
1982
Act
Information
Official
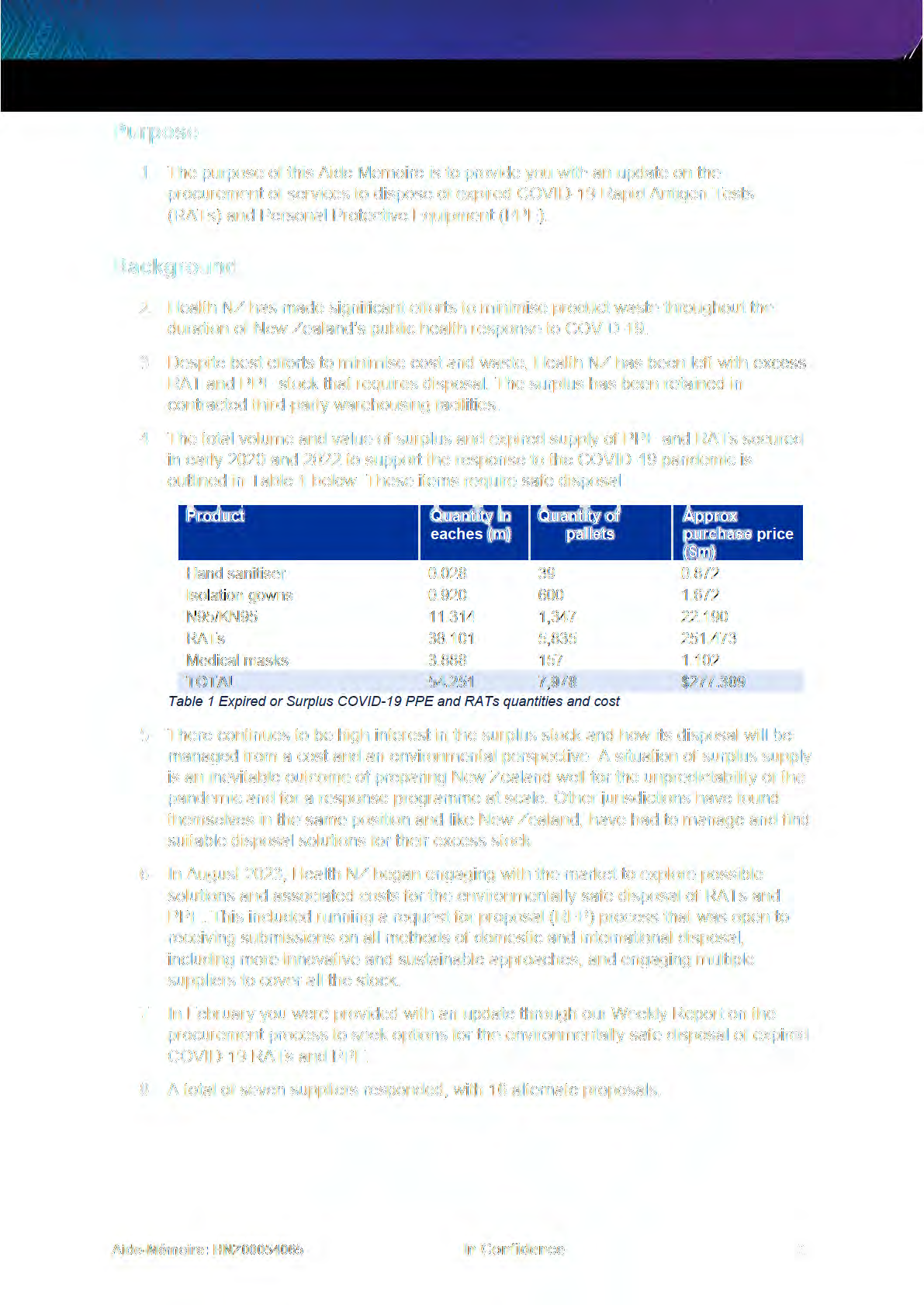
the
under
Released

1982
Act
Information
Official
the
under
Released

 In Confidence
Update on securing a supplier for obsolescence management of
In Confidence
Update on securing a supplier for obsolescence management of
COVID-19 stock
9.
Following a thorough assessment of all submissions and consultation with other
agency partners, New Zealand-based waste management business, Enviro NZ, has
been selected to manage the disposal of the following surplus COVID-19 products:
10. Enviro NZ’s disposal solution uses a combination of three disposal methods to
achieve the most sustainable outcome.
11. Of the 9400 pallets of stock requiring disposal, approximately 56% (5220 pallets) will
be processed into alternative fuels, with the remaining 44% (4180 pallets) to be
disposed of via a method of part recycling, part landfill. In total, an estimated 65% of
the waste will be diverted from landfill.
12. A further explanation of this three-fold approach is explained below:
i.
Co-processing into alternative fuels – once fully operational, 90 pallets per
day will be handled via this method which will see all stock materials disposed of
through the cement manufacturing process. Waste will be co-processed alongside
wood waste and tyre derived fuel to produce thermal energy as a substitute for coal
to make cement clinker.
ii.
Recycling – once fully operational, 72 pallets per day will be handled via a
dual method of part recycling / part landfill, starting with recycling the tertiary
packaging from RATs and PPE stocks. This approach will see storage pallets re-
used, shredded into coloured garden/playground decorative for safety purposes, or
shredded and used as a biofuel; and plastic materials sold for further processing
into slip sheets, plastic decking or similar.
iii.
Landfill – In our commitment to disposing of leftover product materials
responsibly to reduce our environmental impact as much as possible, a New
Zealand based landfill provider with a good track record in sustainable landfill
management has been selected to take remaining waste for disposal. The local
facility is operated safely and sustainably to ensure the surrounding land, air and
water is protected.
13. These disposal methods will be run in parallel to ensure efficiencies and completion
within an expected four-month period. Enviro NZ will commence from the middle of
July 2024.
