; and
(ii)
the provision, in accordance with regulations, of information (in any medium)
relating to vaping products within those premises or on that Internet site.’
The intention is that regulations wil prescribe requirements for specialist vaping premises and
different requirements for generic retailers. Given that the corresponding regulation-making power
was not included in the Bill as introduced, we recommend that the Bill be amended to enable
regulations to deal with the above matters.
Act
If the Committee agrees to this proposal, the Ministry will use the feedback from submitters to assist
with developing the regulatory proposals for new section 24(1)(g)(i ). Any regulatory proposals wil be
subject to ful public consultation, with a view to taking policy decisions to Cabinet to amend the
1982
Regulations.
Advertising targeted at young people and non-smokers
The Ministry acknowledges submitters’ concerns about advertising being targeted at young people
and non-smokers, for example, using colourful displays, naming e-juices after confectionery or using
terms that may appeal to young people.
New section 24(1)(g)(i) provides an exemption for the display of vaping products within a retail
premises ‘in accordance with regulations’. As with the exemption for the provision of information by
retailers outlined above, the display exemption was intended to be linked to the prescribing of
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 23
regulations, but this was inadvertently not included in the Bil . We therefore recommend that a
regulatory power is added to the Bill for the exemption for display of vaping products in new section
24(1)(g)(i).
If the Committee agrees to include a regulation-making power for new section 24(1)g)(i), we will
consider submitters’ comments about the display of products in generic retail shops (particularly
Released
their appeal to young people) when we are developing the regulatory proposals for Cabinet’s
consideration.
We do not intend to make any regulations for the display of vaping products in specialist vaping
retail premises as people under the age of 18 years are not permitted to enter those stores.
However, we recommend that the scope of the regulation-making power includes all retailers
(including specialist retailers) to ensure the Bill is future-proofed should issues arise.
under
Adopting an advertising code for regulated products
The Ministry does not agree with the submitters’ recommendation that New Zealand introduce a
vaping product advertising code such as the one in the United Kingdom or New Zealand’s alcohol
advertising code.
the
In our view, a code would add an unnecessary layer of complexity to the advertising restrictions
given that there are only limited exemptions to the broad restriction on advertising. For example,
Official
the Bill does not allow television, radio or bil board advertising; online marketing; endorsements;
product placement or promotion by social media influencers.
The regulation of advertising for vaping products in the United Kingdom (and alcohol advertising in
New Zealand) is more permissive and al ows for some advertising and marketing of those products.
It is therefore appropriate for detailed guidance to be provided in those circumstances.
Information
The Therapeutic and Health Advertising Code does not apply to vaping devices and substances
because clause 5(4) of the Bill explicitly excludes these products from the scope of the Medicines Act
1981.
Exemption for research and genuine media articles
There was no intention to prohibit research or non-sponsored media articles regarding reduced-
harm alternatives to smoking. We therefore recommend that the Bill is amended to provide for the
following additional exemptions:
Act
• publication, dissemination and discussion related to research into vaping and smokeless tobacco
products, or ways of encouraging smokers to switch to reduced-harm products
•
1982
publication of non-sponsored media articles/blogs etc encouraging the use of reduced-harm
products.
Other exemptions proposed by submitters (eg, manufacturers, direct communications with customers
by specialist vape retailers)
The Ministry agrees that there may be some scope to relax the advertising provisions in respect of
some of the other proposals made by submitters, for example, to allow:
24 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
link to page 31
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
• manufacturers and importers to provide retailers with information about the use of vaping and
smokeless tobacco products (eg, how to use a device and its components, nicotine concentration,
safe charging, etc). As submitters pointed out, there is a multitude of different products on the
market and many of these wil require more detailed instructions than traditional tobacco
products where the use is simple and wel known to al smokers
3
Released
• specialist vape retailers to communicate with their existing customers (eg, by email) to promote
new products or discounts, which is something they can do in-store and on their websites.
The Ministry is broadly supportive of the above proposals and recommends that additional
exemptions be added to new section 24 of the Bill to allow for:
• manufacturers and importers to provide retailers with information about the use of vaping and
smokeless tobacco products, in accordance with regulations
• specialist vape retailers to communicate with their existing customers about vaping products, in
under
accordance with regulations.
Any exemptions for these purposes would need to be tightly prescribed to avoid them being used to
circumvent the advertising restrictions in the Bill. We therefore recommend that the Bill allows a
regulation-making power to prescribe limits on these exemptions if needed.
the
If the Committee agrees to this proposal, the Ministry will work with stakeholders to develop
proposals for the regulations to ensure they are consistent with the Bill’s purpose and will be
workable in practice.
Official
Expert opinions, ‘lay person’ advice and recommendations, and online peer support groups
We acknowledge submitter concerns that the advertising restrictions in the Bil may prohibit the
publication of expert opinions on reduced harm products or advice and recommendations made by
friends, family members or iwi leaders that a person should switch to a reduced-harm alternative to
smoking.
Information
We also note submissions from vaping consumers who are concerned that online peer support
groups would not be able to continue due to the advertising restrictions in the Bill.
In the Ministry’s view, if the group is made up of private individuals sharing their experiences and
recommendations about vaping and vaping products, it is unlikely that any enforcement action
would be taken. This is because this type of activity would not meet the public interest test required
for prosecution. However, it was not our intention that the Bill would prevent these types of
communications, and we agree that the matter needs to be addressed.
In our view, these matters require further consideration to ensure any exemption is not used to
Act
circumvent the advertising restrictions in the Bill. We therefore recommend that the Bill be
1982
3 New section 24(1)(a) exempts manufacturer (and retailer) price lists from the advertising restrictions, provided
the information in the list complies with price list regulations and includes any required health messages. This
provision was carried over from the current Act and extended to al regulated products, including vaping
products. The purpose of providing for regulations in this exemption is to prescribe the content al owed in price
lists to prevent them from being used for promotional purposes (eg, by providing information to retailers on
volume incentive schemes and product promotions).
There is also an exemption for manufacturers in new section 24(1)(c), which exempts magazine publications
that are intended for the manufacturer’s employees, but this exemption does not extend to manufacturer’s
communications with retailers.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 25
amended to exempt communications in specified circumstances described in regulations in relation
to vaping and smokeless tobacco products (with a corresponding regulation-making power).
If the Committee agrees to this proposal, the Ministry will work with stakeholders to review whether
the above activities should be exempted from the advertising restrictions in the Bill, with a view to
taking policy decisions to Cabinet to amend the Regulations. The review will take into account
Released
concerns that the potential implications of any exemption does not undermine wider government
objectives in relation to the purposes of the Bill.
Public health messages
Several submitters expressed support for new section 24(1)(f), which exempts public health
messages approved by the Director-General from the advertising restrictions in the Bill.
under
The intent of new section 24(1)(f) was to allow the Director-General to approve public health
messages for use by publicly funded healthcare services, for example, in smoking cessation
campaigns.
As it stands, the exemption for approved public health messages would apply to al people, including
the
retailers. We therefore recommend that the Bill is amended to limit the applicability of new section
24(1)(f) to public health messages by publicly funded healthcare providers only, in line with the
intended purpose of this provision.
Official
The reduced-harm messaging that retailers can use in their communication material was intended to
be prescribed in regulations under new section 24(1)(g)(i ) (if the Committee agrees to include the
regulation-making power). Stakeholders (including retailers) will be consulted on any regulatory
proposals in this regard.
The Ministry also recommends that new section 24(1)(f) is amended to change the word ‘approved’
Information
to ‘issued’ (ie, ‘public health messages
issued by the Director-General’) to clarify that the Director-
General will actively issue the public health messages rather than there being an application process.
Smoking cessation advice by qualified health professionals
The Bill already provides an exemption for suitably qualified health workers to provide advice to an
individual for the purposes of supporting them to switch from smoking to vaping (new section
24(1)(i)). We want to ensure this allows suitably qualified health workers to be able to provide
advice to
groups of individuals as well just individuals and recommend that new section24(1)(i) is
clarified in this respect.
Act
Public vaping expos/events
1982
The Ministry does not support an exemption from the advertising restrictions for public vaping expos
or events because such expos/events have the potential to influence young people and non-smokers
to start vaping, which is inconsistent with the purposes of the Bil .
Monitoring, enforcement and penalties
Breaches of advertising restrictions wil be managed through the Ministry’s monitoring and
enforcement processes, which includes regular compliance checks on retailers.
26 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
The Ministry of Health consulted the Ministry of Justice on the penalty levels in the Bill as is best
practice, including the maximum fines for a breach of advertising restrictions. No changes to the
penalty levels in the Bill are proposed.
Released
Advertising by overseas retailers
We acknowledge the concerns raised by submitters about overseas advertisers targeting domestic
consumers (known as ‘cross-border’ advertising). The Bill regulates this type of advertising if the
person or company has a presence in New Zealand and the target audience includes New Zealanders.
However, it is not possible for New Zealand law to apply in other circumstances.
Recommendations:
under
We recommend that the Bil is amended to:
• amend new section 24(1)(g) along the following lines:
(i)
the display, in accordance with regulations, of vaping products within any
retail premises specified in regulations or on any Internet site specified in
regulations; and
the
(ii)
the provision, in accordance with regulations, of information (in any
medium) relating to vaping products within those premises or on that
Internet site:
Official
• add regulation-making powers for new section 24(1)(g)(i) and (i ), as was intended
• delete new section 24(1)(h) (advice and recommendations that specialist vape retailers can
give to customers)
• provide for the following additional exemptions in new section 24:
o publication, dissemination and discussion related to research into vaping and
smokeless tobacco products, or ways of encouraging smokers to switch to reduced-
Information
harm products
o publication of non-sponsored media articles/blogs etc encouraging the use of
reduced-harm products
o manufacturers and importers to provide retailers with information about the use of
vaping and smokeless tobacco products, in accordance with regulations (with a
corresponding regulation-making power)
o specialist vape retailers to communicate with their existing customers about vaping
products, in accordance with regulations (with a corresponding regulation-making
power)
• exempt communications in specified circumstances described in regulations in relation to
Act
vaping and smokeless tobacco products (with a corresponding regulation-making power)
• amend new section 24(1)(f) to limit the application of the exemption to publicly funded
healthcare services only and delete the word ‘approved’ and replace it with ‘issued’
1982
• clarify new section 24(1)(i) to ensure that advice can also be given to groups of individuals by
qualified health professionals.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 27
Restrictions on sponsorship
Clause 26: New sections 28, 29 and 30 – Restrictions on sponsorship and
related activity
New sections 28, 29 and 30 provide that a manufacturer, importer, distributor or retailer of
Released
regulated products must not sponsor an organised activity or enter into an arrangement involving an
exclusive supply arrangement involving the use of a regulated product trademark, etc.
Submissions
Most submissions that referenced sponsorship were in the format of a questionnaire template that
asked the question ‘Should al advertising and/or sponsoring by vape companies be stopped?’ Most
under
submitters responded either yes or no and did not explicitly state which provision they were
responding to (advertising and/or sponsoring) nor provide a rationale for their answers.
The few submissions that did specifical y comment on sponsorship were general y from health sector
non-governmental organisations (NGOs) who were supportive of a prohibition on sponsorship.
the
Comment
Official
The new sections 28, 29 and 30 carry over the equivalent provisions in the current Act and extend
them to al regulated products, including vaping products. Sponsorship involving al regulated
products is prohibited under the Bil , and submitters that commented specifical y on this provision
were supportive of the provisions. No changes are proposed.
Information
Recommendation
No change.
Act 1982
28 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Distribution, inducements and rewards, etc
Clause 26: New sections 32, 33 and 34 – free distribution, discounted
products and rewards
New section 32 provides that a manufacturer, distributor, importer or retailer of regulated products
Released
must not free of charge or at a reduced price:
• distribute any regulated product (unless they are a specialist vape retailer)
• supply any regulated product to any person for subsequent distribution.
In addition, a retailer must not supply regulated products free or at a reduced charge to another
person for the purpose of that retailer’s business.
New section 33 provides that a manufacturer, distributor, importer or retailer of regulated products
under
must not:
• distribute a regulated product with a non-regulated product
• supply a regulated product with a non-regulated product to another person for later
distribution.
the
In addition, a retailer must not supply regulated products with non-regulated products to another
person for the purpose of that retailer’s business.
Official
New section 34 provides that (except for specialist vape retailers) a person must not offer any gift or
cash rebate or the right to participate in any contest, lottery or game to:
• the purchaser of a regulated product as consideration for the purchase of the product
• any person in consideration for the provision of evidence of the purchase of a regulated
product.
Information
New section 34 also provides that a person (including a specialist vape retailer) must not offer any
gift or cash rebate etc to a retailer as an inducement or reward in relation to:
• the purchase or sale of regulated products by that retailer
• the advertising of regulated products inside that person’s business
• the location of regulated products in a particular part of that retailer’s business.
Submissions
Act
More than 15 submitters commented on the exemptions for specialist vape retailers to provide
rewards involving a discounted product or to provide products discounted or free of charge.
1982
Just over half of these submitters did not support the exemptions. Of these, just over half (mainly
NGOs and district health boards) did not support any exemptions to give discounts, free samples and
loyalty points. Some submitters said that vaping products were already significantly cheaper than
tobacco products and that the provisions encouraged people to keep vaping rather than eventual y
become vape free.
Just under half the submitters supported the exemptions only being offered to those vaping to quit
smoking. Of these, just under half partial y supported the exemption but recommended that the
discounted products and rewards should only be available to those vaping to quit smoking. Some of
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 29
these submitters recommended only smoking cessation services be able to distribute discount
vouchers (etc).
One submitter stated that the free distribution of a regulated product should include an exemption
for smoking cessation services, stating that, under the Bill, it would be illegal for a service to provide
clients with devices, creating barriers to delivering smoking cessation support. One submitter
Released
supported the exemptions being applied more broadly, including to liquor stores, while another
submitter said that generic retail stores being unable to participate in loyalty schemes was ‘anti-
competitive’.
Comment
The intent of these provisions is to allow specialist vape stores, which must be R18, to continue
under
business largely as usual. These exemptions may support smokers to try new products, which may be
more effective for them.
These provisions do not prevent stop smoking services from providing vaping products free of cost.
the
Recommendation
No change.
Official
Information
Act 1982
30 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Information and warnings at point-of-sale and on Internet
Clause 26: New sections 37 and 38 – Internet and point-of-sale health
information or warnings
Released
New sections 37 and 38 contain requirements relating to point-of-sale and Internet-sales health
information or warnings.
Submissions
Around 20 submitters commented on these sections.
Submitters generally supported the provision of appropriate health information and warnings at
under
point-of-sale at both premises and online stores.
One submitter suggested that health information and warnings available at point-of-sale (including
online sales) should make clear that these products are intended to be used for quitting smoking.
Another submitter argued that only minimal marketing should be al owed for vaping products at
the
point-of-sale and that this should include permissible product claims only made available from a set
of pre-approved statements.
Official
Comment
The Ministry considers that there is benefit in having evidenced-based health information and
warnings at point-of-sale, including for Internet sales. This is permitted by the exemption in new
section 24(1)(g)(ii). Details will be set out in regulations that will be publicly consulted on before they
are finalised (if the Committee agrees to include a regulation-making power to correct the drafting
Information
omission).
Recommendation
No change.
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 31
Prohibition on sale and distribution to people under 18 years
of age
Clause 26: New sections 39, 40 and 44: Sale, distribution and supply to
Released
people younger than 18 years of age prohibited (and repeat offenders)
Sale and distribution
New section 39(1) prohibits a person from selling a regulated product to an under-18-year-old. It also
prohibits a person who has sold a regulated product to a person of any age from delivering or
arranging for that product to be delivered to someone under 18 years of age.
New section 39(2) provides that a person who contravenes section 39 commits an offence and is liable
to a fine of up to $10,000 for a body corporate or up to $5,000 for anyone else.
under
New section 39(3) provides a defence to a charge under section 39(2) if the contravention occurred
without the person’s knowledge and they had taken reasonable precautions and exercised due
diligence to prevent the contravention.
New section 39(4) specifically provides that the requirements in new section 39(3) would be satisfied
the
if the person can prove that they sighted an evidence of age document that indicated the purchaser
was 18 years or older.
Official
New section 44 enables the Court to make additional orders against repeat offenders who sell or
distribute to those under 18 years old in certain circumstances. These orders include prohibiting the
person from selling a regulated product for a particular time period or imposing conditions on the sale
of that product.
Information
Supply
New section 40(1) prohibits a person supplying a regulated product to an under 18-year-old in a public
place. New section 40(2) provides that a person who contravenes section 40(1) commits an offence
and is liable to a fine of up to $2,000.
New section 40(3) provides a defence to a charge under section 40(2) if the contravention occurred
without the person’s knowledge and they took reasonable precautions and exercised due diligence to
prevent the contravention. This requirement is satisfied if the person can prove that they sighted an
evidence of age document that indicated the person was 18 years or older (new section 40(4)).
Act
Submissions
1982
Over 800 submitters commented on the restriction on sales to those under the age of 18 years.
An appropriate age limit
Most submitters who commented on this provision thought it was appropriate to have a minimum
age restriction of 18 years for vaping products. Many commented that the age limit should be aligned
with age restrictions for other harmful products, such as alcohol and tobacco.
32 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Other submitters commented that nicotine was very addictive or that the purpose of vaping was to
quit cigarettes, so an age limit of 18 years was therefore appropriate.
A smal number of submitters felt that the age limit should be lower, for example, to align with the
minimum age for marriage (16 years with the permission of the Family Court) or the minimum age
for entering the army (17 years).
Released
A smal number of other submitters thought that the minimum age should be higher, with
recommendations ranging from 20–25 years. One submitter thought that the minimum purchase
age should be increased annually, with a view to New Zealand becoming both vape- and smokefree
in time.
The ease with which young people can access to vaping products
under
Many submitters were concerned that young people are currently able to access vaping products, but
there were conflicting views about where they were getting these products.
Some submitters thought that young people were buying their products local y from smal retailers or
online or that they were being given it by friends or relatives. However, many small retailers and
the
several online retailers said they always require identification and/or that they never sell to young
people.
Official
Strict age-verification requirements and technology use
A number of submitters thought that the Bil needed to have a stronger regulatory framework to
prevent young people from purchasing vaping products online.
Many submitters thought that there should be a requirement for age verification at the point of
purchase and/or delivery. Some submitters recommended that online vape retailers be required to
Information
use 18 plus courier delivery for vape products, which requires the courier to sight evidence of age
when delivering a product to a customer.
A few submitters thought there should not be any online sale of vaping products.
Several submitters commented that many online sites only require a person to click a box saying that
they are over 18 years of age but do not require any additional verification. Another submitter
commented that very few online retailers required evidence of identification on delivery. However,
several online vaping retailers commented that they have already implemented age verification
processes for online purchases, and many smal retailers commented that they always check age
Act
identification.
A number of submitters recommended that the Committee consider requiring technology-based
1982
online age verification, for example government identification schemes such as RealMe®, requiring
purchasers to use an age-verified PayPal account or using a third-party company authorised by the
Department of Internal Affairs.
Some submitters thought that online purchasers should have to provide government-issued age
identification before they can view products on a website (sometimes referred to as an ‘age gate’).
Another submitter commented such requirements as age gating were unrealistic and
disproportionate. The submitter also commented that no other consumer product is required to
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 33
provide proof of age just to view that product’s website and that equivalent legislative requirements
for the alcohol industry (which do not require ‘age gate’ verification) have been successful.
One submitter said that there should be a limit on the number of products a customer can purchase
online to mitigate the risk of an adult of legal age purchasing high quantities of product for
distribution to minors.
Released
One submitter referred to a ‘track and trace’ initiative in the United States whereby devices
confiscated from minors can be tracked using serial numbers to flag ‘bad actors’ in the supply chain.
Overseas retailers
A number of submitters were concerned that overseas online retailers that do not require age
verification will actively market and sell to young people in New Zealand. The submitter warned that,
under
while the government has the power to regulate domestic retailers, it has very little ability to control
the actions of sites based overseas.
Minors’ access to vaping products
the
Many submitters thought that minors who smoke should have control ed access to vaping and
reduced-harm products to help them switch to a less harmful alternative. Several submitters
commented that parents and guardians should be able to encourage minors in their care to switch
Official
from smoking to vaping should the need arise.
One submitter recommended that new section 40 be replaced with a framework similar to section
241 of the Sale and Supply of Alcohol Act 2012, which permits the supply of alcohol by or with the
permission of a parent or guardian.
Information
Stronger monitoring, enforcement and penalties for sales to minors
Some submitters thought that the monitoring and enforcement regime should be strengthened to
ensure that regulated products do not fal into the hands of young people, and several commented
that additional resources will be needed to carry this out effectively.
Several submitters recommended significant fines or suspension for retailers that sold to minors, while
others thought that a person’s ability to sell regulated products should be suspended either
temporarily or permanently if this were to occur.
Act
Comment
1982
New sections 39 (relating to sale and distribution) and 40 (relating to supply) carry over the existing
restrictions in the Act on supplying tobacco and herbal smoking products and extend these provisions
to all regulated products, including vaping products.
One of the purposes of the Bill as set out in clause 6 in new section 3A is to discourage non-smokers,
especial y children and young people, from taking up vaping or smokeless tobacco products. The
Ministry therefore considers that a minimum age restriction of 18 years for the sale of all regulated
products is appropriate.
34 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
link to page 41
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Online vendors are subject to the same requirements as other retailers and must ensure they take
reasonable precautions and exercise due diligence in ascertaining the purchaser’s age before
completing a sale. This includes sighting an appropriate evidence of age document that indicates
that the person is 18 years or older.
New technology is regularly being developed, and what is reasonable in terms of the level of
Released
precaution and due diligence an online retailer should undertake will change over time as
technology advances. In the Ministry’s view, the provision would be less enduring if the Bill included
a prescriptive requirement for a particular type of online age verification.
Risks associated with both online and in-person sale of regulated products to young people wil be
managed through the Ministry’s monitoring and enforcement processes, which include regular
compliance checks on retailers using control ed purchase operations.
4
New section 44 enables the Court to make additional orders against repeat offenders who sell or
under
distribute to people younger than 18 years of age. These orders include prohibiting the person from
selling a regulated product, or imposing conditions on the sale of those products, for a particular
time period.
We acknowledge the concerns raised by submitters about overseas advertisers targeting domestic
the
consumers (known as ‘cross-border’ advertising). The Bill regulates this type of advertising if the
person or company has a presence in New Zealand and the target audience includes New
Zealanders. However, it is not possible for New Zealand law to apply in other circumstances.
Official
The Ministry of Health consulted the Ministry of Justice on the penalty levels in the Bill, including the
maximum fines for a breach of the restriction on sales to under-18-year-olds. No changes to the
penalty levels in the Bil are proposed.
New section 40 applies to the supply of a regulated product ‘in a public place’ and would not prevent
a person from supplying minors with regulated products in a private setting.
Information
No changes are proposed to new sections 39 or 40.
Recommendation
No change.
Act
1982
4 Controlled purchase operations (CPOs) are designed to monitor and enforce the provisions relating to the sale
of regulated products to people under 18 years old. CPOs involve supervised volunteers aged between 14 and
17 years of age attempting to buy regulated products from retail premises, including online sellers.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 35
Packaging and label ing requirements
Clause 26: new sections 49 and 51: Packaging and label ing requirements
New sections 49 and 51 enable standardised packaging requirements to be set in regulations for al
regulated products, including requirements for messaging or information on packaging.
Released
Submissions
Around 600 submitters commented on standardised packaging. Over half of these submitters agreed
that the packaging of regulated products should be standardised.
Most of these submitters commented in the format of a questionnaire template that asked ‘Do you
think packaging should be sta
under ndardised (including non-nicotine)?’ Submitters provided a yes or no
answer, and most did not provide a rationale. Most of the submitters who completed the
questionnaire template stated that they were vape consumers. There was an even split
(approximately) between submitters who agreed and those who disagreed that packaging should be
standardised.
the
Most submitters considered there should be ful information on the packaging of the product (eg,
including nicotine content, ingredients and volume of liquid in a container) so consumers would be
ful y aware of what they were purchasing and could make informed choices.
Official
Over 50 submitters supported the inclusion of health information and health warnings on packaging.
Some of these submitters recommended that al nicotine-containing regulated products should
contain warning labels stating that nicotine is highly addictive.
A few submitters disagreed with ‘big’ warnings on packaging of vaping products, stating that there
was no evidence that nicotine was harmful.
Information
A few health sector submitters stated that health information on the packaging of vaping products
should include the potential benefits compared with tobacco products.
Comment
Standardised packaging requirements wil only come into effect for vaping products if regulations
are made. It is the Ministry's intention to develop tailored requirements for vaping products and
smokeless tobacco products (which at present are subject to the existing tobacco standardised
Act
packaging regulations).
Labelling requirements (eg, nicotine content, ingredients) will also be set out in regulations.
1982
The Ministry will publicly consult on the proposed labelling and packaging requirements, including
messages and information, for vaping and smokeless tobacco products that wil be set out in
regulations before any final decisions are made.
Recommendation
No change.
36 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
link to page 43 link to page 43
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Products labelled for chewing or other oral use
Clause 26: New section 53: Regulated products cannot be advertised or
label ed, etc, as suitable for chewing
New section 53(1) provides that a person must not publish a regulated product advertisement that
Released
directly or indirectly states or suggests that a regulated product is suitable for chewing or for any other
oral use.
New section 53(2) provides that a person must not import for sale, sel , pack or distribute any
regulated product labelled or otherwise described as suitable for chewing or for any other oral use.
Oral use is defined in new section 53(4) as the absorption of the product primarily through the oral
mucosa.
under
Submissions
Around 10 submitters commented on this section. Of these, most considered that the sale of
Swedish snus
5 should be al owed, referring to evidence for its long-term safety and its contribution
to low smoking rates and smoking-related il ness among men in Sweden.
the
These submitters considered that snus could contribute to reducing harm among New Zealand
smokers. One submitter noted that it may also be suitable as a reduced-harm alternative to vaping,
given the uncertainties around the long-term risks associated with vaping.
Official
A few submitters objected to the Ministry’s view that snus is prohibited under section 29(2) of the
current Act, arguing that this section was clearly intended to apply to chewing tobacco. One
submitter also took issue with the Ministry’s interpretation of the judgment in
Ministry of Health v
Phillip Morris (New Zealand) Ltd [2018] NZDC 44786 and the addition to the Bil of a definition of oral
use.
Information
Several submitters raised concerns that oral nicotine products that do not contain tobacco leaf and
are not approved medicines are not adequately regulated and suggested that this should be
addressed in the Bill.
One submitter opposed any relaxation of the prohibition on the sale of oral tobacco products. The
submitter’s reasons included that snus and other oral tobacco products undermine the Māori
community’s desire to be rid of tobacco and nicotine addiction, that there is evidence of harm (albeit
to a much lesser extent than that caused by smoking) and that there is evidence from Norway that
snus has attracted young non-smokers into nicotine addiction.
Act
Comment
1982
New section 53 is carried over from section 29(2) of the current Act, and a definition of oral use has
been added:
‘In this section,
oral use, in relation to a product, means the absorption of the product primarily
through the oral mucosa.’
5 Snus (pronounced ‘snoose’) is a moist, smokeless tobacco powder variant of dry snuff that is packed under
the top lip.
6 See: www.districtcourts.govt.nz/al -judgments/2018-nzdc-4478-moh-v-morris/
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 37
Snus is captured by section 29(2) of the Act because the use of snus fits directly within the words
‘other oral use’ in that section, as snus is used by being placed in a person’s mouth and absorbed
through the lining of the mouth and not smoked or inhaled. The Ministry is satisfied that the
judgment in
Ministry of Health v Phillip Morris does not change this. The definition of oral use is new
and simply clarifies the scope of this clause.