14. The total costs for disposal will utilise allocated funds for obsolescence management
under the Official Information Act 1982
from the COVID-19 response budget and are expected to total approximately S9(2)
, with an additional cost of S9(2)(b)(ii) for continued warehousing while the
(b)(ii)
solution is implemented. Notably this is commercially sensitive information.
15. The disposal of expired and surplus supplies as a result of COVID-19 will be
completed by end of November 2024. Beyond this date, we do not anticipate any
further requirement for disposal of supplies specific to COVID-19. This is reflected in
our scaled down management of supply since 01 July 2023 to include rotational
stock through hospital services of the 12-week medium pandemic reserve holdings in
Released
the event of a future health emergency or pandemic response. We intend to exhaust
the recently purchased supply of RATs over the winter and early spring period
reflecting these are high usage months, particularly with the prevalence of higher
levels of undifferentiated respiratory illness.
Aide-Mémoire: HNZ00054065
In Confidence
3

 In Confidence
Next steps
In Confidence
Next steps
16. We will continue to monitor the progress of obsolescence management project specific
to COVID-19 supply. PPE and other medical devices used in standard infection
protection and control processes will be managed through normal demand and supply
management processes as part of our quality management of medical devices and
supplies for use in health emergencies.
under the Official Information Act 1982
Released
Aide-Mémoire: HNZ00054065
In Confidence
4







... �
In Confidence
Aide-Memoire
COVID-19 Options for Testing with Rapid Antigen Tests from
01 March to 30 June 2024
To:
Hon Dr Shane Reti, Minister of
Reference:
HNZ00035731
Health
From:
Dr Nick Chamberlain, National
Due Date:
18 January 2023
Director, National Public Health
Service
Copy to:
Security level: Sensitive
Contact for telephone discussion (if required)
£ �-
Name
Position
Telephone
1st contact
Dr Nick
National Director, National Public s9T2>ca>
Chamberlain
Health Service
I
X
Deputy National Director and
Matt Hannant
Director Planning, Purchasing and
Performance, National Public
� 9(2)(a)
I
Health Service
The following departments/agencies have been consulted
ManatO Hauora - Public Health Agency
Aide-Memoire: HNZ00035731: COVID-19 Options for Testing with Rapid Antigen Tests from 01 March to 30 June 2024
In Confidence