Released
The Ministry agrees that the evidence supports submitters’ comments that Swedish snus is
significantly less harmful than smoking. The Government is, however, not supportive of expanding
the range of nicotine-delivery products lawfully able to be sold in New Zealand.
The legal position of oral nicotine products that do not contain tobacco leaf is unclear. It is highly
likely that these products contain nicotine manufactured from tobacco, in which case their sale
would be prohibited under section 29(2) of the Act. However, whether the nicotine in these
products is manufactured from tobacco cannot be proved to the standard of evidence required for a
under
prosecution. Therefore, the Act cannot be adequately enforced for these products.
The Ministry agrees that the sale, supply and advertising of all oral nicotine-containing products
should be regulated. Despite a few submitters’ comments, there is no robust evidence to date to
support claims of the safety and effectiveness of these newer nicotine products in supporting people
to quit smoking.
the
The Ministry proposes that the Bil clarify that nicotine products for oral use (other than products
that have received consent for distribution under section 20, or provisional consent under section
Official
23, of the Medicines Act 1981) fall within section 29(2) of the current Act. This would mean that, like
chewing tobacco and snus, their import for sale and their sale, package, distribution, etc. would be
prohibited.
We consider that nicotine-containing products, other than those for oral use, should be regulated
under the Medicines Act 1981 (eg, nicotine products applied topical y, such as gels) and that this
Information
should be made clear to avoid any possible uncertainty.
Recommendations
We recommend that:
• the Bill be amended to regulate oral nicotine products (other than products that have received
consent for distribution under section 20, or provisional consent under section 23, of the
Medicines Act 1981) under new section 53 of the Bil
• a consequential amendment to the Medicines Regulations be made to clarify that al non-oral
Act
nicotine-containing products are medicines.
1982
38 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Obligations on retailers, including notifications and flavour
restrictions
Clause 26: New section 63
Released
New section 63 provides that a retailer must not sell:
• a notifiable product that has not been notified and does not comply with product safety
requirements, etc
• vaping products that are not listed in Part 1 of Schedule 2 (ie, tobacco, menthol, and mint),
unless they are a specialist vape retailer selling from their approved premises or Internet site.
• a prohibited flavour listed in Part 2 of Schedule 2 (there are none currently proposed).
under
Submissions
Over 1,000 submitters commented on new section 63. The overwhelming majority of these focused
on the restrictions on flavours that generic retailers can sel .
the
Obligation on retailers to only sel products that meet product safety requirements
One submitter disagreed with the retailer obligation that they only sel products that meet safety
Official
requirements, expressing concern that it would unduly shift the responsibility to the retailer. Their
view was that responsibility for compliance with product safety requirements should sit solely with
the notifier (ie, the manufacturer or importer) and that retailers should rely on the notification
scheme to uphold safety standards.
Information
Restriction for generic retailers to sell only tobacco, menthol and mint-flavoured vaping products
A large majority of submitters commented on the flavours that can be sold in generic retail stores.
Many of these submitters were either small retailers, vape consumers, vape businesses or
organisations in the health sector, including health research organisations and NGOs.
Most submitters disagreed with the restrictions on flavours. Those who provided further comments
said the restrictions would:
a. discourage smokers from transitioning or maintaining their transition to vaping as tobacco,
menthol and mint flavours are unappealing
Act
b. negatively affect health equity by disproportionately impacting smokers who live in rural or low
socioeconomic areas who cannot purchase products online or who live in areas without specialist
vape retailers (a higher proportion of these people being Māori)
1982
c. encourage people to tamper with vaping products by adding their own flavours and/or create a
black market for flavours.
Almost all small retailers disagreed with the proposed flavour restriction and raised further
concerns. For example, that the flavour restriction:
a. will significantly impact on their income from vaping products as vape consumers prefer fruit
and/or dessert flavours
b. will create an ‘unfair commercial advantage’ between generic retailers and specialist vape stores
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 39
c. will not affect youth vaping rates as businesses routinely ask for ID when selling vaping products
d. is disproportionate as generic retailers are allowed to sell a range of tobacco products.
Some vape consumers who disagreed with the restrictions stated that it negatively impacted on
consumers’ ‘choice’ and ‘right’ to purchase and use different flavours.
Released
More flavours in generic retail stores and store exemptions
Some submitters suggested that the number of flavours in generic retail stores should be increased
to four, six or ten flavours (including fruit, vanil a and/or dessert flavours).
Other submitters suggested that to ensure smokers can access a range of flavours, the Bil should
permit at least R18 stores, liquor stores, internet vendors, pharmacists or tobacconists to sel more
flavours and allow generic retailers, who are ‘outside a certain distance from a specialist vape store’,
under
to apply for a special licence, which would allow them to stock more flavours.
Agreement with the restriction on flavours
Approximately two-thirds of health organisations who commented on flavours either supported the
the
restriction on flavours in generic retail stores or recommended further restrictions to prevent young
people from accessing vaping products. They, along with other submitters who supported additional
restrictions, suggested only allowing generic retailers to sell tobacco-flavoured vapes or only
Official
permitting three flavours to be sold provided that generic retailer stores cannot sell vaping devices.
A minority of vape consumers and vape businesses agreed with the proposed flavour restrictions for
generic retail stores. Those who gave reasons said:
a. the restrictions would reduce the attractiveness of vaping to young people
b.
Information
generic retailers do not provide good-quality vaping products or advice and specialist vape stores
are better able to support smokers to transition than generic retailers
c. restricting flavours would have little impact on vaping consumers as vaping products can be
purchased online.
Support for broader restrictions on flavours, including in specialist vape retailers
Over 40 submitters stated that al flavours that appeal to young people should be banned.
A very smal number of submitters suggested limiting the range of flavours that could be sold in
Act
specialist vape stores. One submitter suggested that restrictions could later be reduced for specialist
vape stores if ‘supported by evidence’.
A smal number of submitters specifical y focused on the definition of ‘flavour’. They disagreed with
1982
‘the use of the broad term flavour’ and said it is important to be clear about how a flavour is being
defined. Submitters noted that a flavour can be defined in at least three different ways.
a. A chemical formulation (focusing on specific ingredients)
b. A subjective sensation (ie, the person believes it tastes like apple)
c. A descriptor (ie, the name given to the flavour, eg, ‘unicorn puke’).
The submitters suggested that the focus should be on chemical formulation, with one submitter
suggesting a focus on flavour descriptors or trademark violation.
40 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Some submitters considered that the focus should shift from flavours or, in addition to focusing on
flavours, to a focus on regulating:
a. flavour descriptors that may be attractive to young people, for example, label ing a vape flavour
as ‘strawberry’ rather than ‘berry delicious’
b. youth-centric marketing of vape flavours
Released
c. the toxicity of particular ingredients and flavours.
We comment on descriptors and youth-centric marketing in the advertising section of this report. The
toxicity of ingredients will be considered in the development of product safety regulations.
Comment
Notification requirements
under
The retailer has the obligation to take reasonable precautions to ensure a product meets safety
requirements at the time of sale. Retailers will be able to use the notification database to check that
a product is approved and all requirements have been met before they sell a product.
the
Flavours
The Bill aims to strike a balance between protecting young people from the risks associated with
Official
vaping products and supporting smokers to switch to much less harmful alternatives.
A brief summary of the evidence on flavours has been provided to the Committee (as part of the
Ministry’s additional advice, dated 9 April 2020).
There is no strong evidence that particular flavours are important for smoking cessation. However,
there is evidence to suggest that flavours add to the appeal of vaping. For smokers, especial y those
Information
who are unable to quit by conventional means, having a product available that is less harmful and at
least as satisfying as smoking is important.
Some studies have highlighted concerns that some flavours are appealing to young people and may
be a factor in youth vaping uptake. A recent review showed that young people prefer non-tobacco
vaping products, especially sweet flavours. There is also some evidence that sweet flavours are
perceived as less harmful. For young people who have never smoked, there are likely to be some
health risks associated with regular long-term vaping. Therefore, policies and interventions that
reduce access and uptake are warranted.
Act
If the Committee wishes to consider whether the Bil has the balance right and is supportive of
proposing amendments to the restriction on which flavours may be sold by generic retailers, then
increasing the scope of flavours available in generic retail stores would be the simplest way to
1982
achieve this (ie, by adding further flavours or categories of flavour to Schedule 2, Part 1).
Recommendation
No change.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 41
link to page 48
Powers of the Director-General of Health
Clause 26: New sections 24 and 67–72
This part of the departmental report discusses all the Director-General’s powers and related
submissions together because most submitters commented broadly rather than on individual
Released
provisions. Below are the relevant clauses of the Bill that involve the exercise of Director-General
powers.
New sections 67-72 provide that the Director-General may:
• declare a substance to be a prohibited ingredient
• require a notifier to provide information relating to the safety of the product
• issue a public warning or statement if a product poses a risk of harm to people, or require the
notifier to arrange for the product to be recal ed
under
• suspend or cancel a product notification.
The exemptions in new section 24 that involve the exercise of Director-General powers are:
• a public health message approved by the Director-General
the
• approval of suitably qualified health workers to provide advice or messages for the purpose
of supporting a person to switch from smoking to vaping.
Official
Submissions
Over 600 submissions were received on the powers of the Director-General, most of which were
responding to a form submission questionnaire that asked: ‘Should al power for future changes be
left to the Director-General?’
Information
Most submitters simply answered ‘no’, or that it would be unfair or undemocratic for al power to be
with one person. A few submitters were concerned that the Director-General was ‘biased’ or did not
have the technical knowledge to make decisions under the Act.
Submitters who provided a more substantive response suggested various changes to the Bil ,
including that there should be greater transparency about how the powers wil be used or that there
should be consultation with experts, industry, suppliers and end users. One submitter was
particularly concerned that new section 67 (declaration of prohibited ingredients) was tantamount
to a delegated power to make regulations.
Act
Comment
1982
Concern about powers residing in one person
As with the current Act, in practice, the Ministry wil carry out many of the regulatory powers under
delegated authority (eg, product safety wil be managed within Medsafe
7). In response to
submitters’ concerns that the Director-General may not be qualified to make decisions and
assessments about vaping products, in practice, the Ministry seeks and provides the Director-
General with advice on available international best practice.
7 The New Zealand Medicines and Medical Devices Safety Authority, a business unit of the Ministry of Health
and the authority responsible for the regulation of therapeutic products in New Zealand.
42 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
From time to time, the Director-General also seeks advice from technical specialists and specialist
advisory committees under several regulatory regimes that the Ministry administers. The Bil does
not currently require the Director-General to establish an advisory committee to inform the exercise
of their powers.
There are various approaches to advisory committees in health legislation, including no requirement
Released
to establish a committee, providing the discretion to establish one when necessary or requiring their
establishment.
An example of where the regulator must establish a committee is the Psychoactive Substances
Expert Advisory Committee (PSEAC) established under section 11 of the Psychoactive Substances Act
2013. PSEAC comprises up to six members with technical expertise in pharmacology, toxicology,
neurosciences, medicine and any other areas the PSEAC considers relevant.
The Ministry’s preference would be to empower the Director-General to, from time to time,
under
establish and have regard to advice from technical advisory committees on the exercise of the
Director-General’s powers. This is in preference to requiring a specific standing committee to be
established. For example, a new provision could be inserted that sets out the kinds of matters that
an expert committee advises on and that signals the intent that such committees could include
members of the vaping industry, wher
the e appropriate.
The kinds of advice the advisory committee could give might include: the specific effects of a
product, any risks to public health, the likelihood of the product creating physical or psychological
Official
dependency, its appeal to vulnerable populations, the likelihood of its misuse and any other matters
the Director-General considers relevant.
Concerns about the extent of the powers
In terms of the extent of the Director-General’s powers, the Bil adopts a similar approach to pre-
Information
market entry notification features in other established regulatory schemes; either those
administered by the Ministry or those in similar jurisdictions. The Ministry does not recommend
removing any of the Director-General’s powers, but we recommend the Committee consider
introducing safeguards, as set out below.
Industry involvement in rule changes / prohibition of ingredients
In response to submitters’ cal s for industry involvement in ‘rule’ changes, this must be subject to
the limitations on industry involvement under the World Health Organization’s (WHO’s) Framework
Act
Convention on Tobacco Control (FCTC), to which New Zealand is bound as a party.
The Director-General and the Ministry must act to protect public health policies from commercial
1982
and other vested interests in the tobacco industry (Article 5.3). The New Zealand government must
observe complete transparency in any dealings with the tobacco industry. The Ministry considers
some similar considerations apply to engaging with the vaping industry, some of which are also
tobacco industry stakeholders. However, the Ministry acknowledges that, with respect to vaping
products, this needs to be balanced by drawing on appropriate technical expertise, some of which
will reside within vape businesses.
In terms of the suggestion to impose a specific requirement on the Director-General to consult
before declaring an ingredient to be prohibited under new section 67, the Ministry considers that,
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 43
for public safety reasons the Director-General must be able to act immediately once there is
sufficient information available. Despite this, the expectation is that the Director-General will
consult, where reasonably practicable in the circumstances, with a range of technical specialists.
However, to address submitter concerns, the Ministry recommends tightening new section 67 to
specify criteria that the Director-General must take into account before declaring an ingredient to be
Released
prohibited. Criteria could include the risk of harm arising from the use of the ingredient, the history
of any beneficial use of the ingredient, the stance taken by other recognised regulatory authorities
overseas and any other matter the Director-General considers relevant.
Consultation on application of powers
As best practice, the Director-General’s delegates will in many cases consult with those affected
before making a final decision. However, considering the nature of the Director-General’s powers,
under
most would be less effective if the Bill required prior consultation with those affected in all cases.
For example, the Director-General may need to act very swiftly to protect the public by recalling a
product or prohibiting an ingredient that has been the subject of adverse event reports.
However, in relation to powers to suspend or cancel a product notification under new sections 71 or
the
72, the Ministry recommends including an express Director-General obligation to give the notifier a
reasonable opportunity to be heard before any suspension or cancellation occurs. This is taking
account of natural justice principles and the impact on the notifier’s livelihood and reputation. The
Official
obligations to act reasonably and to provide reasons already apply to the Director-General under
these draft sections, but there is no explicit right to be heard.
Providing a right of appeal against suspension or cancel ation
The Bil does not provide a right of appeal against the Director-General’s decision to suspend or
Information
cancel a product notification of a notifiable product. Under general law, the notifier would have a
right to apply to the High Court to judicially review the process leading to cancellation. However,
that is not an appeal on the merits of the decision, and judicial review may be outside the reach of
smaller businesses.
While the Director-General’s decision to suspend is by its nature an interim measure, as with
cancel ation, it may result in the vape business going out of business. For this reason, we
recommend that the Bil should provide the notifier with a right of appeal for decisions to
suspend or cancel. The committee may wish to consider whether other aspects of the Bill
Act
conferring Director-General powers should give right of appeal (eg, recal of a product).
In terms of what the right of appeal would look like, the Psychoactive Substances Act 2013 (section
45) includes a right of appeal to an appeals committee of three members appointed by the Minister
1982
and a further right of appeal on questions of law to the High Court. An alternative (and older) model
under regulation 65 of the Medicines Regulations 1984 allows a right of appeal to the District Court
within 14 days of being notified of the decision in writing, and the decision of that court is final. The
Ministry has a slight preference for the first model.
44 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Requiring reasonable grounds for recal
The Ministry considers that the Director-General’s power to recal a product under new section 70
should be on reasonable grounds. New section 70 requires that the Director-General be ‘satisfied
that the continued availability of the product poses an unacceptable risk of harm to people’. In
requiring that the Director-General must be satisfied ‘on reasonable grounds’ before resorting to
Released
recall, this implies the Director-General’s grounds for doing so must be clearly identified and
transparent.
Recommendations
We recommend that the Bil be amended to:
• empower the Director-General of Health to set up technical advisory committees to assist in
decision-making and exercise of powers under the Bil once enacted
under
• provide the notifier a reasonable opportunity to be heard before the Director-General of Health
can suspend or cancel a product notification of a notifiable product
• provide a notifier with a right of appeal against a decision to suspend or cancel a product
notification of a notifiable product similar to the appeal right in section 11 of the Psychoactive
Substances Act 2013 (ie, a right of appeal to an appeals committee of three members appointed
by the Minister of Health, and a further right of appeal on questions of law to the High Court)
the
• require the Director-General of Health to be satisfied on reasonable grounds that the product
presents an unacceptable safety risk before making a recall order.
Official
Information
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 45
Prohibited ingredients and colouring substances
Clause 26: New section 66: Substances that notifiable products must
not contain
New section 66 provides that a vaping substance must not contain a prohibited ingredient, a
Released
prohibited flavour or a colouring substance.
Submissions
Prohibited ingredients
Several submitters commented on proposals to prohibit ingredients. Some made specific
suggestions, for example, to prohibit diacetyl, all non-water-soluble oils and vitamin E acetate.
under
One submitter suggested that maximum concentrations (rather than a complete ban) be set for
prohibited ingredients. Another submitter suggested that prohibited ingredients should be listed
alongside prohibited flavours in schedule 2 of the Act.
the
Colouring substances
In addition, several submitters commented on colouring substances.
Official
A couple of submitters agreed with prohibiting colouring substances, with one submitter stating that
it is not a ‘major disincentive’ for vaping consumers. One submitter noted that flavours are
‘inadequately regulated’ under the Australia New Zealand Food Standards Code.
One submitter disagreed with the placement of this section. They contend that it belongs in the
regulations (rather than the Act itself) as it can be amended more easily if the evidence on colouring
Information
substances in vaping products changes.
Comment
The Bill provides that the Director-General may declare a substance a prohibited ingredient if
satisfied that the substance is unsafe. The Ministry has prepared a draft list of prohibited ingredients
that will be consulted on before it is finalised.
The Ministry agrees that it would be desirable to allow the Director-General to set limits for
Act
ingredients so that, in some cases, an ingredient may be al owed up to a maximum concentration.
The Bill does not currently allow for this.
The prohibition on colouring substances is appropriately placed in the Bill rather than regulations.
1982
There is no evidence to support the safety of colouring substances and no good reason for their use
(eg, unlike flavours, they do not play an important role in supporting smokers to switch).
Recommendation
We recommend that new section 67 be amended to enable the Director-General of Health to set
maximum limits for ingredients, in addition to outright prohibiting ingredients.
46 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Establishment of notification database
Clause 26: New section 73: Establishment of database and
confidentiality of certain information
New section 73 provides that the Director-General of Health must establish and maintain a
Released
database, ensuring that confidentiality of information is protected.
Submissions
One submitter proposed that the Ministry specify the information to be col ected through the
notification system and that the notifiable product industry should be responsible for implementing
and maintaining a low-cost, online, self-service notifications system.
under
Comment
The Ministry is planning to implement a low-cost, online, self-service system to support notifications
(ie, the same objectives as proposed by the submitter above).
the
The planned system wil be a bespoke configuration of a common Ministry-wide platform that
supports workflow for processing and publishing applications and a number of other functions as
Official
well as notifications.
The system must meet key security, performance and availability requirements, and a common
Ministry platform will be better able to support these requirements than a sector-developed system
for vaping notifications.
The common platform wil initial y support two Ministry regulators, but current plans are to extend
Information
its use to several other areas over the next five to ten years. This will enable the Ministry to establish
a standardised workflow for receiving, processing, responding to and publishing applications, etc,
across multiple business units. Common core systems and a standardised workflow are expected to
provide significant efficiency savings in each area.
The scope of the planned system is significantly beyond what could reasonably be delivered by the
vaping industry. The overal cost to the industry is expected to be significantly lower on the common
platform compared with a bespoke industry solution as sectors served by other business units will
also be using the common platform.
Act
Recommendation
1982
No change.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 47
Regulations
Clause 26: New sections 75–80
New sections 75–80 provide for making regulations for specified purposes, including:
Released
• forms, registers and other documents
• health messages on automatic vending machines
• exemptions
• acceptable forms of delivery and visibility
• health information and warnings at point-of-sale and on the Internet
• information that must be contained in annual tax returns
• specifying requirements relating to the standardised packaging (including messages and
information)
under
• notifiable products (including the prescribing of product safety requirements)
• imposing fees and levies.
Submissions
the
Around 70 submitters commented on these sections. These comments were mostly related to the
details of the regulations, for example, maximum nicotine level. Proposals for the regulations will be
Official
developed and publicly consulted on in due course.
One submitter suggested that the scope of the regulations was too broad and that health officials
may give effect to prejudices with respect to harm reduction. The submitter argued that Parliament
should retain ownership over the regulations or be able to review them.
Another submitter argued that more of the provisions of the Bil should be in regulations to al ow
Information
greater flexibility should issues develop in the future that might require action faster than a change
to primary legislation allows.
Yet another submitter felt that the regulations needed to be responsive to new products and
technologies.
Comment
The Ministry is satisfied that the current scope of the regulations is sufficient for the legislative
regime to work effectively and that there is appropriate oversight, that is, by Cabinet and the
Act
Regulations Review Committee. Full public consultation will occur before any regulations are
created.
1982
We agree with the suggestions that maximum limits should be able to be set for ingredients in
addition to ful prohibition.
Recommendation
No change (except for the regulation-making powers recommended in the body of this report).
48 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Infringement offences
Clause 26: New sections 81–84
New sections 81–84 set out the procedure for the infringement offences in the Bill, including notices
and payment of infringement fees.
Released
Submissions
Very few specific comments were made by submitters on the infringement offences. One submitter
suggested adding a further range of offences to the infringement notice regime.
under
Comment
The Act currently provides for infringement notices for a range of offences related to tobacco
products. The Bil proposes the establishment of an infringement regime for al regulated products.
The infringement regime can only apply to strict liability offences. We have followed Ministry of Justice
guidelines on the establishment of infringement offences and the setting of the infringement fees
the
associated with these offences.
Official
Recommendation
No change.
Information
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 49
Enforcement officers
Clause 26: New sections 85–93
New sections 85–93 provide for the appointment of enforcement officers by the Director-General.
These sections provide safeguards relating to the powers of entry and inspection and the power to
Released
require information.
An enforcement officer is provided with protection from civil and criminal liability and granted
powers of entry and inspection, including being able to apply for a search warrant in certain
circumstances.
The provisions also provide for an enforcement officer to require a person to provide identifying
information in specified circumstances.
under
Enforcement officers are required to identify themselves when exercising certain powers. Offences
for intentional y obstructing, hindering or resisting enforcement officers and providing false or
misleading information are also included.
the
Submissions
Appointment of enforcement officers
One public health unit and an individual submitter suggested that police and customs officers should
Official
have reciprocal powers as both play key roles in the regulation and enforcement of tobacco laws.
Search powers
One submitter was concerned about the search powers listed in section 87, which provides for
Information
warrantless searches if ‘the officer believes on reasonable grounds that it is a place to which this
section applies’. They believed this violates section 21 of the BORA (unreasonable search).
The submitter was also concerned that section 87(4) al ows police to accompany the health
inspector on the warrantless search, which weakens police controls under the Search and
Surveil ance Act 2012, and that police powers should not be included in a health bil . The submitter
considered the search warrant provisions in section 89 to be appropriate but noted that the Bill does
not provide any reporting, oversight or complaints processes. The submitter recommended that
section 87 be removed from the Bill and that the Bill be amended to acknowledge that the reporting
requirements in section 171 of the Search and Surveillance Act 2012 apply to all searches.
Act
One public health unit submitted that enforcement officers are currently only allowed to ‘inspect’ a
retailer, which suggests officers can only look but not touch anything, such as opening a cupboard to
look inside without a search warrant. The submitter considered that this is too much of a constraint
1982
on enforcement officers. This view was supported by another submission from an individual.
Resources
One submitter stated that additional resources will be required to effectively monitor and enforce
the legislation to ensure minors do not access tobacco or vaping products. Another submitter was
concerned that vaping products wil be subject to the same lack of monitoring and regulation
enforcement as the alcohol licensing regime.
50 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Comment
The new enforcement officer provisions have been carried over from the provisions in the current
Act and extended to al regulated products. The Ministry believes the provisions contain appropriate
safeguards regarding the powers of entry and inspection.
Released
Recommendation
No change.
under
the Official
Information
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 51
Annual returns and reports
Clause 26: New section 94
New section 94 contains annual reporting requirements for manufacturers and importers of
regulated products and specialist vape retailers.
Released
Submissions
A smal number of submitters supported the requirements for annual reporting. One submitter
recommended that all retailers selling vaping or smokeless tobacco products, not just specialist
retailers, should be covered by these requirements.
under
Comment
This is a carry-over provision and is a current requirement for manufacturers and importers of
tobacco products. New section extends the scope of the current provision to al regulated products.
The requirement on specialist vape retailers to report is new and does not exist for generic retailers
the
or for retail of tobacco products. The reporting requirement for specialist vape retailers aims to
ensure that those retailers continue to satisfy the sales requirement in new section 14A(2)(b).
Official
Recommendation
No change.
Information
Act 1982
52 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Section 4: Minor and technical changes
Recommendation
We recommend that the fol owing minor and technical changes be made to the Bill.
Released
• The following definitions in section 2(1) and (2) of the current Act should be amended to
include al regulated products (they currently just apply to tobacco):
o
Automatic vending machine
o
Distributor
o
‘Of the same kind’
• The Bill should be amended to ensure that a vaping substance does not include medicinal
cannabis or a cannabidiol (CBD) product (as these are regulated under the Medicines Act 1981
and the Misuse of Drugs Act 1975).
• The Bil should be amended to enable the current regulations to continue to apply to tobacco
under
products and, where applicable, herbal smoking products, until a new set of regulations
applying to regulated products comes into force, as was the policy intent.
the Official
Information
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 53
Section 5: Out-of-scope comments noted in submissions
Comments on tobacco and tobacco products
Over 60 submitters provided comments on tobacco and tobacco products. Over half of these
submitters (including many DHBs and NGOs) commented on the need to reduce the retail supply of
Released
tobacco products. Most of these submitters recommended that the Bil be extended to prohibit the
sale of tobacco products by general retailers, including dairies, service stations and supermarkets.
A couple of submitters commented that substantial restrictions on smoked tobacco products would
be consistent with the more restrictive availability of vaping products as proposed in the Bill.
These comments/recommendations are out of scope for the Bill. In 2019, Associate Minister of
Health, Hon Jenny Salesa indicated her intention to introduce an action plan to accelerate progress
towards the Smokefree Aote
under aroa 2025 goal. A draft plan wil be publicly consulted on before it is
finalised. This consultation will provide the public with the opportunity to submit on issues relating
to tobacco products.
the
Vaping products should be regulated under the Medicines Act
New subsection 2(4) in clause 5(4) stipulates that vaping devices are not medical devices and vaping
substances are not medicines.
Official
Seven submitters, including two from a single organisation, proposed that vaping products should be
assessed through the same safety standards applied to other ‘quit smoking’ products, such as
nicotine replacement therapies (NRTs).
NRTs are currently regulated as medicines or medical devices under the Medicines Act 1981.
Information
The approach proposed by submitters is contrary to government policy on the regulation of vaping
products, which is to strike a balance between protecting young people from the risks associated
with vaping products and supporting smokers to switch to much less harmful alternatives.
Regulating vaping products as medicines or medical devices would place both up-front and ongoing
compliance obligations on manufacturers and importers, which would have a significant impact on
the availability of products for existing smokers.