In Confidence
COVID-19 Options for Testing with Rapid Antigen Tests from
01 March to 30 June 2024
Purpose
1. This Aide-Mémoire responds to your 8 January 2024 request for the options we
considered for continued community testing using COVID-19 Rapid Antigen Testing
(RATs) for the period of 01 March to 30 June 2024.
2. We are proceeding to procure a small supply of five million RATs within existing
appropriations to continue distribution during March – June 2024 in line with the current
access criteria.
ACT 1982
Background/context
3. A number of public health non-mandatory measures for COVID-19, such as testing,
remain in place and continue to provide an effective approach to managing COVID-19,
including reducing its impact on hospital admission rates.
4. RATs have been publicly-funded as part of the COVID-19 Public Health Response since
February 2022 to support COVID-19 testing in community settings. No further RATs were
secured in the 2022/23 financial year. This was due to high stock levels held within a
centralised supply construct and reduced COVID-19 funding.
INFORMATION
5. Our remaining COVID-19 RATs stock has a March 2024 expiry date (between mid and the
end March). To ensure sufficient time for use, RATs are distributed from our warehouses
with a minimum remaining shelf life of three to four weeks. In line with this practice, we
are therefore unable to distribute any currently-held RATs beyond the end of February
2024.
6. Clinical and public health advice supports continued testing for COVID-19 to enable
access to antiviral therapies (AVTs) and access to RATs for the public and healthcare
sector to ensure the ongoing public health management of COVID-19 outbreaks.
7. In Australia, while approaches differ by state and territory, the Government has
committed to investing an additional $517 million to ensure continued supply of RATs to
priority groups and clinical settings.
8. Information was provided to you on 19 December 2023 (HNZ 00034528 refers) on
COVID-19 Personal Protective Equipment (PPE), vaccine, and diagnostic testing supplies,
alongside forecast requirements over the coming three years. That advice noted that a
small volume of publicly funded RATs will be secured to cover the period 01 March to
30 June 2024.
9. s 9(2)(b)(ii)
10. The number of five million RATs has been derived based on May to October 2023
distribution levels, taking into account a peak demand
RELEASED UNDER THE OFFICIAL for winter and the phasing out of
that peak. This stock would continue to be distributed to Primary Care, public hospitals,
pharmacies (for public distribution), Aged Residential Care, home care, first responders,
the Māori and Pacific distribution network, and others such as family planning.
11. The volumes of RATs distributed to date is higher than what we expect to distribute over
2
Aide-Mémoire: HNZ00035731: COVID-19 Options for Testing with Rapid Antigen Tests from 01 March to 30 June 2024
In Confidence