Act
1982
54 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Appendices
Appendix 1: Submitters’ details
Oral
Released
Category / Name of submitter
Organisation
submission
Businesses: Tobacco and vaping industries
British American Tobacco New Zealand
British American Tobacco New Zealand
Yes
Chris Woods
Japan Tobacco International
Imperial Brands New Zealand
Imperial Brands New Zealand
Yes
Dr James Murphy (BAT)
Nicoventures Trading Ltd
Yes
Kaine Thompson, Claas H Schberg
JUUL Labs
Yes
Lion Labs
Lion Labs
Yes
under
Mission Limited
Mission Limited
Yes
Vaping Trade Association of NZ
Vaping Trade Association of NZ
Yes
Businesses: Other
Paul Rayner
A tourism company
the
Anne Harris
Pfizer NZ
Myriad Pharmaceuticals
Myriad Pharmaceuticals
Yes
Sue and Teresa Taylor
T and T Consulting Ltd
Yes
Official
Business associations
Business NZ
Business NZ
Dr Eric Crampton
The New Zealand Initiative
Yes
Greg Harford
Retail NZ
NZ Food and Grocery Council
NZ Food and Grocery Council
Dave Hooker
NZ Association of Convenience Stores
Yes
Information
New Zealand Law Society
New Zealand Law Society
Large general retailer organisations
Matthew Lane
Night n Day Foodstores
Yes
Melissa Hodd
Foodstuffs (N.Z) Limited
Yes
Vape stores
Antifun Ltd TA Premium Vape
Antifun Ltd TA Premium Vape
Coastline vapes
Coastline vapes
Cosmic
Cosmic
Yes
Easy as E-Cigs Ltd
Easy as E-Cigs Ltd
Act
Michael Brader on behalf of team
Hawkes Bay Vapour
Yes
Tracy Pile
Infused Oamaru
Yes
Paul Elton
Jock’s Vapes, Upper Hutt
1982
Jubby's Juice Ltd
Jubby's Juice Ltd
NZ Vapour
NZ Vapour
Morris Lazootin and Savvas Dimitriou
Te Wairua Limited
Yes
Clint Baxter
Vape Merchant Ltd
Yes
VAPO staff survey
VAPO
Vaporium
Vaporium
Kevin Carroll
Vive Vape Co
Yes
Robert Reid
Global Innovations Ltd
Yes
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 55
Oral
Category / Name of submitter
Organisation
submission
District health boards (DHBs) / public health services
Richard Portch
Auckland Regional Public Health & Auckland
Yes
Metro DHBs
Released
Evon Currie
Canterbury DHB
Yes
Nicholas Jones
Hawke's Bay DHB
Waikato DHB
Waikato DHB
Whanganui DHB
Whanganui DHB
Dr Jose M Ortega Benito
Nga Tai Ora Public Health Northland
Toi Te Ora Public Health
Toi Te Ora Public Health Tauranga
Primary health organisations (PHOs) / entities
Anoop Gopalakrishnan
Wel South Primary Care Network
under
Irihāpeti Mahuika
Pegasus Health (Charitable) Ltd
Yes
Mahitahi Hauora Primary Health Entity
Mahitahi Hauora Primary Health Entity
Health profession associations
Dr Felicity Dumble
NZ College of Public Health Medicine
Dr Jeff Brown
Royal Australasian Col ege of Physicians
Yes
the
Dr Kate Baddock
NZ Medical Association
Yes
Nicola Hill
Royal Australisian College of Surgeons
Yes
NZ Nurses Organisation
NZ Nurses Organisation
Official
Pharmaceutical Society of NZ
The Pharmaceutical Society of NZ
Phlip Pattmore
The Paediatric Society of NZ
Yes
Prudence Stone
Public Health Association of NZ
Yes
Royal NZ College of General Practitioners
Royal NZ College of General Practitioners
Health sector NGOs, coalitions and councils
ASH (Action for Smokefree NZ)
ASH (Action for Smokefree NZ)
Yes
Information
Associate Professor Colin Menelsohn
Australian Tobacco Harm Reduction Association Yes
Bridget Forsyth
Smokefree Murihiku
Barbara Hol and and Barbara Robson
Federation of Women's Health Councils
Cancer Society
Cancer Society of NZ
Yes
Carly McDowell
Smokefree Mid Canterbury
Catherine Manning
Takiri Mai te Ata Regional Stop Smoking Service Yes
Dr Alex Wodak
Australia Drug Law Reform Foundation
Yes
Dr Nicki Jackson
Alcohol Healthwatch
George Laking
End Smoking New Zealand
Yes
Act
Hawkes Bay Smokefree Coalition
Cancer Society
Heart Foundation
Heart Foundation NZ
1982
Jo Miller (for Healthy Families Hutt Valley
Healthy Families Hutt Valley
Strategic Leadership Group)
Letitia Harding
Te Ha Ora Asthma and Respiratory Foundation
Yes
Lisa Hesp
Executive Group, Smokefree Canterbury
Yes
Mark Vivian
Stroke Foundation of NZ
New Nicotine Alliance
New Nicotine Alliance
Philip Hope
Lung Foundation NZ
Yes
Ross Bell
NZ Drug Foundation
Yes
Sophie Carty
Smokefree Otago
56 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Oral
Category / Name of submitter
Organisation
submission
Trish Fraser
Auckland Women’s Health Council
Yes
West Coast Tobacco Free Coalition
West Coast Tobacco Free Coalition
Robyn Harris
The Pharmacy @ Your Community Charitable
Yes
Released
Trust
Iwi and Māori health providers
Te Runanga o Ngati Whatua (iwi)
Te Runanga o Ngati Whatua
Fred Sadler
Te Hau Ora O Ngapuhi
Collective of Māori health and iwi health
Cancer Society Northland (on behalf of the
Yes
providers
collective)
Hapai te Hauroa Tapui Ltd
Hāpai te Hauora Tapui Ltd
Yes
Rebecca Ruwhiu-Collins
Vape2Save
under
Taki Tahi Toa Mano
Taki Tahi Toa Mano
Yes
Pacific health providers
Edward Cowley
Tala Pasifika
Maria Meredith
PACIFICA Tamaki (Women’s) Branch
Yes
Tofilau Bernadette Pereira
PACIFICA Inc
Yes
the
Other: NGOs, advocacy groups, student association
Coalition of Asia Pacific Tobacco Harm
Coalition of Asia Pacific Tobacco Harm
Advocates
Advocates
Official
Nancy Loucas
Aotearoa Vapers Community Advocacy (AVCA)
Yes
Jordan Williams
NZ Taxpayers Union
Yes
NZ Council for Civil Liberties
NZ Council for Civil Liberties
Yes
Matt Holden
Free Speech Coalition
Francesca Dykes
Otago University Students Association
Yes
Sisi Tuala Leafa
Hash Tags (Youth Empower)
Yes
Information
Universities
Chris Bullen and Natalie Walker
National Institute for Health Innovation, School
of Population Health, University of Auckland
David Sweanor
Advisory Board, Centre for Health Law, Policy &
Ethics, University of Ottawa
Various people
Tobacco and Alcohol Research Group,
Yes
University College of London
Martin McKee
London School of Hygiene and Tropical
Yes
Medicine, University of London
Various authors
Adolescent Health Research Group, University
Yes
Act
of Auckland with University of Otago
Janet Hoek
ASPIRE 2025, University of Otago
Yes
Schools
1982
Harbour Montessori College
Harbour Montessori College
John Rogers
Sancta Maria College
Territorial authorities
Brendan Anstiss
Christchurch City Council
Small general retailers
Abdal Soltan
Auckland city central dairies
Alan Dinning
Alankar Patel
The Corner dairy
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 57
Oral
Category / Name of submitter
Organisation
submission
Albert Street Dairy
Albert Street Dairy
Ali Mahmood
Alpesh Patel
Grove Store, Papakura
Released
Yogesh Patel
Te Puke Street Food Store
Amarijit Singh
store
Amit Patel
Mirrabooka Superette
Anjana Rama
Eastbourne Dairy
Anju Patel
McDivitt Superette,
Anthony Tea
Mobil Mart Mt Albert
Ara Smith
Aro Val ey Mini Mart
Aro Val ey Mini Mart
under
Arvindbhai Patel
Ashish Patel
Mini Mart Herne Bay
Asvin Patel
Avinesh Mudalair
Piopio Superette
Baljinder Singh
Yes
the
Balvir Singh
Belt Road Supermarket
Belt Road Supermarket
Bharat Patel
Shree Superette
Official
Bhavesh Patel
Masala's Convenience Lower Hutt
Bhavna Patel
Jyotis Dairy
Bid Basket Foodstuffs Bell Block
Bid Basket Foodstuffs Bell Block
Bimal Singh
Albany Highway Superette and Lotto
Bina Sheth
Puhinui Mini Mart
Information
Brad Sissons
Liquor store- Bay of Islands
C R Dairy
C R Dairy
Cambridge Corner
Cambridge Corner
Capital Market
Capital Market
Capital Mart Featherston
Capital Mart Featherston
Chao Weng
Polson St Foodmarket
ChengWei Ge
Discounter Stores
Chet Ankumar Amrutbhai Patel
Larnoch Superette
Chintu Gandhi
One Stop Super Shop
Act
Choices Dairy and Takeaways
Choices Dairy and Takeaways
Chris Ayto
City Cards and Mags
City Cards and Mags
1982
City Mart Hamilton
City Mart Hamilton
City Mart Wellington
City Mart Wellington
City Mini Market group
City Mini Market group
City Stop Manners Street
City Stop Manners Street
Coinsave
Coinsave
Coronation Dairy
Coronation Dairy
Craig Pitman
Crescent Dairy
Crescent Dairy
58 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Oral
Category / Name of submitter
Organisation
submission
David Davies
Edmund Road Auto Mart (Gul )
David Zhang
Ponsonby Superette
Delowar Hossian
Parkland Superette, Ramarama
Released
Dhansookh Dajee
Plaza Superette Mt Albert
Yes
Dharmesh Jarem
Coronation Superette
Dharmesh Modi
Rongotea Food Square
Dharmesh Patel
Dimpal Kumar
Dipak Patel
Dipika Patel
Woodward Dairy
DJ's Dairy
DJ's Dairy
under
Fang Niu
Midtown Superette
Four Square Eketahuna
Four Square Eketahuna
Frankleigh park dairy
Frankleigh park dairy
GandT Happy Variety NZ Ltd
GandT Happy Variety NZ Ltd
GAS Eketahuna
GAS Eketahuna
the
Gas Linton
Gas Linton
Gas Waikari
Gas Waikari
Gaurang B Patel
Westpark Superette
Official
Gaurang Pandya
Gaurang Patel
Handy Store, Papatoetoe
George Ding
Discount Mal
Good Value
Good Value
Greens Dairy
Greens Dairy
Information
Hao Xu
Hardik Rokadia
Fenton Park Dairy, Rotorua
Harry Dahya
Herbert Ave Dairy
Harshad Patel
Chivalry Foodmart
Hataitai Dairy
Hataitai Dairy
Haven Road Store
Haven Road Store
Hawera Discount Specialist
Hawera Discount Specialist
Hemal Gandhi
Carnation Superette
Heretanga Hospital Store
Heretanga Hospital Store
Act
Hermant Patel
Hiren Ahir
Alexander Food Market, Palmerston North
HiWay Dairy
HiWay Dairy
1982
Ikramulhaq Patel
Elm Street Dairy, Waiuku
Imran Keten
Kirons Convenience Store
Inglewood Dairy
Inglewood Dairy
Jagroop Singh
Village Foodmart Tuakau
James Godinet
Metromart group
Jane Ling
Jatinder Singh
Jay Modi
Southbridge Superette, Canterbury
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 59
Oral
Category / Name of submitter
Organisation
submission
Jayesh Patel
Beach Haven Discount Superette
Jennifer Gin
dairy owner
Jeremy Dunedin
Mobil Group
Released
Jian Li
Discount Dairy & Vapour
Jimil
Prachi Enterprises Ltd
Jitesh Patel
Creswik Food Market
Jo-anne Thomson
Food for Though Café & Takeaways
John Zao
Kaimanawa Food Market
Kaimanawa Food Market
Karamjit Kaur
JVR Pricecutter
Karen Mills
under
Kbeez Putaruru Ltd
Kbeez Putaruru Ltd
Kelvin Grove Mini Market
Kelvin Grove Mini Market
Ken Chan
Tofu Shop Henderson
Kiran Patel
Gloriana Dairy. Palmerston North
Kirit Patel
Patels Foodmarket, Whangarei.
the
Kirst Soma
Kishor Rupan
Yes
Kiwimart
Kiwimart
Official
Kolotex Fielding Ltd (WN Chamberlain)
Hooked on A Habit, Feilding
Yes
Kolotex Gisborne Ltd (WN Chamberlain)
The Discount T, Gisborne
Kolotex Holdings NZ Ltd (WN Chamberlain)
The Discount T, Lower Hutt
Yes
Kolotex Kilbirnie NZ Ltd (WN Chamberlain)
Kilbirnie The Discount T store.
Kolotex Naenae NZ Ltd (WN Chamberlain)
The Discount T Upper Hutt
Information
Kolotex Newtown NZ Ltd (WN Chamberlain)
The Discount T Newtown
Kolotex Porirua NZ Ltd (WN Chamberlain)
The Discount T Porirua
Kolotex Riccarton NZ Ltd (WN Chamberlain)
The Discount T Riccarton
Krhitig Gupta
Kulraj Singh
Dairy264
Kunal Saluja
Allenton Foodmarket
Lalit Patel
Juliet Superette Pakuranga
Lance Kennett
The Bul ring
Langley Mini Market
Langley Mini Market
Act
Limbrick St Maxi Mart
Limbrick St Maxi Mart
Lingsu Deng
Duncan St Dairy. Whanganui
Liz Annette
GAS Maratei
1982
Liz Watson
GAS Maratei
Mahendra Patel
Cheltenham General Store
Mahesh Lathiya
Mahesh Patel
Kaiapoi Super 7
Mamonur Rahman
Bairds Road Pricecutter
Manav Sharma
McLean Park Store, Napier
Manav Sharma
McLean Park Store, Napier
Manhar Patel
Summerhays Corner Superette
60 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Oral
Category / Name of submitter
Organisation
submission
Marfell Superette
Marfell Superette
Max Wholesale Foodmarket
Max Wholesale Foodmarket
Midhurst Dairy
Midhurst Dairy
Released
Mirrabooka Superette
Mirrabooka Superette
Mihir Patel
Minesh Patel
The Corner Dairy, Papakura
Mitchell Horner
Mitesh Shankarbhai Chaudhari
Mobil Dannevirke
Mobil Group
Mobil Taihape
Mobil Group
Mt Victoria Food Market
Mt Victoria Food Market
under
Narendra Somabhai Patel
Whenuapai Mini Mart
Naresh Patel
Natu Patel
Jayna Superette
Nelson Chamberlain
New Zealand Tobacconist
Nilesh Shah
convenience store in Auckland CBD.
the
Ninad Joshi
The Bulls Superstore. Bulls
Nirmal Kaur
Cee Jaes 278 Shakespear St
Niteen Patel
Westshore Corner Store Napier
Official
Nolantown Dairy
Nolantown Dairy
Opunake Discounter
Opunake Discounter
Palak Nayak
Yes
Palak Zaveri
Park Store Hawera
Park Store Hawera
Information
Parth Patel
NZ Convenience Store
Parul Patel
Sherwood Superette, Browns Bay
Patea Dairy and Food
Patea Dairy and Food
Pengkun Luke Liu
Thirsty Liquor Northcote
Penny Chen
Pooja Sharma
Pradip Patel
Rewa Dairy. Palmerston North,
Prakash Patel
Pranav Patel
South End Mini Market. Marton,
Act
Prasant Patel
Mananui Dairy, Whakatane
Pratiksa Patel
Redberry Supermarket, Cambridge
Pravin Dahya
Winsford Superette
1982
Pushpa Moore
Queen Drive Dairy
Queen Drive Dairy
Queen Mart City
Queen Mart City
Raj Kingra
Store
Raj Kumar
Raj Modi
Raj Patel
Hillpark Superette
Rajeshbhai Gopalbhai Patel
Tui Superette, Kaikohe
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 61
Oral
Category / Name of submitter
Organisation
submission
Rakesh Sunni
Rakesh Umar Kirithumar Raval
Merivale Superette
Raman Kaur
Released
Ramesh Patel
Porchester Road Superette
Rangiora Mini Market
Rangiora Mini Market
Ravi Patel
Onekawa Store, Napier
Raviesh Dhillon
Ravji Patel
Connifer Grove Dairy
Rendezvous Dairy
Rendezvous Dairy
Rima Naidu
Kennedy Road Dairy, Napier
Rinkesh Pater
under
Ripal Patel
Alfiston Road Diary
Rishab Bharewaj
Gilbert Road Superette
Ritesh Kapadia
Tawa Foodmarket, tawa
Robin Young
Rocky Cao
Discounter store, New Plymouth
the
Rocky Cao
Discount Specialist Strandon
Rocky's Superette
Roshan Patel
Crofton Downs Dairy
Official
Ruby (Hong) Wu
Rucha Patel
Northland Dairy
Sai Dairy
Sai Dairy
Sai Simram
Sai Simram Ltd
Sai Simran ltd
Sai Simran ltd
Information
Saurav Madan
store
Yes
Saurin Gandhi
Shailesh Vallabh
Yes
Shirish Patel
Tuakau Food Market,
Shital and Jignesh Patel
Windsor Park Store Hastings
Shital Patel
Windsor Park Store Hastings
Shiv Patel
Shiv Patel Enterprises Ltd
Shonit Chandra
Fenton Park Dairy, Rotorua
Shyam Foodmarket
Shyam Foodmarket
Act
Spirit Stratford
Spirit Stratford
Stratford Dairy
Stratford Dairy
Sukhvinder Singh
VIP Superette, Kawakawa
1982
Sukwinder Parmar
Line Road Pricecutter, Glen Innes.
Sunil Kumar
East End Dairy, Kaikohe
Super Liquour New Plymouth
Super Liquour New Plymouth
Suresh Jarem
D Jairam and Sons
Yes
Tao Mo
Tavistock Dairy
Tavistock Dairy
Telstar dairy
Telstar dairy
The Hempstore Aotearoa
The Hempstore Aotearoa
Yes
62 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Oral
Category / Name of submitter
Organisation
submission
Thien Quang Huynh
Thomas Pul upparambil
Tim Jeffs
Released
Tinkal Morar
Weymouth Superette
Top Cook Dairy and Takeaway
Top Cook Dairy and Takeaway
Tracy Jiang
Elles Rd Mini Market
Trilochan Singh
Xpress Mark, Flat Bush
Trupti Patel
Gemini Dairy
Tucker Box
Tucker Box
Tushar Patel
Sunnybrae Dairy
Yes
Tusharkumar Limbachiya
under
Umesh Patel
Southgate Superette, Takanini
Viral Patel
Marten Ave Superette
Vivek Ganta
Waipukuarau Store
Waipukuarau Store
Wei Lin
the
West End Store
West End Store
William Kweon
Willis Street Superette
Willis Street Superette
Official
Windmill Dairy
Windmill Dairy
XiaoJuan Wang
Discounter Stores, Auckland
Xin Tan
Yang El en Zhang
Yining (Ivan) Wan
Information
Yogesh Patel
Te Puke Street Food Store
Yogihari Retail Ltd
Yogihari Retail Ltd
Yuan Wang
Thirsty Liquor Glenfield
Yun kim
The Point Store
Zan Li
Z Star Ltd and Vigor Brown Store
Yes
Individual submitters
Aaron Sowry
Greg Sutherland
Mike Kuzman
Abby Poole
Gregory Jackson Ertel
Minette Hanekom
Act
Abilash Thomas
Gregory Price
Miriam Gabriel
Adam Porter
Gretchen Wade
Mitchell Bocking
Adam Smith-Holley
Halle Mitchel
Monica Higgins
1982
Adam Timmins
Hamish Lawson
Morag du Bois
Adrian Borrowdale
Hamish McCrae
Morgan Pritchard
Adrianne Swinburn
Hamish Quigg
Mukesh Chhika [+Oral]
Aengus C
Hannah M Parker
Nancy E Loucas
Agnes Walker
Harlen Wilkinson
Nancy Peters
Ahmad Alzahrani
Harrison Ross
Narelle Constable
Aiden Curtis
Harry Hughes
Narendra Somabhai Patel
Aimee Bradley
Harshad Patel
Natali Manic
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 63
Individual submitters
Alan Bromley
Hayden Hughes
Natasha Broadley
Albert Soek
Hayden Ross Bacon
Natasha Judd
Alexander Bukh
Healther Mccarthy
Nathan Andrews
Alexandra Hickman
Heidi Greig
Nathan Brown
Released
Alexandra Smith
Helen Beswick-Cousins
Nathan Cowie
Ali Sarabi
Helen Kourounis
Nathan Grey
Alicia Goss
Helen McCaul
Nathan Krutz
Alison White
Hendrik van der Merwe
Nathan Ward
Allan Rapley
Henry Bacon
Natu Patel
Amanda Bennett
Hinemoa Macpherson
Neale Cooper
Amanda Casey
Hiren Vather
Neil Briscoe
Amanda Chisholm
Hitesh Kumar
Neil Riley
under
Amanda Dodd
Holly Johnson
Neil Rossin
Amanda Donald
Hori Meilak
Nelson Chamberlain
Amanda High
Ian Hutcheson
Nerissa Hawkins
Amanda Lipsham
Imogen Adolph
Nicholas Fletcher
Amanda Martin
Irene Tufuga
Nicholas Hannan
the
Amanda Roberts
Ireni Ireni
Nicholas Reid
Isabelle Finau Sepi jnr Tufuga
Amber Bishop
Braddick
Nicholle Nicholle
Official
Amber Cordy
Isabelle Stacey
Nick Greene
Amber Rutledge
Ivan Cul pitt
Nick Wiles
Amera Morrigan
Izak Townsend
Nicola Hainton
Amii Pritchard
J Chanesman
Nicola Mangos
Amy Jackson
Jacinta Trounson
Nicola McDermott
Information
Anahera Horomona Tuhou
Jack Jesson
Nicole Ritchie
Andrea Mcewan
Jack Smith
Niki Ash
Andrea Santos
Jack Toepfer
Nikke Fernie
Andrea Thompson
Jackie Liggins
Norman Alexander
Andrew Keehan
Jackson Taylor
Norman Scott
Andrew Keesing
Jacob Clarke
Nur Gencel
Andrew Ralph
Jacqui Kilburn
Olly Fatherly
Andrew Rooke
Jacquie Forsyth
Pamela Kapila
Andrew Thompson
Jaimee Lisa Brough
Patricia Hall
Act
Andrew Young
Jaimie Horan
Patricia Heem
Angela Burney
Jake Patterson
Patricia Philcox
Angela Hauk-Willis
James Isaac Galbraith
Patricia Soon
1982
Angela Helen Stones
James Pearson
Patrick Copeland
Angela Park
James unknown
Patrick Fruean
Angelique Naoum
Jamie Robinson
Paul Browne
Ann Duncan
Jamie Shearer
Paul Button
Ann Peterson
Jan van Dyke
Paul Fraser
Anna Leslie
Jan Walsh
Paul Haliday
Anne Andrew
Jane Cartwright
Paul Harlow
Annie Mcmullen
Jane Ellis
Paul Leahy
64 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Individual submitters
Ansar Pasha
Jane Mitchell
Paul Martin
Anthony McDonald
Jane Murdoch
Paul Meani
Anthony Ryan
Janice Hill
Paul O'Connel
Anton Mather
Jarod Thompson
Paul Watson
Released
Antonia Wihongi
Jase Smithyman
Paula Hodges
Aramoana de Feu [+Oral]
Jason Clark
Pearce Stephens
Arana Tamepo
Jason Francis
Penelope Scott
Aria Aani Moore
Jason Green
Pengkun Luke Liu
Arnia Tamihana-Simich
Jason Mateni
Peter Cartwright
Aron Bailey
Jason Wright
Peter Foster
Arron Conn
Jay Dempster
Peter Greenwell
Arron Peacock
Jayesh Patel
Peter Kenehan
under
Ashlee Ratcliff
Jazz Leat
Peter Liggins
Ashley Koning
Jen Wiig
Peter Meagher
Atil a Erbasan
Jennifer Bennett
Peter Melrose
Axel Salsmark
Jennifer Byrn
Peter Reddaway
Ayden Collins
Jennifer Chatfield
Phil Lattaney
the
Azriel Ritchie
Jennifer L McGinnis
Philip David Hunter
Barbara Edwards
Jennifer Sage
Philip de Weck
Official
Bedette Van Wyk
Jenny Buckley
Philip Gowers
Ben Pritchard
Jess Sternbeck
Philip Greshoff
Bernie Disney
Jesse Arnold
Philip Hamilton
Beth Jenkinson
Jessica Brunn
Philip Hardman
Bethany Hughes
Jessica Lim
Philip Simpson
Beverly Hunter
Jessica Mayfield
Phil ipa Bourke
Information
Bhrent Bingley
Jessica Short
Pippa Edwards
Bhupen Patel
Jessica Tasker
PT
Bodie Hutchinson
Jewel Peters
Quinton Satchell
Bodie Newman
Joan Evans
Rach Mac
Bohdan Palatchie
Joanna Wild
Rachael Courtney
Bonnie van der Bult
Joanne Blue
Rachel Morse
Boudewijin Boogaard
Joanne Thornton
Rachel Smithers
Boyd Hicks
Jodi Henry
Rade Naumoski
Bradley Keith
Jodie Wiseman
Rakesh Umar Kirithumar Raval
Act
Bradley unknown
Jody Barber
Ralph Kohi
Brady Sharrett
Joel Haydon
Ray Burns
Brenda McGregor
Joella Allcott
Rebecca Gilbert
1982
Brenda Tuffery
Joey McIsaac
Reefe Hinga
Brendon Cameron [+Oral]
John Alexander Campbel
Reilly Gardner
Brendon Edward Hoare
John Coppin
Relay for Life
Brendon White
John Eddy
Rian Swart
Brent Bary
John Elliott
Richard Butterfield
Brent Carter
John Finnie
Richard Dykes [+Oral]
Brent Wallace
John Hornyak
Richard Hain
Brett Redwood
John Lear
Richard Holmes
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 65
Individual submitters
Breviss Wolfgramm
John Russell
Richard John Emery Fry
Brian Johnson
John Stella
Rick Webster
Brian Peters
Johnny Meeuws
Riki Trevor Huntley
Brian Thomas
John-Paul Dimmers
Riley Powell
Released
Brian Walters
Jonathan Deane
Rob Cross
Bridget Forsyth [+Oral]
Jonathan Geoffrey Hal
Rob McCardle
Bridget Lummis
Jonathan Lole
Robert Beaglehole & Ruth Bonita
[+Oral]
Brock Ferrar
Jordan Whibley
Robert Bell
Brodie Whitnell
Jordan Whycroft
Robert Cleary
Bronson Wairau
Joseph Clifford
Robert Todorovski
Bronson Wharehinga
Joshua Dutton
Robyn Berry-Luke
under
Brooke Anderson
Joshua Leask
Robyn Ede
Bruce Cross
Joshua Marseden
Robyn Leatrice Gallagher
Bruce Macdonald
Joshua Moa
Robyn Sayer
Bud Wagstaff
Joshua Morris
Rodney Comer
Caela Adams
Judy Stevens-Morehu
Rodrigo Souza
the
Caitlin Harvey
Julian De Fresne
Roger McPherson
Call Morrison
Julian Emsley
Roimata Mangu
Callum Mason
Julian Morrel
Romayne Helen Mcdowell
Official
Calum Reardon
Julie Hicks
Ron Ron Swenson
Cam Lockie
Justin Honey
Ronald Hey
Cameron Rutten
Justin Liao
Ronald Liew
Campbell Lange
Justin Timms
Ronni Cullen
Candace Bagnall [+Oral]
K Lee
Rosemary Seddon
Information
Candice Robb
Kahu Pekepo
Ross McKay
Carl Arundel
Kane Hughes
Ross Soroka
Carl Dale
Kara Cowin
Roy Ratahi
Carl Mann