In Confidence
the March – June 2024 period. This is due to the removal of remaining COVID-19
initiatives such as supplying RATs to travellers arriving at New Zealand borders, ceasing
supply to other government agencies (such as Corrections and Education), and no-longer
offering RATs to non-healthcare providers.
Clinical and Public Health risks if RATs are no longer available
through the Public Health Response
12. We considered clinical and public health risks if free RATs are no longer available.
13. RATs are used for our most clinically vulnerable population groups to meet the Pharmac
eligibility criteria for AVTs. If RATs were unavailable for this group, it would likely create a
barrier to AVTs access, resulting in treatment not being sought (due to not being able to
ACT 1982
test) and increased emergency department presentations and hospitalisation of cases.
14. Community and Primary Care providers may revert to Polymerase Chain Reaction (PCR)
testing, leading to an increase in COVID-19 PCR testing numbers. This would result in a
delay in commencing treatment while awaiting test results and an increase in costs due
to a higher number of PCR samples being processed
.
15. A delay in supporting the management of healthcare workers with respiratory COVID-19
symptoms could result in increased staff absenteeism or delays in return to work as they
await PCR results, and could lead to a significant increase in PCR testing.
16. It would not then be possible to manage cases and outbreaks in high-risk settings such
INFORMATION
as hospitals and Aged Residential Care in a timely and effective manner. This could have
a significant impact in leading to more widespread outbreaks due to the delay in
diagnosis and isolation of cases with an impact on patient flow.
17. It could lead to an increase in the hospitalisation of cases (since people would be unable
to test themselves at home and self-manage) and a possible increase in death rates due
to delayed diagnosis, treatment and management.
Discussion
Options we considered for RATs and Community Testing
18. Alongside the Public Health Agency, we considered three options. It was determined that
Option 1 would best manage the public health risk that the JN.1 variant and the wider
COVID-19 continue to pose for those who are clinically vulnerable within community
settings This option provides an assurance that the risks identified in paragraphs 12 - 17
would be addressed.
19.
Option 1 (proposed) - Purchase a limited number of RATs to enable continued access
for the period 01 March to 30 June 2024. Populations or settings for which this would
enable continued access to free RATs include:
-
The public, including those eligible for AVTs.
-
Primary and Urgent Care, along with community pharmacies.
-
High-risk settings such as public hospitals and Aged Residential Care
RELEASED UNDER THE OFFICIAL
-
Māori and Pacific distribution channels.
20. Option 2 – A smaller number of RATs are purchased and eligibility for free RATS is
limited to those eligible for AVTs. This option would reduce access to healthcare
providers to support cheaper diagnostic testing (e.g. RAT vs PCR), healthcare worker
3
Aide-Mémoire: HNZ00035731: COVID-19 Options for Testing with Rapid Antigen Tests from 01 March to 30 June 2024
In Confidence


In Confidence
testing and testing for outbreaks. This would therefore reduce the ability to manage
onward transmission and outbreaks.
21.
Option 3 – RATs are not publicly-funded or freely available beyond 29 February 2024.
The risks associated with this includes barriers to access AVTs for the most clinically
vulnerable population group in a timely manner, leading to an increased risk of
hospitalisation. It would also increase the volume of PCR testing and therefore increase
laboratory processing costs and turnaround times for COVID-19 results, further delaying
access to AVTs. Healthcare providers would need to purchase their own RATs. Primary
Care, pharmacies and Emergency Departments would become overburdened with
testing and care for undifferentiated respiratory illness. This does not align with clinical
and public health testing advice.
ACT 1982
Next steps
22. We intend to proceed with
Option 1, with the first batch of RATs purchased by Friday 26
January 2024 to enable continuity of supply.
23. These purchases of RATs will be phased to manage obsolescence risk. The expiry date
of this stock will be at least 12 months (possibly longer). If the demand is lower than
expected over the period March - June 2024, we will be able to repurpose the stock
across the health system.
24. We are developing a communications plan regarding continued access to RATs through
to June 2024. This will be shared with your office.
INFORMATION
25. RATs that are unable to be distributed before they expire will be disposed of. There is a
procurement process underway exploring options for the disposal of expired and surplus
COVID-19 stock. We will be in a position to update you once this process is completed.
26. Provision of RATs beyond 30 June 2024 will be subject to demand levels during March-
June, and future budget availability. At this time, we have not made any commitment to
purchase further RATs beyond the end of this financial year. We are working closely with
the Ministry on future options and risks.
RELEASED UNDER THE OFFICIAL
4
Aide-Mémoire: HNZ00035731: COVID-19 Options for Testing with Rapid Antigen Tests from 01 March to 30 June 2024
In Confidence




