Karandeep Singj
Rudie Pansegrauw
Carl Van de berg
Karen Carter
Russell M
Carly Gaskel
Karen Horrel
Ryan Andrews
Carmen Gregan-Ford
Karen Lupe
Ryan Cameron
Carol Board
Karina Huang
Ryan Gibson
Carolyn McKay
Karina Liman
Ryan Harris
Act
Carson Smith
Karolyn Baumann
Ryan Johnson
Casey Annabel
Kasey Calogaras
Ryan Lee
Casey Wallace
Kate Burton
Sacha Kawe
1982
Cass Metcalfe
Kate Fullerton
Saffron Melanie Mason
Catherine Thompson
Kate Marjetich
Sahne Martin
Cedric Tan
Kath Fowler
Sally Liggins
Celina Matila [+Oral]
Katherine Lynch
Sam Turner
Chantel Smolenski
Katherine Russel
Samantha Barry
Chanttal Lewis
Katherine Zibell
Samantha Copeland
Charles Boston
Kathleen Fay Pile
Samantha Kirikiri
Charles Nicholson
Kathrynne Stokes
Samantha Politi
66 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Individual submitters
charmaine pompey
Katie Burrows
Samathan Symlie
Chedyn Beach
Kayla unknown
Sandra Wood
Cherie Murphy
Kayle Stevens
Sara Christiansen
Cherry Morgan
Kayley Pania Mitchell
Sarah B
Released
Cheryl ford
Kayne Metcalfe
Sarah Barwise
Chester De Castro
Kegan Develin
Sarah Cunningham
Chet Ankumar Amrutbhai Patel
Keith Oliver
Sarah Harkness
Chirag Makwana [+Oral]
Keith Wallace
Sarah Melissa
Chirag Shah
Kelly Tattersall
Sarah Scott
Chris Andrews
Ken Chan
Sarah Webster
Chris Barwise
Ken LotuI'inga
Saskia Zwanikken
Chris Bold
Kera Gifkins
Scott Clarke
under
Chris Clarkson
Kerri Kilner
Scott Fraser
Chris Dearsley
Kerrine O'Connor
Scott Jones
Chris Verstappen
Kerry Hocquard
Scott Murdoch
Chrissy unknown
Kerry Johnston
Scott Radford
Christian weaver
Kevin Dsouza
Sean Bardwel
the
Christie Cooper
Kevin Parsons
Sean Hillgrove
Christina Corbett
Kevin Wilton
Sean Hsin-Shyuan Lee
Official
Christine Anderson
Kieran Richard Smith
Sebastian Lloyd
Christine Francks
Kim Ashford
Shae Ryder
Christine Hemming
Kim Han
Shalonne Scobbie
Christine McLean
Kim Lingham
Shane Bradbrook [+Oral]
Christine Pike
Kim Oakley
Shane Comber Froggatt
Christopher McMeekan
Kim Papesch
Shane Pratt
Information
Christopher Koenig
Kim Powell
Shane Purcell
Christopher lee archer
Kiona Graham
Shannon Anderson
Christopher smith
Kirsty Alty
Shannon unknown
Chrystal Smith
Kirsty Jones
Sharon Coomber
Claire Doole
Koko Tuffery
Shaun Sweet
Claire Hynd
Kris Anton
Shaun Vukic
Clare Parry
Kristcyn Knipe
Shawn Laurence
Clive Bates [+Oral]
Kristian Pilgrim
Shawn Whitworth
Colin Villiers
Kristie McGoldrick
Shayna Coleman
Act
Coral Graham
Kristy Dench
Shayne Walker
Courtnery Carter-Smith
Kurt Harrison
Shelly Elvin
Craig Alan Dawson
Kye Frank
Sheree Patmore
1982
Craig John Dance
Kylie Garrad
Sherrill Lewis
Craig Kilpatrick
Lamees Ramahi
Sheryl Olsen
Craig Milne
Lana Hunter
Shinay Maraea
Csandra Ogle Plunkett
Lane Berghan
Shiva unknown
Cynthis sideris
Lannden Bower
Shreya Rao
Daisha Zilionis
Laura Ewing
Sian Burgess
Daisy Dee
Lea Tolofua
Sidney Sampson
Daisy Walker
Leann Cartwright
Simon Henwood
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 67
Individual submitters
Dale Foote
Lee Taylor
Simon Stack
Damelza Elizabeth Burnard
Lee Teoh
Simon Watkins
Damien R
Leigh Williams
Sina T
Damon Cooper
Leila Kleyn
Sonali Dutt
Released
Dan Collett
Leitu Tufuga [+Oral]
Sonja Maitland
Dan Walsh
Leon Monastirski
Sophie Carty
Daniel Akast
Lesley smith
Sophie Hancock
Daniel Anderson
Levi Hines
Sophie Stickland
Daniel Farr
Levi Ryan
Stacey Girardin
Daniel Frances O'Toole
Lewis Lewis Read
Steohine Joffrin
Daniel Milne
Lexi-Jayy-jack Critchley
Stephan Butler
Daniel Reid [+Oral]
Linda Buxton
Stephanie Winkler
under
Daniel Spearpoint
Linda Lavin
Stephen Christie
Daniel Veen
Linda Simonsen
Stephen Galvin
Daniel Vos
Lindsay Zelf
Stephen Phillips-Dargavil e
Daniel Vuksic
Linley Barrett
Stephen Piner
Danielle Grice
Lisa Brady
Stephen Smith
the
Danny Carley
Lisa Cowe
Steve Brad
Darren Alan Johnston
Lisa Graham
Steve Daniel
Official
Darren Johnston
Lisa H Ponga
Steve Dohmen
Darren Smith
Lisa Rutherglen
Steve Hamilton
Dave Hack
Litsa Skepathianos
Steve Munford
Dave Morgan
Lloyd Casey
Steven Brown
David Bowen
Logan Fletcher
Steven Gribble
David Clarke
Lorna Johnston
Steven Mclachlan
Information
David Farrell
Lorraine Hamilton
Steven Smith
David Hunt
Louise Mainvil
Stuart Dally
David Lines
Louise Tawhai
Sudha Bhana
David Miller
Louise Tischendorf
Sue Purgmire
David Moyle
Lucy Kennedy
Sue Sue
David Peter Vitali
Luise Gortz
Susan Gibbling
David Sopper
Luke Baker
Susan Letchford
David Turner
Luke Hardiman
Suscan Ritchards
Dawn Newton
Luna Rivers
Suzanne Bielby
Act
Dean McCondach
Lydia Liew
Suzette Laws
Dean Robb McCondach
Lynne Montgomery
Tabitha Day
Debbie Wallace
M E Miller
Tai Faalogo
1982
Debra Pugh
Maddison Booth
Tamara Schliebs
Declan Tessier
Maggie Prentice
Tanan Zorigt
Dee Carter
Mahmoud Adbuo
Tania Eliason
Del Logan
Mansukhbhai Patel
Tania Lumb
Denise Drendel
Manu Dodd
Tayla Harris
Destri Head
Mara Schneider
Te Rukutia Tongaawhikau
Dharmesh Jarem
Marc HenryWright
Teaio Maki
Diana Orvan
Marcella Angela Dodanis
Terijoy Wilton
68 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Individual submitters
Diane Prescott
Marcus Ohlson
Terri Ellmer
Dianne Morison
Marcy McCormick
Terri-Mae Fitzgerald
Dierdre Legg
Maree Candish
Thomas Bell
Dion Bly
Marewa Glover [+Oral]
Thomas Power
Released
Dominic O'Grady
Margaret Dinnan
Thomas Russ
Dominic Ritchie
Marian Bartram
Tiarna Lee Hopkinson
Donna Hancock
Marie Hart
Tiata Malosi
Donna Morpeth
Marie Wilkins
Tiffany Cresswel
Donna Torrie
Marion Freimuth
Tim Childs
Dr Allan Wyllie
Mark Broadway
Tim Hinton
Dylan Mama
Mark Francis McElhinney
Timothy Moffitt
Ed Red
Mark Green
Timothy Stewart
under
Edward Rhind
Mark Hodgson
Timothy Wilson
Edward Wil iams
Mark Linton
Tineke Tu
Eiizabeth Burdett
Mark O'Neill
Tirika Adam
Eileen Brown
Mark Paterson
TJ Bishop
Elaine Epiha
Mark Schofield
Tom Johnson [+Oral]
the
Eli Buck
Mark Swaney
Tom Kim
Eli Sherwood
Marko Peselj
Tom Morawetz [+Oral]
Official
Elizabeth Nairn
Marshal Ashworth
Tony Hulme
Elizabeth Prosser
Martin Witt
Tony Johnson
Elizbeth Belshaw
Mary Evans
Tony Lawson
Emily Fleet
Mary Karas
Tony Le Ross
Emily McKenzie
Mary Martik
Traa Riarn
Emily Thomsett
Mary-Anne Macaskil
Tracey Collins
Information
Emma Griffin
Mathew Josland
Tracey Waho-Blayney
Emma Louis
Matilda Fry
Tracy Carrol
Eric Lim
Matt Honeycombe
Tracy Michel
Erica Harland
Matthew Bird
Trent Keith
Ernest Munro
Matthew Boswell
Tricia Warren
Eve Bon
Matthew Cathro
Trish Bostad
Faran Gil banks
Matthew Driscol
Trudi Hill
Fiona McNair
Matthew Emson
Tui Ziarno
Florence Pirotais
Matthew Perpere
Tyler McKinley
Act
Frances Bish
Matthew Rogers
Tyler Sands
Francesca Sullivan
Matthew Rutherglen
Venessa Minkley
Frankie Lee Matthews
Maureen Farr-Cross
Vickie Geraghty
1982
Freison Mitchell
Maureen Hall
Vicky Noble
Freya Noel Barnes
Maxine Gunning
Victoria Findlay
Gail Cannan
Maxine Lynch
Victoria Walters
Gareth Miles
Meghan Kirk
Vincent Xie
Gareth Ollerenshaw
Melanie McFarlane
Vincent Zeng
Gareth Pritchard
Melanie Yardley
Virginia Smith
Gareth Robinson
Melinda Sigley
Vivian Murphy
Garry Farmer
Melissa Fergusson
Wade Maxwell
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 69
Individual submitters
Garry Grant
Melissa Jull
Warren Hartley
Gary Grive
Melissa Reynolds
Warrick Carter
Gaurang B Patel
Melissa Stead
Warwick Carter
Gavin Christensen
Melody Tulaiu
Wayne MacKinlay
Released
Gavin Rae
Michael Boylen
Wayne Ritchardson
Genevieve Ussher
Michael Crook
Wendy Carew
Geoffrey Dawson
Michael Francis Caton
Wendy Stratton
Geoffrey Mairs
Michael Gabbitas
William Alexander
George Barlis
Michael Gartner
William George Frewin
Geraldine Atkins
Michael Hoy
William Gianoncelli
Gerri Brown
Michael Sibbes
William Shankar
Ghia M
Michael unknown
William V
under
Glenn Henson
Michaela Billings
Wymond Gripp
Glenn William Anderson
Michaela Henderson
Yang El en Zhang
Gordon Stuart
Michele Mullins
Yolande Jaffares [+Oral]
Grace Murdoch
Michele Nimmo
Yuan Wang
Graham Peters
Michelle Dayman
Yvonne Menary
the
Graham Slattery
Michelle Gibson
Zachary Adams
Grant Hewison
Michelle Mollard
Zane S
Official
Grant Mason Daubney
Michelle Taylor
Zara Jasmine Cole
Greg Davies
Mikayla Prosser
Zorawar Singh Ahluwalia
Eliana Golberstein Rubashkyn
Greg Simpson
Mike Burke
[+Oral]
Information
Act 1982
70 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Appendix 2: The Ministry’s views on the risks and
benefits of vaping products
Submitters presented a wide range of often conflicting evidence to the Committee on the evidence
for the risks and benefits associated with vaping.
Released
The Ministry’s position is that vaping products can benefit smokers, especial y those who are unable
to quit smoking using conventional means. However, they are not risk free. In particular, the risks
associated with long-term vaping are not yet ful y known. For young people who have never
smoked, there are likely to be some health risks associated with regular long-term vaping.
Harm reduction and support for smoking cessation
under
The many toxins in tobacco smoke, rather than the nicotine, are responsible for most of the harm
associated with tobacco use. Vaping (and smokeless tobacco products) do not combust.
The Ministry is satisfied that vaping is significantly less harmful than smoking and is an appropriate
alternative for smokers who wish to reduce the smoking-related harm to their health and those
the
around them. There also is evidence to show that vaping can help people to quit smoking.
Official
Vaping flavours
Many submitters raised concerns about the restrictions on the sale of vaping product flavours by
generic retailers.
There is no strong evidence that particular flavours are important for smoking cessation, but there is
evidence to suggest that flavours add to the appeal of vaping. For smokers, especial y those who are
Information
unable to quit by conventional means, having a product available that is less harmful and at least as
satisfying as smoking is important.
Some studies have highlighted concerns that some flavours are appealing to young people and may
be a factor in youth uptake of vaping.
A summary of the evidence on the use of vaping flavours was provided to the Committee on 9 April
2020 as part of the Ministry’s supplementary briefing.
Act
Vaping product safety
While it is much less harmful to vape than it is to smoke, there are inherent risks associated with the
use of vaping products. These risks relate primarily to the toxicants present in products, however,
there is also some risk associated with malfunctioning devices.
1982
These risks can be mitigated through the product safety requirements, which will be set by
regulations (eg, for manufacturing standards, prohibitions on harmful ingredients, maximum
nicotine concentration and child-resistant closures).
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 71
link to page 78
Potential impact of vaping on children and young people
There is some concern that experimentation may lead to regular vaping and then to smoking (ie,
vaping acts as a gateway to smoking). However, there is no robust evidence to support this concern.
Two major reviews were published in 2018 that addressed this issue. The National Academy of
Sciences in the United States (which was cited by a few submitters as the best source of evidence)
Released
and Public Health England (which has subsequently updated its review) considered the same
evidence and concluded that there is an association between ever vaping and ever smoking at a later
point in time. Both reports acknowledged that the studies included in the reviews had limitations
and that it is not possible to conclude that vaping causes smoking.
A summary of the data on youth vaping is provided as Appendix 3.
under
Vaping during pregnancy
The Committee has expressed interest in the impacts of vaping in pregnancy, and several submitters
have provided supplementary evidence.
The Ministry’s advice is that it is best to have a smokefree and nicotine-free pregnancy (we know
the
that nicotine can have some adverse effects in pregnancy). However, for women who are struggling
to stop smoking, the use of nicotine replacement therapy (NRT) during pregnancy is appropriate, as
use of these products is associated with significantly less harm than continuing to smoke. The
Official
addition of support, such as that provided by stop-smoking services, is currently the most effective
way to quit smoking.
However, for women who cannot quit using conventional methods, switching to vaping and stopping
smoking, is an appropriate action.
Information
Estimates of lives saved
The Committee has expressed interest in whether there are any estimates for the number of lives
saved by vaping. A 2019 study by researchers at the University of Otago found that New Zealand’s
approach of al owing wide access to e-cigarettes would be likely to result in overall health gains and
cost savings for the health system.
The research modelled the impact of liberalising the sale of vaporised nicotine products, such as e-
cigarettes, compared with a situation where the uptake of these products was very low, as in New
Zealand in 2011.
Act
The study, published in the international journal
Epidemiology,
8 found that liberalising vaping
products would lead to a gain of 236,000 quality-adjusted life-years for the general population and
savings of NZ$3.4 bil ion over the remaining lifetime of these people – a sizeable gain in health for
1982
the New Zealand population and an important benefit in terms of lower health costs.
8 Petrovic-van der Deen FS, Wilson N, Crothers A, et al. 2019. Potential country-level health and cost impacts of
legalizing domestic sale of vaporized nicotine products.
Epidemiology May, Vol 30(3), 396–404. URL:
https://journals.lww.com/epidem/Citation/2019/05000/Potential_Country_level_Health_and_Cost_Impacts_
of.14.aspx (accessed 11 May 2020).
72 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Appendix 3: Youth vaping (14- to 24-year-olds)
Data on youth vaping is reported from the ASH Year 10 Snapshot survey, the Youth Insights Survey
(YIS), the Health and Lifestyle Survey (HLS) and the New Zealand Health Survey (NZHS). The most
recent vaping data from these surveys is summarised in the table below.
Released
The Youth19 Rangatahi Smart Survey (Youth19) is new and has only been completed once so does
not provide any trend data unlike the other surveys.
Table 2: Summary of the findings from the youth vaping surveys
YIS
ASH Year 10
Youth19
15- to 17-year-
15- to 24-year-
Snapshot
olds in the
olds in the HLS
2018/19 NZHS*
Survey
2018
2019
2019
2018/19
2018
under
year
Sample
Nationally
Census style
This is not a
Subgroup of a
Subgroup of a
representative
survey;
nationally
nationally
nationally
survey; 2,689
27,354 year
representative
representative
representative
year 10 students 10 students
survey. It
survey.
survey.
the
aged 14 and 15
aged 14 and
surveyed 7,700
years.
15 years.
adolescents (13-
to 18-year-olds)
Official
from Greater
Waikato,
Auckland and
Northland
regions.
Information
% of those 38%
37%
38%
25%
46%
who have
tried
vaping
% of those 8%
12%
10%
3%
7%
who vape
at least
once a
month
Act
(current or
regular
vapers)
1982
% of those Not reported
Not reported 6%
–
–
who vape
at least
once a
week
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 73
YIS
ASH Year 10
Youth19
15- to 17-year-
15- to 24-year-
Snapshot
olds in the
olds in the HLS
2018/19 NZHS*
% of those 2%
3%
Not reported
2%
5%
who vape
Released
daily
Those
Māori
Not reported Males
–
–
more likely
to be
Māori
current
Students
vapers
attending
medium- to
high-decile
under
schools
Those
Sample size not
Males
Not reported
–
–
more likely enough
to be daily
Gender
vapers
Diverse
the
Māori
Pacific
Official
peoples
Students
attending
low- and
medium-
decile schools
Information
Vaping by
Among never
Fewer than
About two-thirds -
-
smoking
smokers, 2.1%
1% of never
of ever vapers
status
are current
smokers
(65%) and 48%
vapers and 0.5% were daily
of regular vapers
are daily vapers. vapers.
had never
smoked
Current
cigarettes. The
smokers
survey did not
were more
report about
likely to try
Act
daily vaping.
vaping and
vape
regularly.
1982
*Information for 15- to 24-year-olds is also available from the NZHS.
Points to note:
• Al surveys had similar questions on frequency of vaping and smoking.
• ASH Year 10 Snapshot data suggests that daily vaping is more common among students
attending low-decile schools in New Zealand, but Youth19 data says that current vaping is
more common among medium to high decile school students in Greater Waikato, Auckland
74 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
and Northland regions. The 2018 YIS data did not show any significant differences by school
decile group (but the numbers were high in low-decile schools).
• Reporting of vaping prevalence by smoking status is different. ASH Year10 Snapshot and the
YIS data say ‘Out of never smokers, _% are daily vapers’, whereas Youth19 data says ‘Out of
ever vapers and regular vapers, _% had never smoked cigarettes’.
Released
• The YIS was the only survey that asked participants if their last vape contained nicotine.
ASH Year 10 Snapshot survey
The ASH Year 10 Snapshot survey of between 20,000 and 30,000 14- to 15-year-olds (year 10
students) has been carried out annual y since 1999. It has been col ecting data relating to the use of
e-cigarettes (vaping) since 2015.
under
Key findings
Key finding of the 2019 ASH Year 10 Snapshot survey were as fol ows.
• A total of 3 percent of year 10 students reported using e-cigarettes daily.
the
• Fewer than 1 percent of year 10 students who never smoked reported using e-cigarettes
daily.
• Over one-third of year 10 students reported trying an e-cigarette (even a single puff or
Official
vape).
• Students who smoked were almost four times more likely to have tried an e-cigarette (even
a single puff or vape) than students who never smoked.
Comment
Information
These results coupled with the decline in daily smoking over the same period (2015–2019), indicate
that vaping may be displacing smoking. Daily use is important to monitor as a sign that young people
are dependent on vaping. Although students are experimenting, daily use remains low. Daily
smoking in this group has been declining since 1999 as shown in the graph below.
Act 1982
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 75
 Released
under
Released
under
Source: 2019 ASH Year 10 Snapshot, Topline Results – Smoking 2019 factsheet
the
Official
Information
Source: 2019 ASH Year 10 Snapshot, Topline Results – E-cigarettes and vaping 2019 factsheet
Act 1982
76 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
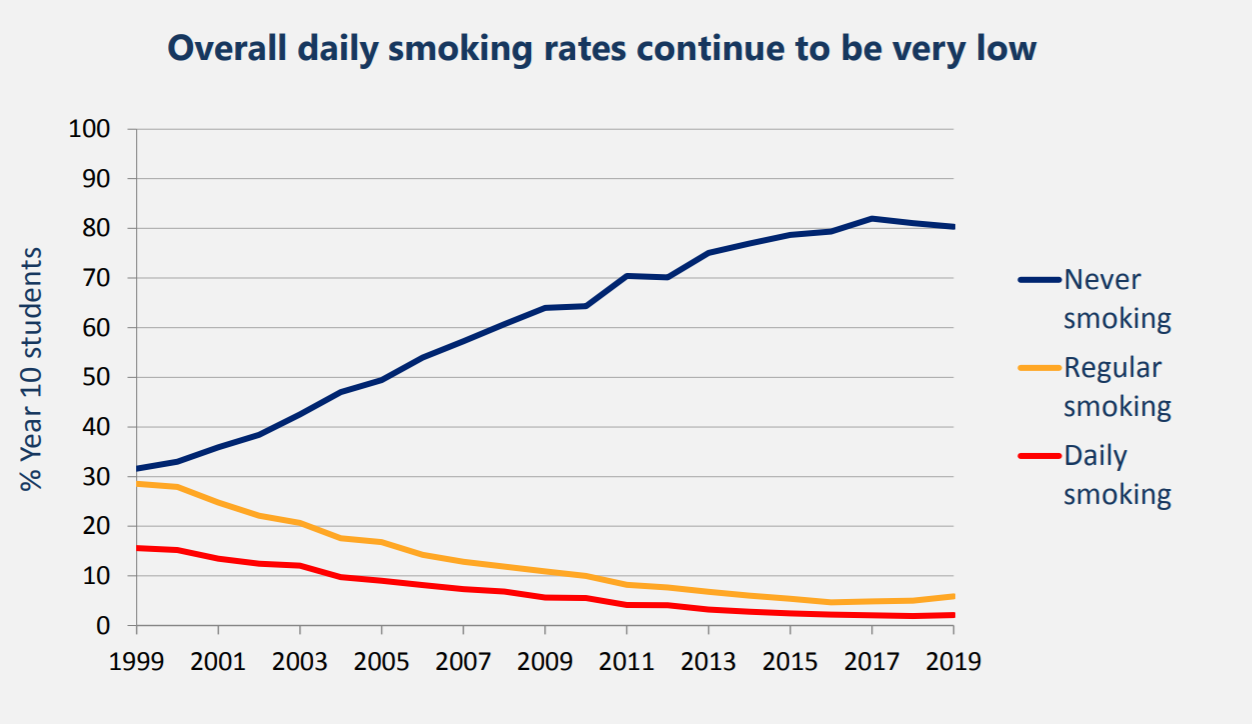 CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Released
under
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Released
under
Note: Regular e-cigarette use is described as students who report e-cigarette use either daily, weekly or monthly.
the
Source: 2019 ASH Year 10 Snapshot, Topline Results – E-cigarettes and vaping 2019 factsheet
Official
Information
Act
Note: Regular smoking is described as students who report smoking either daily, weekly or monthly.
Source: 2019 ASH Year 10 Snapshot, Topline Results – Smoking 2019 factsheet
1982
Youth Insights Survey
The YIS has asked year 10 students about ‘ever use’ of e-cigarettes since 2012. The YIS is conducted
every two years and also asks about smoking.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 77
Key findings
Vaping
The YIS found that while the proportion of students who had tried e-cigarettes (vapes) increased
between 2016 and 2018, the proportion of students vaping daily (daily vapers) remained low. The
results show that ‘ever use’ has been steadily increasing over the years. In 2012, 7.1 percent of
Released
participants said they had tried an e-cigarette. By 2018, this figure has increased to 36.9 percent.
In 2018:
• 38 percent of students had tried vaping (ever vapers), up from 29 percent in 2016 and
20 percent in 2014
• 1.9 percent of students were daily vapers, compared with 0.7 percent in 2014
• 8 percent of students vaped at least monthly (current vapers), up from 3 percent in 2014
(The increase in current vapers was seen across most demographics.)
under the Official Information Act 1982
• Māori were two times more likely to be current vapers than non-Māori.
The top reasons current vapers gave for vaping were that they liked the flavours/taste and they
enjoyed vaping with their friends (both 59 percent).
Among current vapers:
• over half (54 percent) did not vape nicotine (only flavour)
• 23 percent vaped nicotine (with or without flavour)
• 5 percent did not know what their device contained
• 12 percent did not give any response.
Smoking
The YIS found that the proportion of students who were smoking cigarettes at least once a month
(current smokers) in 2018 had not changed since 2016. In 2018, around 1 in 20 (5 percent) 14- and
15-year-olds smoked at least monthly (current smokers), unchanged from 2016 and down from 2012
(7 percent).
Those more likely to be current smokers were:
• Māori (2.5 times more likely than non-Māori)
• Pacific peoples (2 times more likely than non-Pacific peoples)
• attending low-decile schools (2 times more likely than those attending high-decile schools).
Health and Lifestyle Survey
The HLS is a biennial face-to-face in-house survey of New Zealanders aged 15 years and over that has
collected information on e-cigarette use since 2014. In this appendix, we focus on young people
aged 15- to 24-years-old.
78 Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report
CONFIDENTIAL UNTIL THE BILL IS REPORTED BACK TO THE HOUSE OF REPRESENTATIVES
Key findings
In 2018:
• about 7 percent of 15- to 24-year-olds reported vaping at least once a month (a significant
increase from 2014, when it was 0.1 percent)
Released
• around 5 percent of 15- to 24-year-olds reported vaping daily.
Differences by demographics and smoking status were not observed because of the smaller sample
size for 15- to 24-year-olds.
New Zealand Health Survey
under the Official Information Act 1982
The New Zealand Health Survey (NZHS) is published annual y and reports on e-cigarette use in the
population aged 15 years and over. The NZHS has reported on e-cigarette use since 2015/16. This
appendix focuses on the 15- to 17-year old age group results.
Key findings
• Daily e-cigarette use amongst 15- to 17-year olds increased from 0.1 percent in 2015/16 to
1.7 percent in 2018/19.
• Those 15- to 17-year olds who use an e-cigarette at least monthly increased from
0.6 percent to 3.4 percent.
• Those 15- to 17-year olds who had ever tried an e-cigarette increased from 19 percent to 25
percent.
Comment
The results of this survey show that while many young people are experimenting with vaping (25
percent), only a small percent are vaping on a daily basis (around 2 percent). The results do not show
the percentage of the daily vapers that were smokers or ex-smokers.
Youth 19 Rangatahi Smart Survey
Key findings
Overall, 38 percent of students in the Northland, Auckland and Waikato regions reported they had
tried vaping, while 10 percent reported vaping regularly (monthly or more often) and 6 percent
reported vaping weekly or more often.
Comment
The survey found that nearly two-thirds (65 percent) of students who ever vaped and nearly half
(48 percent) of regular vapers had never smoked cigarettes. This was the only survey included in this
appendix that did not report on daily vaping.
Smokefree Environments and Regulated Products (Vaping) Amendment Bil : Departmental report 79
Document Outline





